Breus mole (massive subchorionic thrombo-hematoma), a variant of RPH?
This lesion is a distinct, rounded protruding mass on the fetal surface of the placenta resulting from the upward thrust of a large blood clot (Fig 1).

Fig 1a) This placenta from a 26-week gestation infant who died in the nursery shows a tumor like mass covering most of the fetal surface (within white arrows).

Fig 1b The cut sections of the placenta in Fig 23a demonstrate a thrombohematoma (arrows) over much of the fetal surface. There is a pale, older blood layered over redder, more recent blood suggesting two separate events with the first occurring well prior to delivery.
The blood remains beneath the chorionic surface confined in the intervillous space, yet it shoves the villi downward and expands the chorionic surface upward into the amniotic cavity. The stem villi appear to be stretched rather than ruptured (Fig 2).
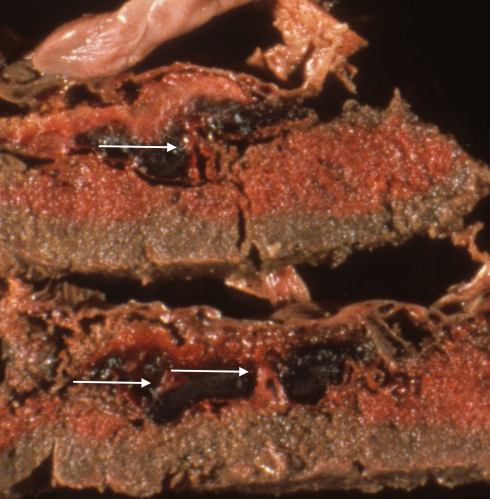
Fig 2 This is a blown up image of the left corner of Fig 23b. The arrows show the placental stem villi being stretched through the thrombohematoma.
The term “Breus mole” is a combination of the eponym of the author of an early paper in German describing the lesion and the term “mole” for a hill or mass. The older terms like tuberose and fleshy mole are very descriptive, but no longer used. I prefer to use “Breus mole” rather than the common descriptive nosology of massive subchorionic (or subchorial) thrombohematoma, which is a confusing term. First, a Breus mole is within the chorion (the placenta is also called the chorion frondosum), but beneath the fetal surface of the chrorion (similar to a submarine, which is below the surface of the water, but not below the bottom of the ocean). A retroplacental hematoma is truly under the placenta. Some confusion exists about the term “subchorionic” in publications particularly of ultrasonographic observations(1-3). Secondly, the term thrombohematoma is in some ways a contradiction of terms. I was once denigrated in court by another placental pathologist for using the term. The problem is that a thrombus forms in a space with flowing blood, but a hematoma forms outside of the vascular system. Breus mole is a special case that combines features of both a thrombus from blood flowing in the intervillous space, and as a hematoma expanding the placenta. Microscopically, the blood shows sparse strands of fibrin confirming the presence of flowing blood in its formation (Fig 3)

Fig 3 This very low magnification photomicrograph shows the fetal surface of the placenta with surface chorionic vessels on the left of the photograph with a thrombohematoma below. Lines of fibrin lamination can be seen in the thrombohematoma (H&E).
.
The blood making up the thrombohematoma is maternal. The volume of the blood clot in some cases would exceed the fetal blood volume. More direct evidence of the blood’s origin is provided when the fetal and maternal blood are morphologically distinct, such as sickled cells in maternal sickle cell disease, the 50% enlarged red cells in fetal triploidy, and many nucleated red cells in fetal erythroblastosis. In these cases, I found no appreciable fetal blood in the Breus mole (Fig 4).
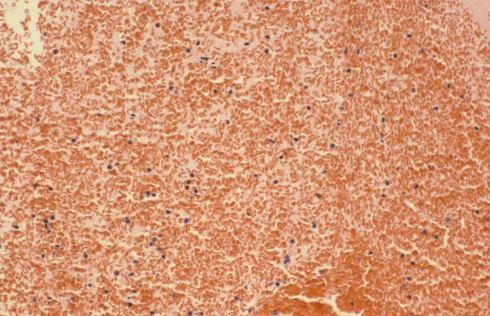
Fi g 4 This is high magnification microphotograph of the blood in a thrombohematoma in which all of the red cells are sickled indicating that the blood is maternal as fetal cells do not sickle. (H&E)
In a triploid placenta that I stained for fetal hemoglobin, no fetal cells were in the thrombo-hematoma (Fig 5).

