Gross:
Hemorrhage:
Subarachnoid Hemorrhage:
Mild trauma: Delivery is likely responsible for small incidental hemorrhages over the subarachnoid surface of the brain in preterm infants.
Intraventricular hemorrhage (IVH):
Intraventricular hemorrhage is usually the result of germinal matrix hemorrhage or hemorrhage from periventricular leukomalacia. The germinal matrix exists from early gestation to term as a reserve of immature cells that will become neurons and glia. The amount decreases after 33 weeks of gestation. The risk of IVH synchronously decreases with the maturation of the germinal matrix. Hemorrhage in this soft embryonic appearing tissue with prominent thin-walled vessels may occur in response to various risks including hypoxia, cytokines, or suddenly changing vascular pressures. Most hemorrhages are identified after birth. However, intraventricular hemorrhage can be found in previable fetuses at autopsy (Fig 1).
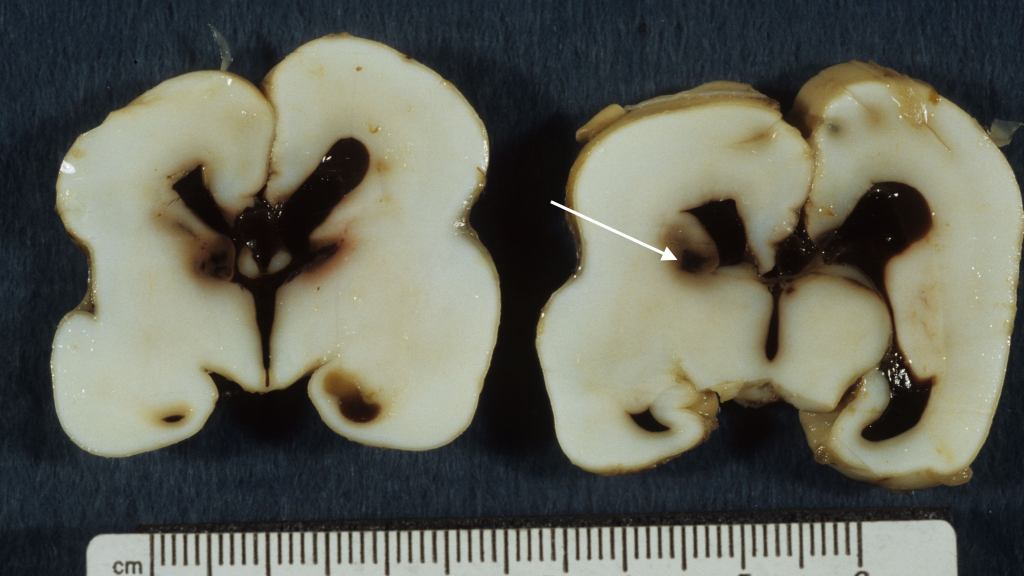
Grossly, in the intact brain, blood in the ventricles often seeps and pools around the base of the brain (Fig 2).
A simple grading system for IVH is: Grade 1: hemorrhage confined to the germinal matrix; Grade 2 hemorrhage into but not expanding the ventricle; Grade3 hemorrhage expanding the ventricle; Grade 4 hemorrhage into the white matter. Since the blood is likely to follow the path of least resistance, these Grade 4 hemorrhages likely flow into areas of periventricular leukomalacia which are also more frequent in more premature infants (Fig 3).

Cephalohematoma:
During birth, the compression around the scalp can produce a caput succendaneum. This is a harmless, usually bloody appearing subcutaneous, edema from compression of cranial veins during delivery. A cephalohematoma can occur with bleeding into the scalp sometimes associated with the use of a vacuum extractor. A subgaleal hematoma beneath the scalp aponeurosis can occur with vacuum extraction, but also with trauma to the underlying bone (Fig 4)