Fig 5a : This is a very low magnification photograph with the thrombohematoma on the right of the picture and placental villi on the left. The tissue has been stained immunohistochemically to detect fetal hemoglobin as a dark brown precipitate. The light brown staining of the thrombohematoma does not demonstrate fetal hemoglobin in the red cells. (Anti-hemoglobin F)
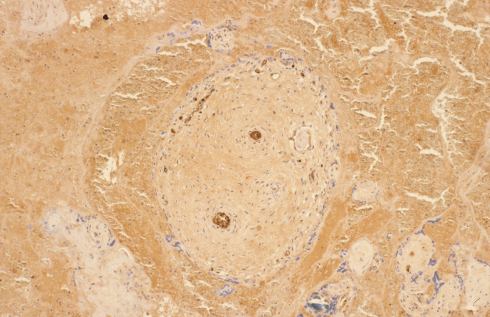
Fig 5b This image is a higher magnification of a villus in Fig 27a. The fetal blood within the vessels inside the villus shows the dark brown precipitate that identifies them as fetal cells. (Anti-hemoglobin F, 10x)
In a published case of triploidy, the authors also confirmed a lack of fetal blood in the clot(4). The lack of fetal blood does not prove that a fetal vessel is never torn in the formation of a Breus mole, but fetal hemorrhage appears to be the exception.
The Mechanism of Breus mole formation
A 1902 paper by D. Berry Hart proposed a reasonable hypothesis for the formation of Breus mole(5). After examining three specimens microscopically, he concluded that: “ There is an undue blocking of the serotinal (basal decidual) sinuses in the large-celled layer. This leads to a slow engorgement of the intervillous circulation. This will bulge out the chorio-basal septa, and as these tack down the chorion at definite points, the amnion and chorion will bulge up between. This produces the tuberose swellings…”. He argued that the hematomas caused fetal death. His specimens were from fetal death followed by a long intrauterine retention, as were those of Breus and many other early observers. As a result of this retention which could be months following fetal death, he gives the following clinical advice “The knowledge of the long retention of such specimens should also make one refuse to express any opinion as to the chastity of a woman who has expelled such a mole, and whose husband has been absent for a longer period than three months.” A review of Breus mole by Dr. Shanklin also proposed a similar mechanism of obstructed intervillous blood outflow(6).
To perhaps visualize this process better consider that the intervillous circulation is divided from the amniotic fluid by a flexible membrane. The flow in the intervillous space is high flow because of decreased vascular resistance in the spiral arteries, and hence must also be at low pressure, but obviously that pressure must equal the amniotic fluid pressure or the placenta would collapse and flow would come to a stop. Now imagine that the outflow of the intervillous circulation became obstructed. The spiral artery blood is at low pressure, but is in a continuous circuit with the systemic blood flow. Blood would continue to flow into the intervillous space until stopped by reaching systemic blood pressure. This would be at a higher pressure than normal flow and higher than the amniotic fluid pressure. As a result, the membrane between the amniotic fluid and the intervillous space would bulge into the amniotic fluid until its elastic limits prevented further expansion then the blood would clot from lack of flow. This bulging clot is the Breus mole. This model is simplified and does not account for some intervillous blood finding its way laterally into other venous drainage.