. The amount of blood from such hematomas may be sufficient to cause hypotension in the infant. With a subgaleal hematoma, there is a possibility of parenchymal hemorrhage as well.
Subdural Hematoma:
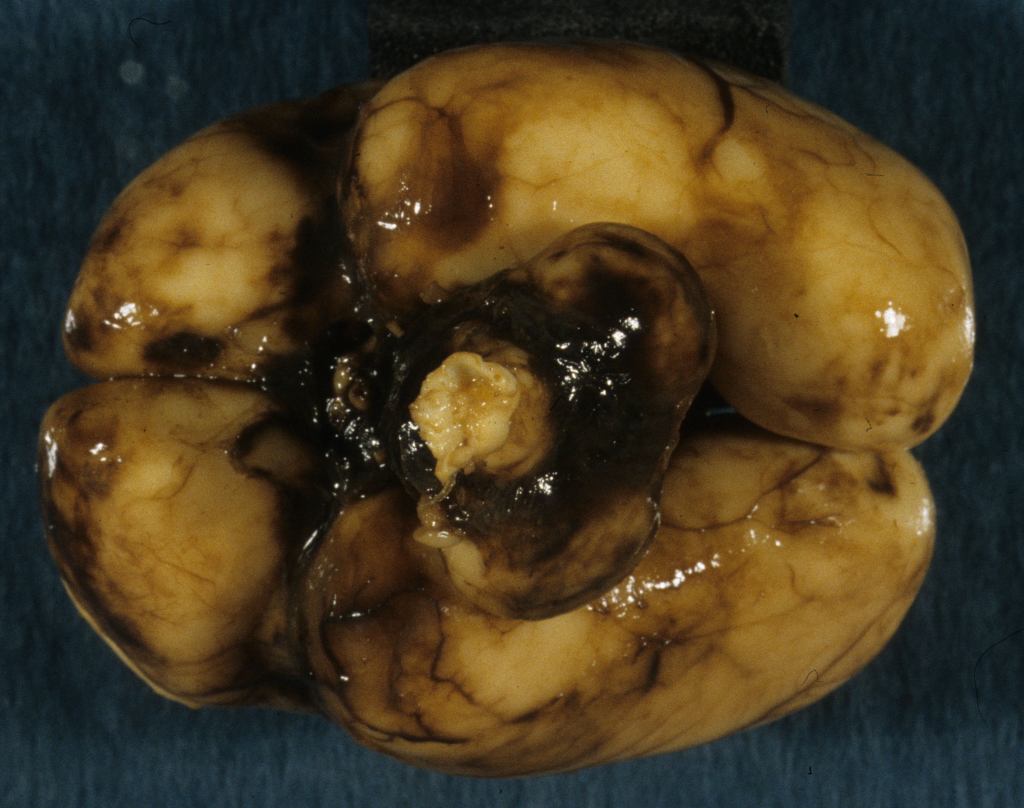
As in adults, this is the result of trauma tearing small veins traversing the subdural space (Fig 5).
There is evidence that lethal subdural hemorrhages can occur prior to birth1. In New Zealand, there is evidence that traditional practices of abdominal massage can lead to such hemorrhages2.
Parenchymal hemorrhage:
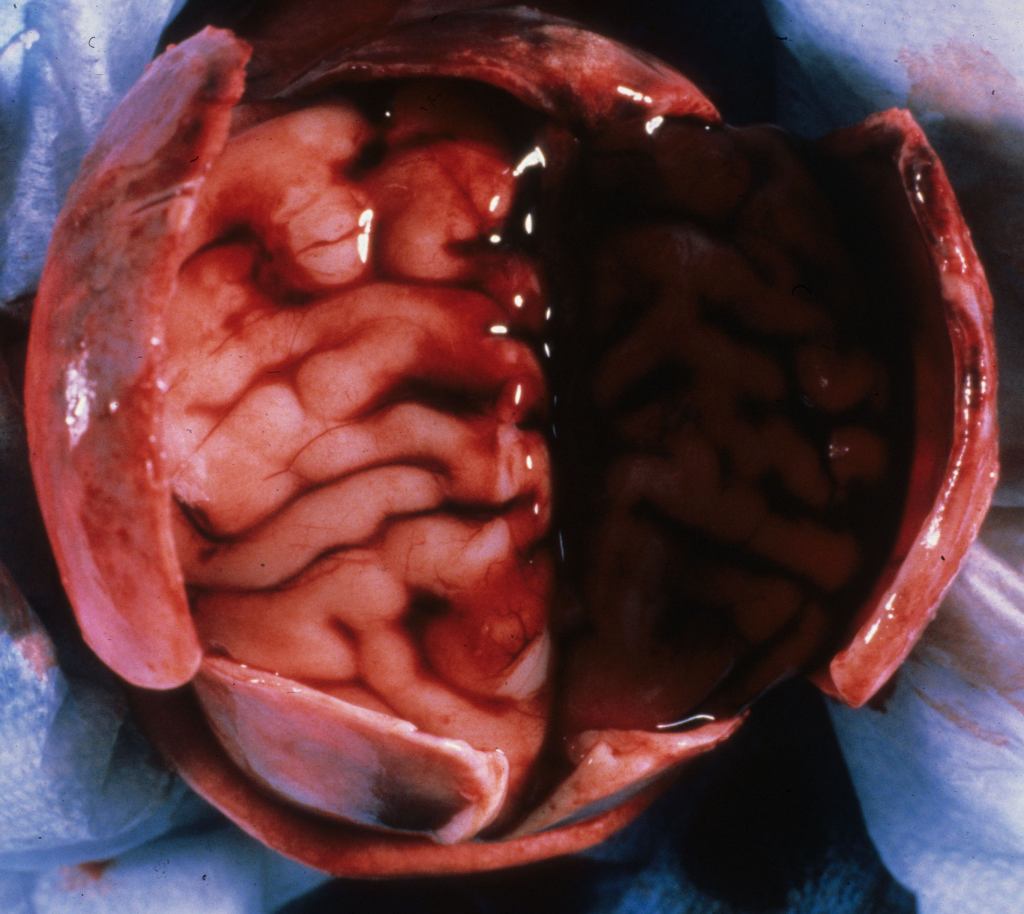
Deep hemorrhages may be evident in the intact brain if the hemorrhage extends to the subarachnoid surface (Fig 6a, b).
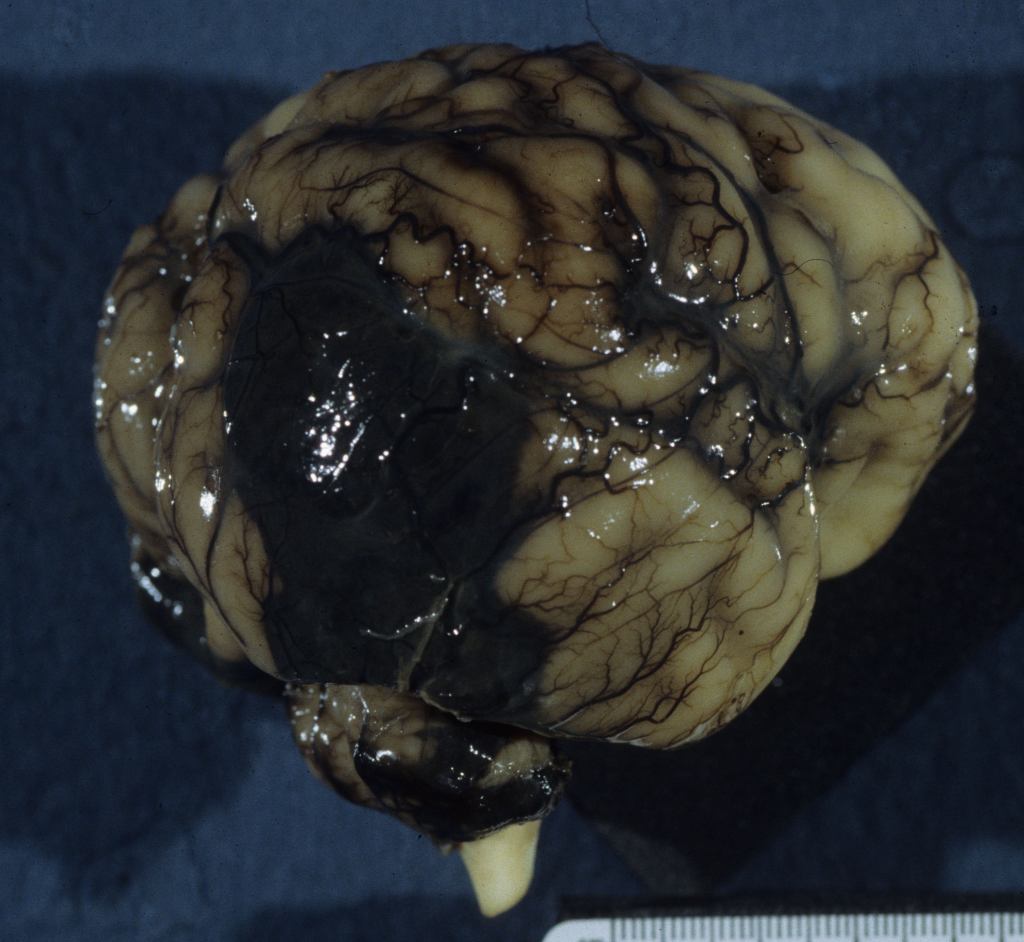

The causes of such hemorrhage may be from vascular malformation such as Vein of Galen aneurysm or from thrombocytopenia especially with anti- platelet antibody A1 in the mother3,4. As with Rh disease, the fetus must be A1 positive in an A1 negative mother. Infarctions of the brain may appear hemorrhagic. Because of the large proportion of spinal fluid and the flexible skull with unfused sutures, hemorrhage into the brain can cause fetal anemia and hemorrhage shock (Fig 7a, b, c).



Edema:
Following prolonged experimental hypoxia in monkeys, Dr. Myers discovered that the mechanism of brain injury involved edema from vascular leaking5,6. In the older gestation fetal brain, edema when marked can be recognized as compression of the lateral ventricles (Fig 8). This edema may also increase the expected weight of the brain for a given gestation.

Herniation:
Whether from edema, hemorrhage or mass lesion, uncal and tonsillar herniation of the brain is rare in the fetus because of the increased proportion of spinal fluid and the expansible skull. Herniation may be present in older gestation infants (Fig 9a b).
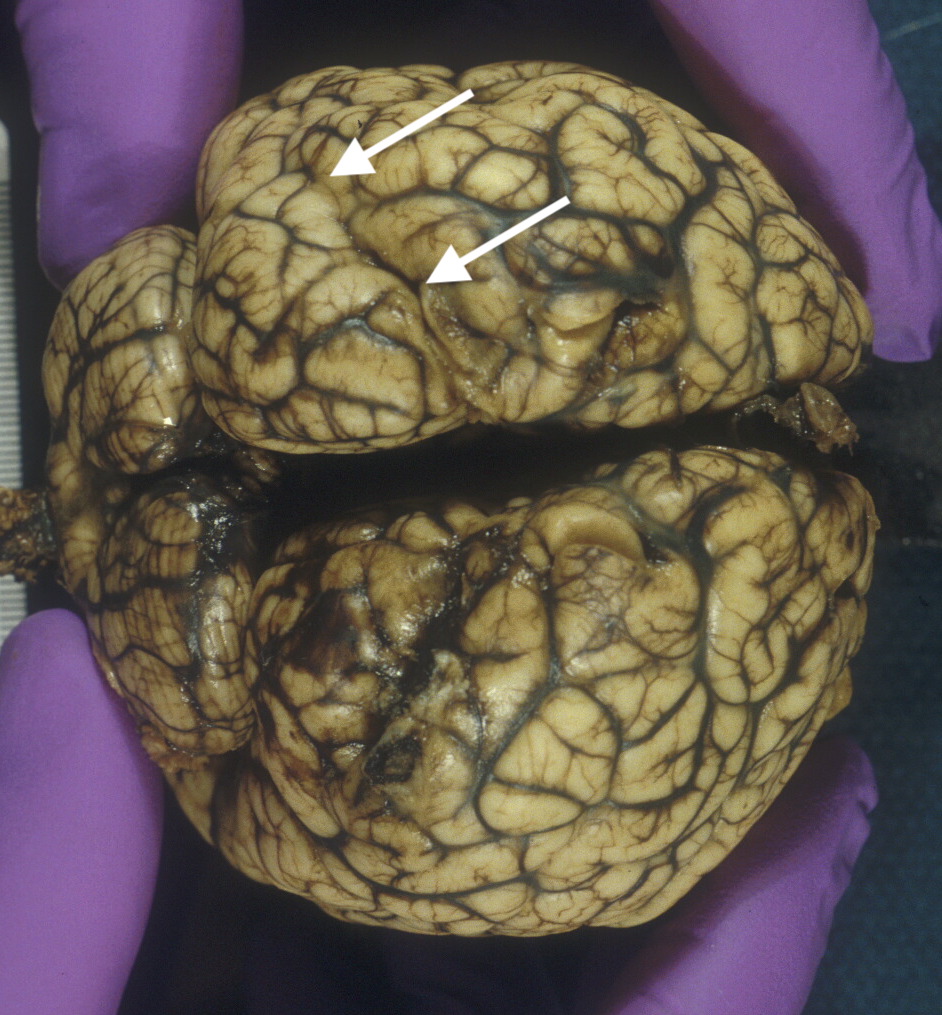

Infarctions:
In my residency training, I saw a stillborn brain with multiple scattered small cortical infarctions and in my fellowship, a typical middle cerebral artery distribution infarction. They were attributed to differential blood flow with hypoxia. However, there is adequate clinical evidence now that infarctions in the fetal brain can occur from severe genetic, or immune acquired, thrombophilia or from emboli from thrombi on the venous side of the placental circulation (Fig 10a, b)7. A cortical brain infarction may not be diagnosed in the nursery in an uncomplicated pregnancy.