If we tentatively accept that obstruction of intervillous venous outflow can produce a Breus mole, then how does the flow become obstructed? One way to approach that question is to look at the clinical associations with Breus mole. The originally reported and most common association is with intrauterine retention of a dead fetus(7). Normally with an early gestation fetus, the placenta separates and is delivered vaginally. However, it is not uncommon that we receive in pathology a curettage because the immature placenta remained adherent to the uterine wall. If the placenta were to remain attached, but the uterus contracted because of the loss of amniotic fluid, then the uterine intramural pressure might stop intervillous blood outflow but not be high enough to block arterial inflow into the placenta. This would produce a Breus mole from total venous obstruction. This is a plausible but unproven scenario. A similar mechanism could occur with a sustained uterine tetany.
Starting with the report of Drs. Shanklin and Scott, Breus mole has been identified in live born infants, not just retained fetal deaths(6). Another explanation for the venous obstruction is needed for these cases. The obstruction of a small venous opening in the maternal floor of the placenta seems unlikely to cause a significant back up of blood. Thrombi in placental veins in the attached basal decidua are occasionally seen incidentally in placental samples without any lesion. However, if a deep venous system became obstructed, then the scenario envisioned for forming a Breus mole could be fulfilled. If thrombosis was cause of such venous obstruction, then there might be an association of Breus mole with thrombophilia. One published report suggests as much. An index case of a stillbirth with Breus mole in a mother with homozygous C677T mutation of MTHFR gene led to discovery of two more cases of stillbirth with Breus mole and various other thrombophilias(8). The original case does not report on the duration of intrauterine retention, but the photograph of the hematoma looks recent, as it is deeply colored. The authors do not try to prove that the mole caused the fetal death but they note that their observation is in keeping with the theory of occlusion of venous outflow from thrombosis as a mechanism producing a Breus mole. Of course, finding three cases does not prove a causative association.
In theory, if blood at systemic pressure were to enter the intervillous space directly it could produce a Breus mole. Most Breus moles extend into the marginal area of the placenta and some are clearly related to a marginal hematoma or even placenta previa (Fig 6).
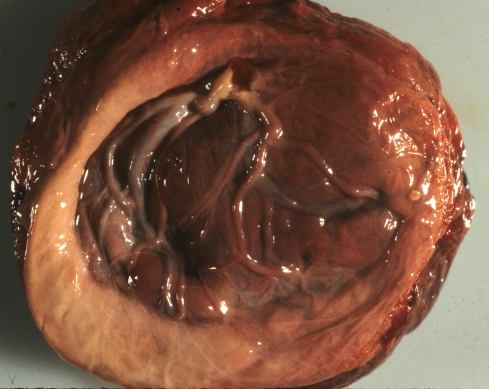
Fig 6a The fetal surface of this placenta demonstrates a raised tan margin that appeared to be a circumvallate placenta with a large Breus mole. The margin however was not typically folded in but appeared simply raised.