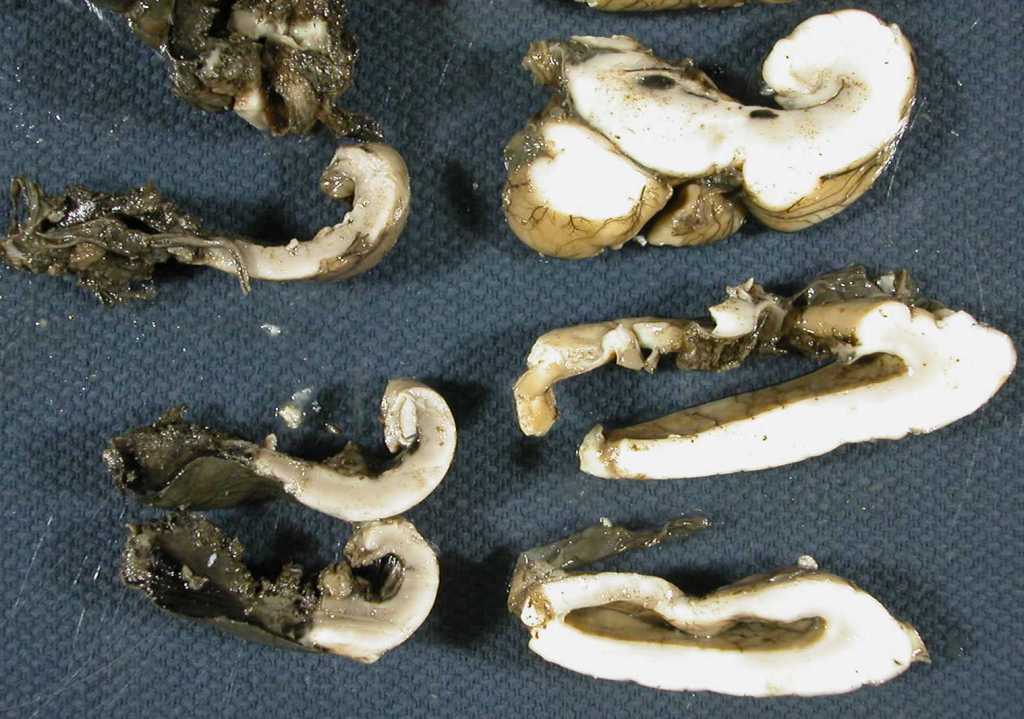
Periventricular leukomalacia and leukotelencephalic necrosis:
In premature infants, brain injury tends to occur in the white matter of the cerebral cortex, often in a periventricular pattern, but it may be more extensive. Early on there may be a chalky coloration, but over time the necrotic areas may develop into cystic cavities (Fig 11)8.

This white matter vulnerability in preterm infants has been attributed to low blood flow, increased glutamate receptors on immature oligodendroglia, etc9,10. In Dr. Myers experiments in monkeys white matter lesions could be produced even in mature monkeys with partial asphyxia and oligoacidotic hypoxia that is a pO2 at 8-10 mmHg while the pCO2 and pH remain minimally affected11.
Malformations:
It is not the purpose of this manual to include malformations, but common malformations include holoprosencephaly, various forms of hydrocephalus (e.g. Dandy Walker (Fig 12), Arnold Chiari, aqueductal stenosis), and midline cyst with absent corpus callosum (Fig 13).



Tumors:
There are congenital tumors of the brain, although uncommon and unlikely to be encountered in stillbirth. The most common malignant tumor is glioblastoma multiforme12.
Gestational age:
Comparing the gyral pattern of the brain to several available charts of gestational age development provides a relatively simple and accurate way to estimate gestation of the infant13(See manual post on gestational age). There are also microscopic changes in the brain that can be used to estimate gestation such as the loss of the external granular layer in different cortical locations (Fig 14a, b)14.


Microscopic:
White matter injury:
Early white matter injury can be difficult to discern, but will show on H&E slides a difference in texture from surrounding areas. As the lesion progresses there will be vascular growth and endothelial swelling, astrocytic gliosis with distinct eosinophilic cytoplasm and macrophage (microglia) ingestion of debris. The final stage may be a gliotic scar or a lacuna of spinal fluid surrounded by glial scarring (Fig 15).

In a stillborn infant, the progression of changes can provide a window of timing of a previous insult (Fig 16a, b).


Grey matter necrosis:
Gray matter necrosis shows neuronal necrosis, and progresses with astrocytic gliosis, vascular proliferation, gitter cells and eventually a glial scar or cavitation (Fig 17a, b).


Neuronal necrosis:
Neuronal necrosis is an apoptotic cell death of individual neurons. In Myers experiments in monkeys, there was a stereotyped progression of brain nuclei showing neuronal necrosis as the period of complete asphyxia increased (Fig 18a, b).

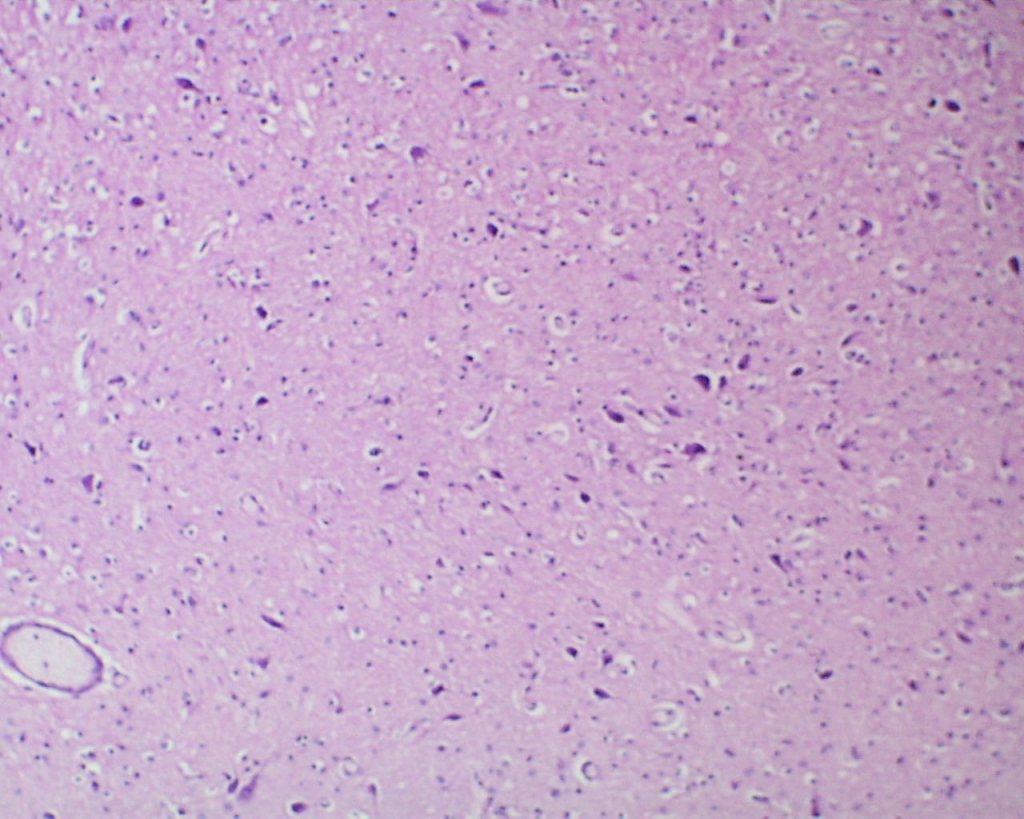
This pattern only occurred if the experimental monkey was initially not acidotic, and the progression showed a relationship to metabolic activity in those nuclei. In the perinatal autopsy, a frequent pattern of neuronal necrosis with karyorrhexis is found in the pons and subiculum (for more detail, see blog on Sept 30,2020 on “Cesarean section association with autism and attention deficit?”).
Hemorrhage:
Intraventricular hemorrhage originating in the germinal matrix can often be identified microscopically (19a, b).