Fig 6b When the placenta in Fig 28a was sectioned, the margins could be seen to be a circummarginal old Breus mole with a second more acute Breus mole reaching from the margin to the center of the placenta (*).

Fig 6c The placental and membrane surfaces are stained with brown hemosiderin. There is an old marginal hematoma from which hemoglobin must have diffused into the amniotic cavity. On the right side is an old (pale) Breus mole (*). The placenta was diagnosed prenatally as placenta previa in a 24 week-gestation fetus with 24 hours of postmortem intrauterine retention. The mother was delivered by Cesarean section.
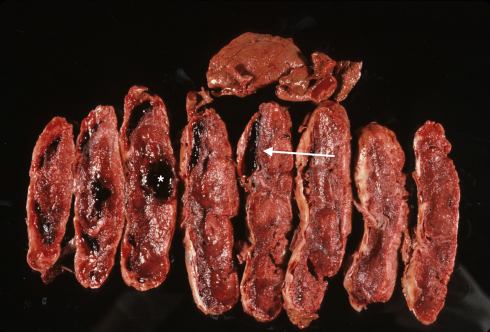
Fig 6d The cut sections of the placenta in Fig 28c demonstrate that a more acute Breus mole underlying the surface fibrin deposition created the raised mass (arrow). The fetal surface is on the left side of the slices. The relationship to the older marginal hemorrhage cannot be determined from the anatomy. There is an equally acute hematoma in the middle of the parenchyma (*) that appeared to be a dissection of the subchorionic thrombohematoma into the parenchyma.
The direction of blood flow, either from the mole to the margin versus from the margin to the mole, cannot be determined from the delivered placenta. Yet, the probable mechanism is the equivalent of a marginal retroplacental hemorrhage that ruptures into the intervillous space rather than spreading laterally in the basal decidua.
Is there any evidence for this mechanism? Admittedly the evidence is indirect. Some reported cases have reported maternal pain as a presenting symptom(9). I have seen cases presenting as abruption without a retroplacental hematoma, only Breus mole (Fig 7).
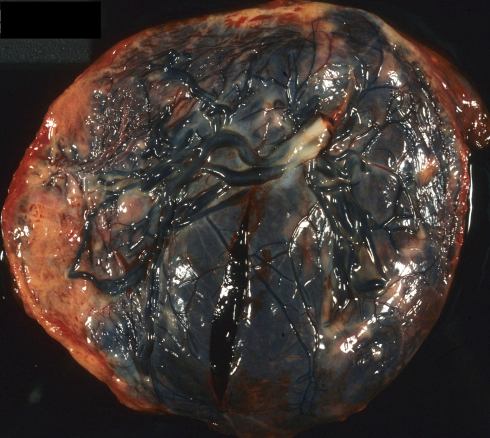
Fig 7a The fetal surface of this placenta shows a large acute (red) Breus mole occupying the right two thirds of the placenta. This 33 week-gestation stillborn infant was delivered to a mother who had presented with acute abdominal pain. The fetal autopsy was consistent with acute asphyxia and a fetal death that occurred between 4 and 24 hours prior to delivery. A retroplacental hematoma would have been demonstrable after a minimum of 4 hours duration. The timing is consistent with the Breus mole causing acute asphyxia and death. The compressed placental parenchyma suggests that intravillous flow was impeded.

Fig 7b The cut sections of the placenta in Fig 29c demonstrate the expansion of the Breus mole at both margins of the placenta, but there was no retroplacental hematoma.
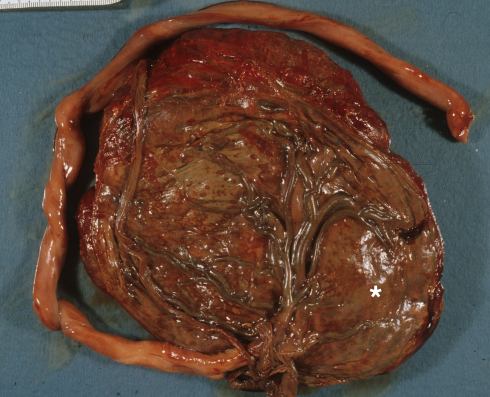
Fig 7c This placenta shows hemoglobin staining of the membranes and cord, and a large Breus mole (*). The placenta was from a 37 week AGA infant, with maternal distress thought to be an abruption, and with Apgars scores of 7/9.
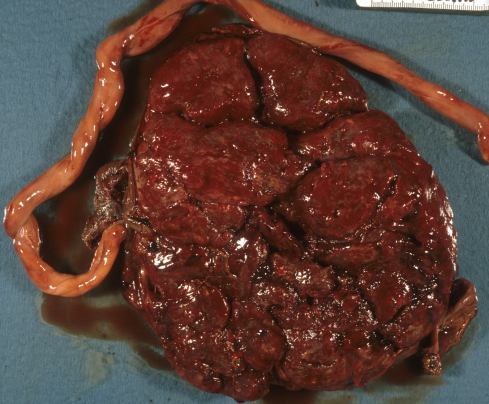
Fig 7d The maternal surface of the placenta showed no retroplacental hematoma, but the surface was redder beneath the hematoma.
One reported case of a Breus mole as a type of abruption disappointingly failed to prove either diagnosis to me(10). A case of Breus mole with a triploid fetus presented with pain, and pathologically the authors suggest that the lesion was either a Breus mole or a retroplacental /marginal hematoma that had ruptured into the placenta(4). The association in early reports of Breus mole in retained fetuses might be explained by the tendency in early gestation for incomplete separation and marginal hemorrhage (Fig 8).

Fig 8 : This sliced placenta is from a therapeutic abortion showing the continuity of an acute marginal hematoma and an acute marginal Breus mole (white arrow). There is a central Breus mole that cannot be fully explained by the marginal lesion (pink arrow).
In retained fetuses the marginal hemorrhage and adjacent Breus mole could have formed from loss of amniotic fluid volume and persisted or occurred during the labor of the retained fetus. The mechanism of a ruptured marginal hematoma seems as anatomically plausible as venous outflow obstruction, but the truth may require more direct observation in situ by ultrasound, or by in vitro experiment.
Unlike a premature placental separation, a Breus mole does not devitalize a volume of placenta. Yet, Breus mole in case reports and case series is often associated with fetal death and/or intrauterine growth retardation (6, 9, 11-13). Some case reports of intrauterine growth retardation, and / or poor fetal outcome including death with a prenatally detected Breus mole have found either absent or reversed end diastolic umbilical venous blood flow, a marker of increased fetal vascular resistance in the placenta(11, 14-16). Perhaps the stretched stem villi between the mole and the functioning placental parenchyma could be narrowed and less distensible, increasing resistance. More likely, the intervillous blood flow beneath the placenta is slowed and the vessels are constricted. However, without evidence from before and after measurements, the growth restriction and fetal vascular resistance could have preceded the Breus mole. One in which the Breus mole and abnormal Doppler studies were identified simultaneously has a pathological description consistent with massive intervillous fibrinoid deposition(17). This latter lesion is strongly associated with severe intrauterine growth retardation that would have preceded the formation of the Breus mole.
If a Breus mole caused a stillbirth, then the age of the mole and the duration of intrauterine postmortem retention of the fetus should be similar (Fig 9).
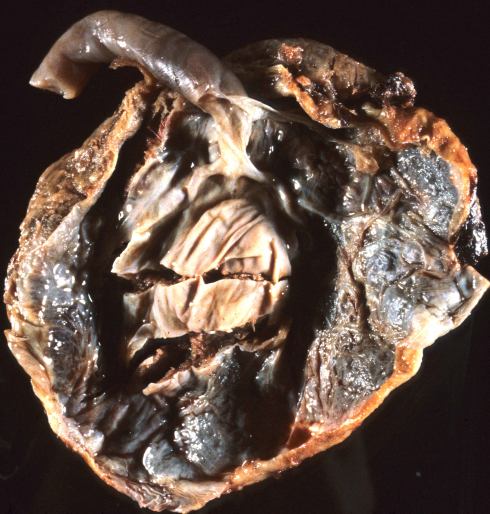
Fig 9a The fetal surface of this placenta from a stillborn infant shows brown staining of the umbilical cord and membrane surfaces. There is a large, central, tan Breus mole. The staining is due to diffusion of hemoglobin both from the Breus mole and the umbilical cord blood. The image does not show well that there was a short furcate insertion of the cord.

Fig 9b The cut section of the placenta in Fig 31a shows that the hematoma is brown tan consistent with the autopsy estimate of postmortem fetal retention of 48 hours. The autopsy also demonstrated a double outlet left ventricle. The surface thrombo-hematoma was subchorionic. A torn umbilical vessel would have created a subamnion hemorrhage or bled into the intervillous blood flow. The parenchyma beneath the Breus mole is not compromised. The timing of this Breus mole is consistent with occurring at the time of death, but the mechanism of death is unclear.
The age of the Breus mole, as with other hematomas, can be estimated by the transition from deep red to brown to tan to white. In a live birth, the old ag e of the Breus mole is evidence that the infant survived it prior to delivery. It is difficult to understand how a relatively small (less than 50% of the surface) could cause fetal death. Further prenatal ultrasound investigation focused on timing and full evaluation of blood flows, as well correlation with the clinical history and pathology should help clarify the origin and significance of Breus mole.
Two clinically insignificant lesions that could be confused with Breus mole.
Subchorionic fibrin plaques
Subchorionic fibrin plaques are white, tan or yellow flat discs of fibrin that form just beneath the fetal surface. On cut section, they are subchorionic in location and from the intervillous side show a smooth maternal surface with an “empty” space beneath it (Fig 10).