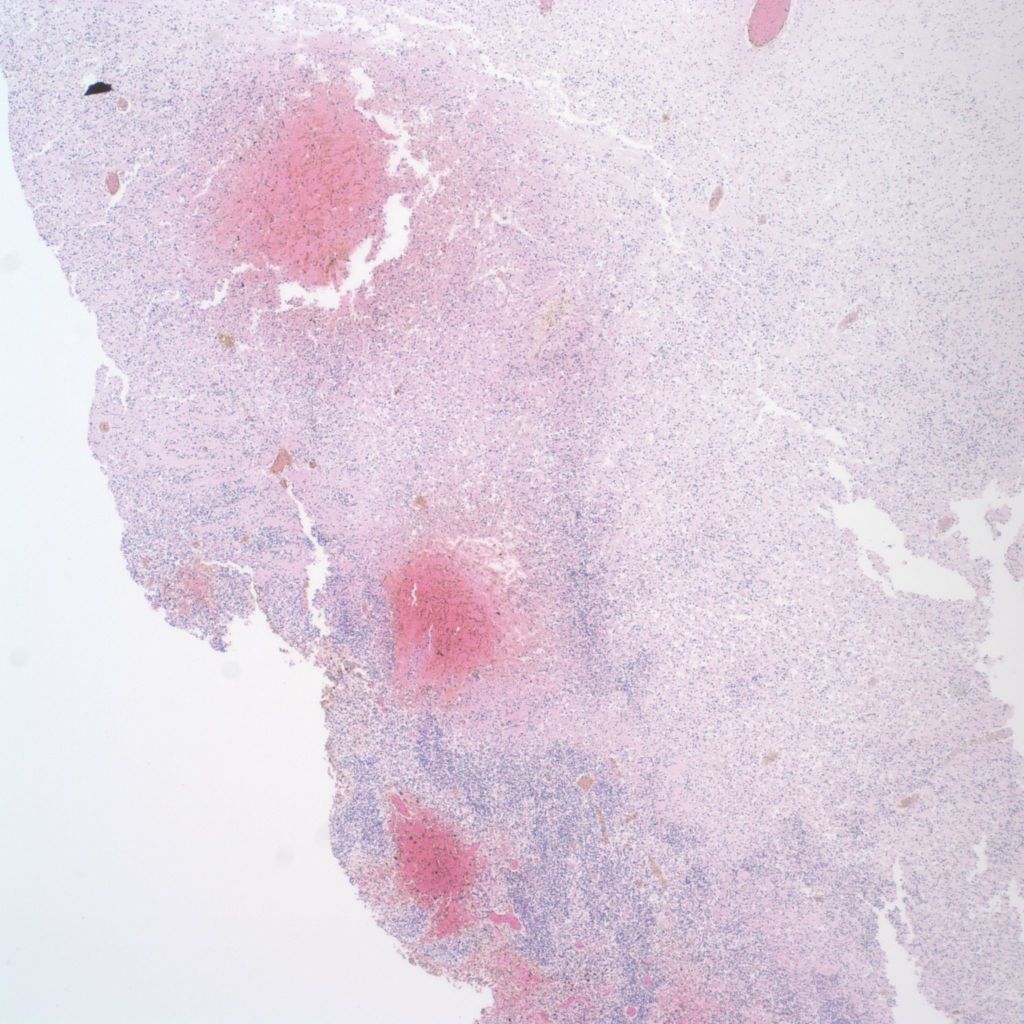
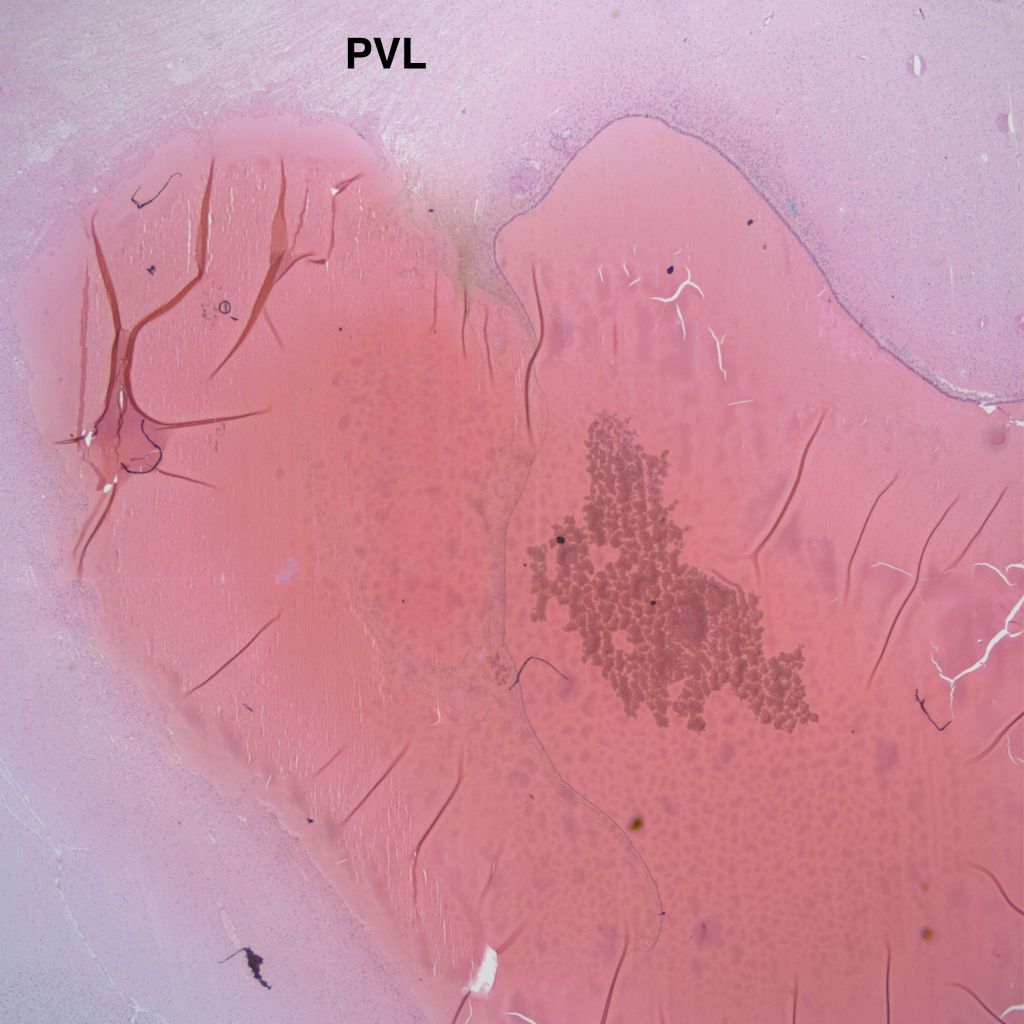
Where the hemorrhage extends into surrounding white matter that portion appears necrotic. From the histology, it may not be clear whether the hemorrhage caused the necrosis or the hemorrhage bled into a necrotic area. If the surrounding non-hemorrhagic white matter is necrotic, a reasonable inference is that the hemorrhage passively entered the necrotic area. Not all intraventricular hemorrhages are from the germinal matrix but may occur with other deep brain hemorrhage (Fig 20).
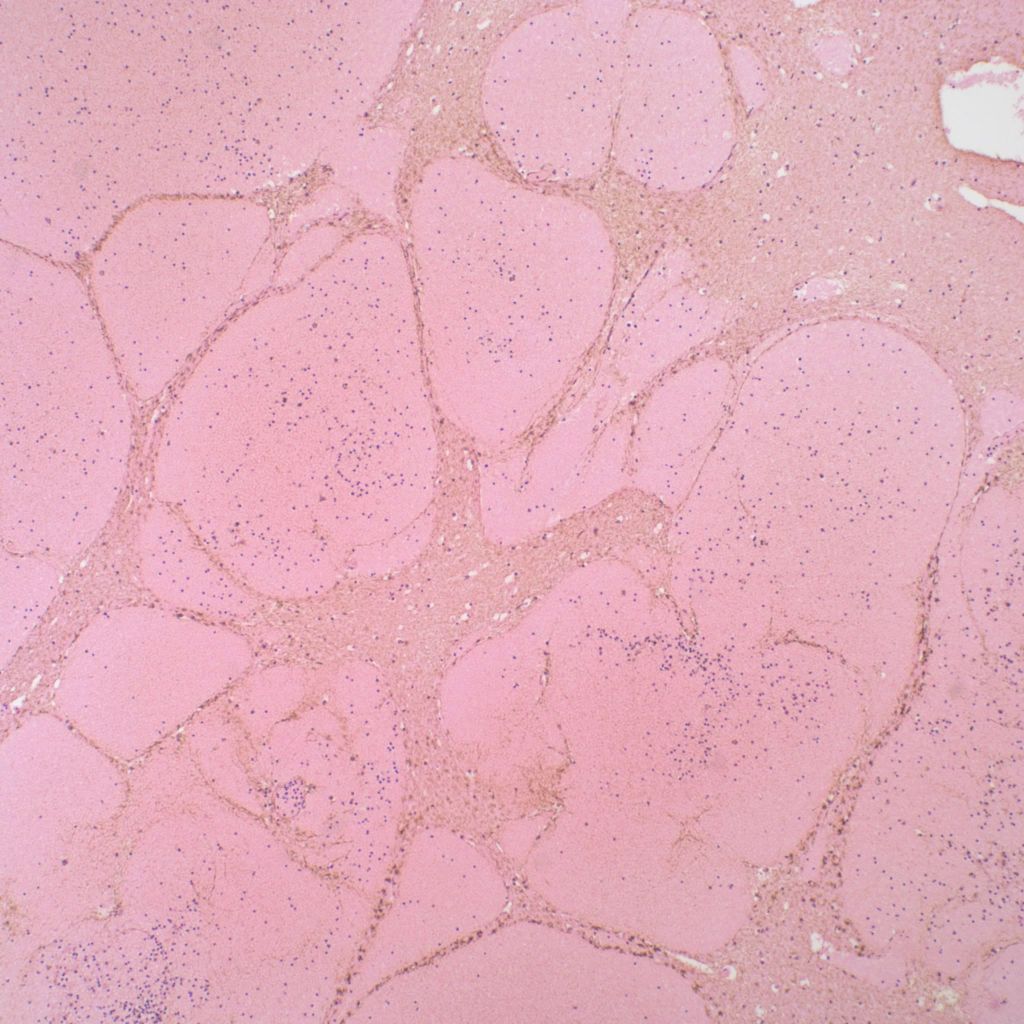
Parenchymal hemorrhages may have underlying vascular lesions that are not perceptible grossly (Fig 21a, b).
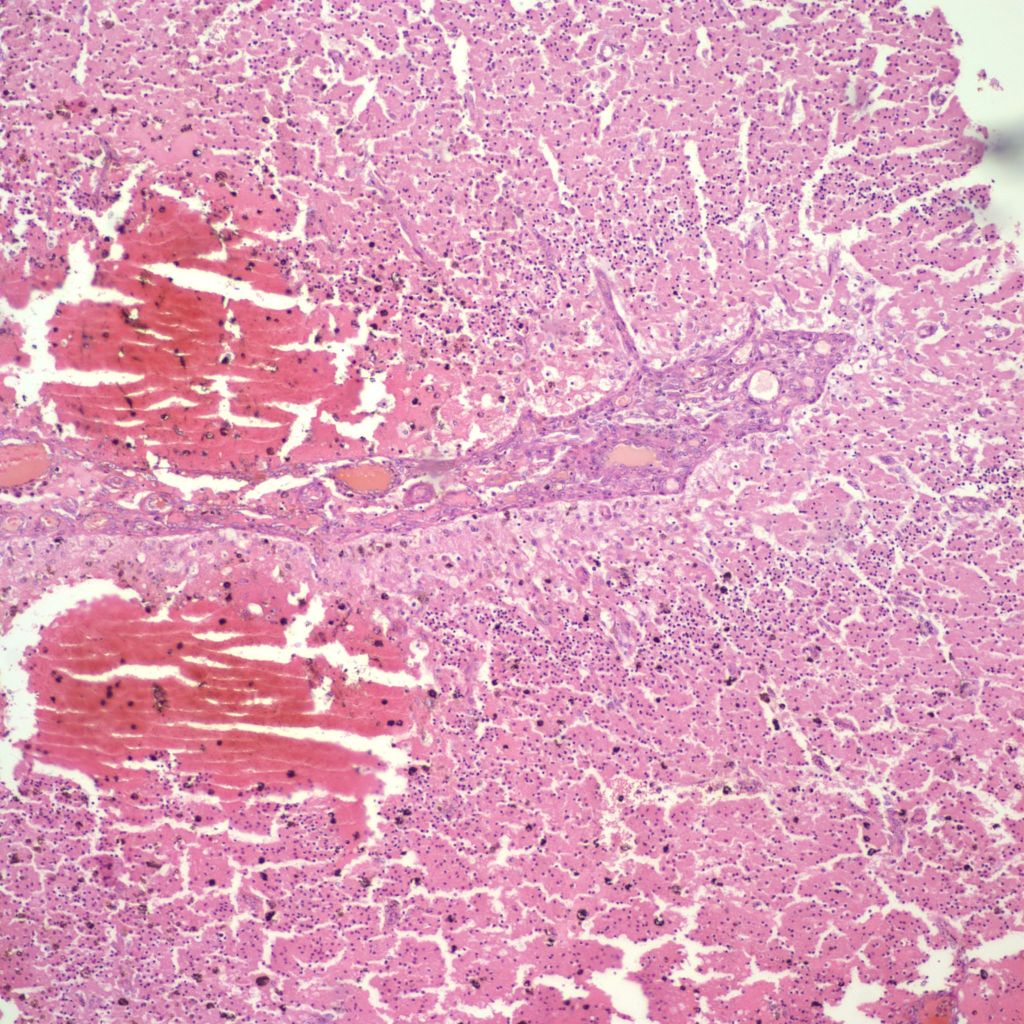
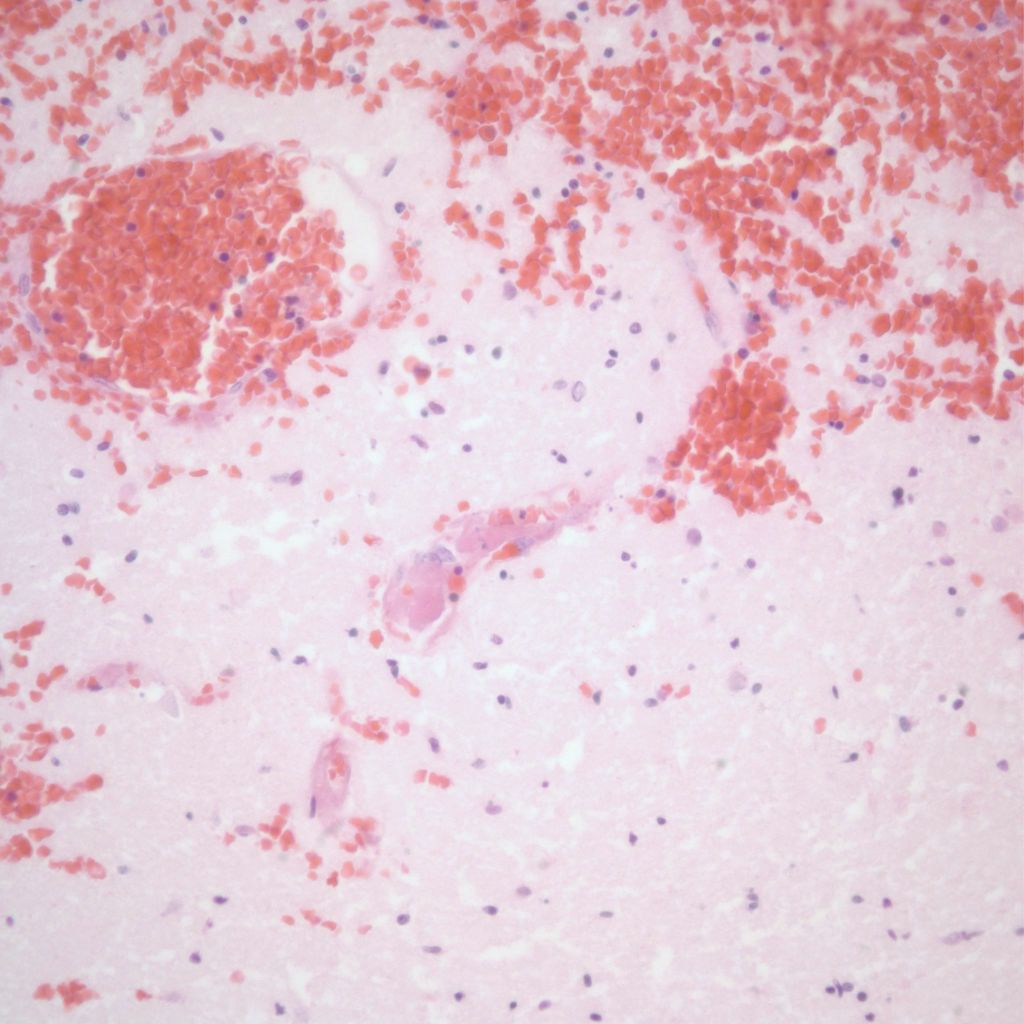
Hemorrhage can also occur into infarctions in the brain (Fig 22)
Infection: The most common fetal infections that directly affect the brain are CMV and HSV. CMV typically produces periventricular calcification. HSV may produce a necrotizing, geographic patterned encephalitis (Fig 23).