Fig 10a This placenta is that noted in the text. The fetal surface shows a small subchorionic fibrin plaque (arrow).
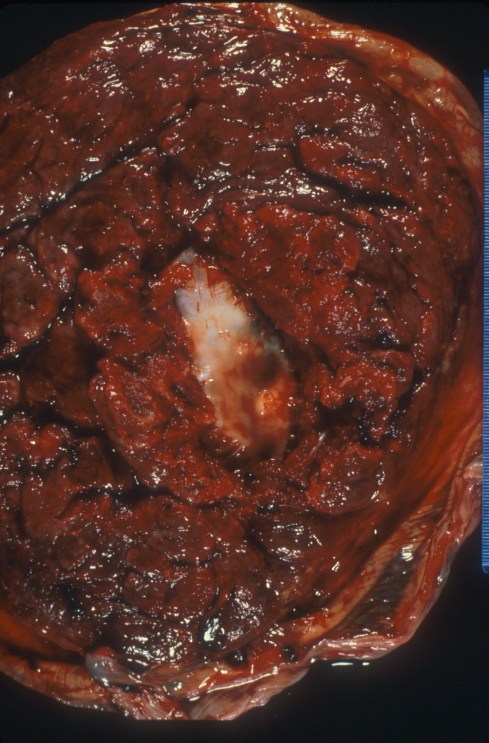
Fig 10b The maternal surface of the placenta has been pulled aside to reveal the smooth undersurface of the plaque seen in Fig 10a.

Fig 10c The ultrasound demonstrates a lucency believed to be the plaque in Fig 10a.

Fig 10d The microscopic sample of the plaque demonstrates tightly laminated fibrin strands. (H&E, 4x)
Grossly and microscopically the lesions show closely spaced lamina of fibrin. Very early in my career, a perinatologist asked me to locate a subchorionic echolucent mass that had slow flow beneath it that he had been seen on ultrasound. The mass was a subchorionic fibrin plaque. The subchorionc fibrin plaque likely formed in an area where the flow upward from the spiral arteries tracks across the upper subchorionic surface before finding a path to the venous outflow. These areas are likely slow flowing and promote the formation of thrombus against the surface. The lesions are unlike Breus mole in that there is no “massive” bulging of the surface, and more under the microscope the fibrin is densely laminated with none of the free blood characteristic of a thrombohematoma.
The lesions are common and vary in number and size from placenta t to placenta. There is no evidence in my experience that they are clinically significant. A study by Dr. Naeye claimed to have demonstrated decreased fibrin plaques in infants with hypotonia[18]. He hypothesized that the plaques were caused by fetal kicking of the surface. This hypothesis seems unlikely, as there is no underlying epithelium to be injured by kicking that would initiate thrombus formation. Until another study confirms this hypothesis, I think there is insufficient evidence to associate absent fibrin plaques with fetal hypokinesia.
A curiosity of the plaques is that they do not organize. Organization is a pathological term for the in growth of new capillaries, and the accumulation of macrophages and of active fibroblasts that eventually reopen a thrombosed vessel or turn the thrombus into a flattened scar. The intervillous space has no surface endothelium to grow into the thrombus. Trophoblastic cells line the intervillous space and inhibit mural thrombus, but they do not grow into thrombi to start new capillaries (neovascularization). The only available endothelium lines the fetal vessels and it is rare for these fetal vessels to disrupt and organize intervillous fibrin. Thus, thrombi in the intervillous blood space tend to be permanent, at least for the short life of the placenta.
Chorionic cysts
Another lesion that can resemble Breus mole is a blood filled chorionic cyst. A chorionic cyst is a fluid filled sac on the surface of the placenta that protrudes into the amniotic cavity and is covered by a thinned layer of chorionic connective tissue that may include chorionic vessels (Fig 11).

Fig 11a The fetal surface of the placenta shows several small, clear chorionic cysts (arrows)

Fig 11b The fetal surface shows a large chorionic cyst with some hemorrhage into it.
The amnion is also above (on the fetal side of) the cyst. The bottom of the cyst sits on subchorionic fibrinoid. The inner lining of the cyst is chorionic epithelium composed of cytotrophoblastic cells of fetal origin, sometimes called “X” cells (18). The underside of the fetal surface of the placenta, that is the portion on top of the intervillous blood spaces starts with a continuous trophoblastic lining. This lining during gestation is lost and the surface becomes covered with a layer of fibrinous fibrinoid, a combination of maternal blood fibrin and cytotrophoblast secretion (annexins, fetal fibronectin etc.) Cytotrophoblastic cells become trapped in this fibrinoid, but remain viable. If a sufficient volume of cyotrotroblast cells is trapped, they may continue to secrete, and create a pseudo-lumen (Fig 12),

Fig 12a This is a photomicrograph of the fetal surface of an immature placenta showing the thick layer of cytotrophoblastic epithelium lining the top of the intervillous space (arrow). (H&E, 4x)
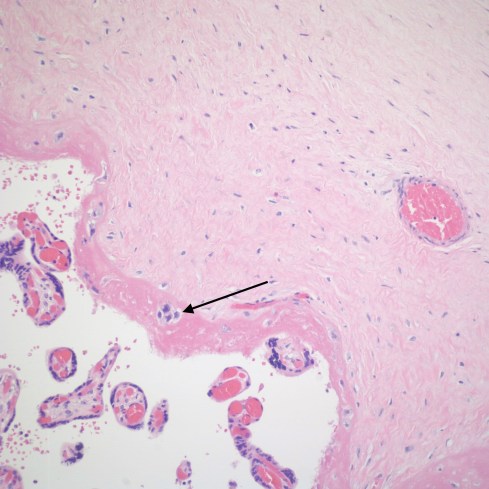
Fig 12b The fetal surface of the placenta is at the top of the photomicrograph with the pink subchorionic fibrinoid running diagonally above the intervillous space. The arrow points to some trapped residual cytotrophoblast. (H&E, 20x)

Fig 12c This photomicrograph shows a larger volume of entrapped cytotrophoblast in the subchorionic fibrinoid that have formed a pseudo

Fig 12d This photomicrograph shows the expansion of a small surface chorionic cyst with fluid. (H&E, 4x)
The fluid in the cyst has not been completely studied, but may also include maternal serum that has “leaked” into the cyst (see footnote). If the cyst re-communicates with the intervillous space, maternal blood will mix with cyst fluid content and could be mistaken for a Breus mole (Fig 13).
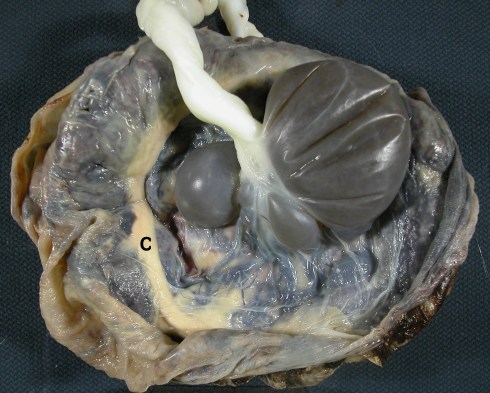
Fig 13 The fetal surface of this placenta shows a large, multilobular chorionic cysts filled with dark blood stained fluid. There is also a partial circumvallate margin of the placenta (c).
Chorionic cysts are often adjacent to subhchorionic fibrin plaques or circumvallate fibrin (which also traps a layer of chorionic epithlelium) suggesting that the local conditions in these areas favors cyst formation (Fig 14).
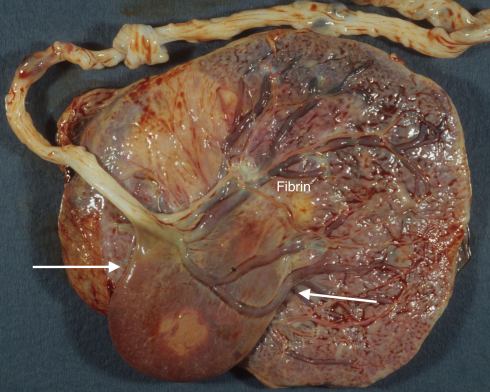
Fig 14a The fetal surface demonstrates a large, drooping, thin walled chorionic cyst (arrows) that has a base at a fibrin plaque (“fibrin”).
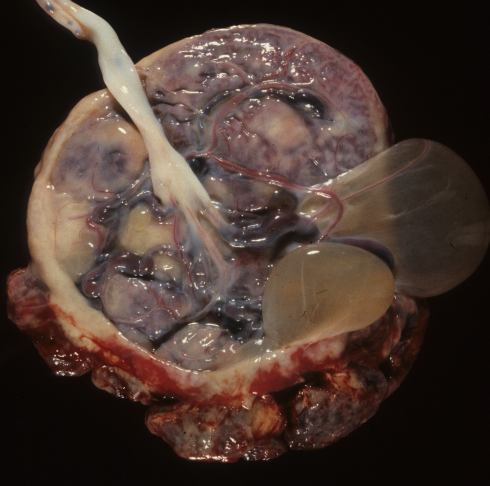
Fig 14b The fetal surface of this placenta has a cyst that appears to arise from the partial circumvallate margin.
Smaller accumulations of cytotrophoblast secretions are seen in the chorion of placental septa and fetal membranes (Fig 15).