The role of fetal septic responses in white matter necrosis has been proposed.
See short case presentation below on an infant with hypoxic/ischemic brain injury.
References:
1. Demir RH, Gleicher N, Myers SA. Atraumatic antepartum subdural hematoma causing fetal death. Am J Obstet Gynecol 1989;160:619-20.
2. Becroft DM, Gunn TR. Prenatal cranial haemorrhages in 47 Pacific Islander infants: is traditional massage the cause? N Z Med J 1989;102:207-10.
3. Bussel J, Kaplan C. The fetal and neonatal consequences of maternal alloimmune thrombocytopenia. Baillieres Clin Haematol 1998;11:391-408.
4. Spencer JA, Burrows RF. Feto-maternal alloimmune thrombocytopenia: a literature review and statistical analysis. Aust N Z J Obstet Gynaecol 2001;41:45-55.
5. Myers RE, Beard R, Adamsons K. Brain swelling in the newborn rhesus monkey following prolonged partial asphyxia. Neurology 1969;19:1012-8.
6. Bondareff W, Myers RE, Brann AW. Brain extracellular space in monkey fetuses subjected to prolonged partial asphyxia. Exp Neurol 1970;28:167-78.
7. Rayne S, Kraus F. Placental thrombi and other vascular lesions classification; morphology and clinical correlations. Path Res Pract 1993;189:2-17.
8. Banker BQ, Larroche J-C. Periventricular leukomalacia of infancy. Arch Neurol 1962;7:386-410.
9. Folkerth RD. Neuropathologic substrate of cerebral palsy. J Child Neurol 2005;20:940-9.
10. Jensen FE. Role of glutamate receptors in periventricular leukomalacia. J Child Neurol 2005;20:950-9.
11. Myers RE. Four patterns of perinatal brain damage and their conditions of occurrence in primates. Adv Neurol 1975;10:223-34.
12. Winters JL, Wilson D, Davis DG. Congenital glioblastoma multiforme: a report of three cases and a review of the literature. J Neurol Sci 2001;188:13-9.
13. Larroche JC. Developmental Pathology of the Neonate: Excerpta Medica; 1977.
14. Friede RL. Developmental Neuropathology, Second Edition. Heidelberg: Springer-Verlag; 1989.p.5