Fig 15a This photomicrograph shows multiple chorionic microcysts (arrows) in the chorion layer of the fetal membranes. (H&E, 4x)

Fig 15b This photomicrograph demonstrates a small cyst in a septum in the middle of the intervillous space. (H&E, 10x)
In these areas, cysts are usually microscopic or at least smaller than a 1 cm diameter(19). One author has argued that the formation of these microcysts is associated with uterine hypoxia, but I have not found the evidence convincing (20-22). He demonstrated comparable associations with microcysts in placental septa and showed an association with maternal diabetes(23). If the microcysts were in response to uterine hypoxia, membrane hypoxia would be due to ischemia of the uterine body, but not necessarily associated with utero-placental ischemia. Placental blood flow in normal circumstances is more dependent on trophoblast invasion lowering vascular resistance in spiral arteries than to overall uterine blood flow. These microcysts are common even in uncomplicated pregnancies. To understand these cysts it is helpful to understand the roles of chorionic cytotrophoblast. Some have regionally specific functions, for example the intervillous cytotrophoblast produces an extracellular matrix, while the invasive cytotrophoblast is capable of killing and replacing maternal vessels. The cytotrophoblastic cells of the fetal membrane and those of the base of the placenta (including the placental septa) form a chorionic epithelium that adheres tightly to the underlying de-epithelialized and decidualized endometrium. This membrane chorion forms several cell layers of a stratified epithelium without the inside versus outside polarity of a glandular epithelium. It is within clefts of this layer and a similar layer in septal cysts that colloid collects and forms microcysts. Similar collections of colloid occur between chorion epithelial layers in the septum of dichorionic twins. A reasonable hypothesis is that chorionic epithelial membrane normally secretes a substance that escapes into surrounding tissue, but if some mechanical factor allows a separation of cells, the secretion accumulates as colloid. These microcysts do not evolve into large macroscopic cysts like those in the fetal chorionic surface of the intervillous space perhaps because they are not exposed to the intervillous circulation (see footnote).
Most chorionic cysts are found incidentally in placentas from uncomplicated pregnancies. Most reports of chorionic cysts detected by antenatal ultrasound also had no complications (24-28). There are reported cases of a chorionic cysts associated with intrauterine growth retardation but these could be biased if the patients were referred for ultrasound examination because of suspected intrauterine growth retardation(27). Indeed three cases were associated with maternal floor infarction (MFI) including two infants from the same mother. This could be a chance association, but it is reasonable to suspect that the increased cytotrophoblast proliferation and turbulent blood flow of MFI could predispose to cyst formation.
In theory a rapidly expanding cyst could put tension on vessels draped over it, causing vascular narrowing and increased resistance. Two reports of ultrasound detection of cysts in patients not referred for intrauterine growth retardation demonstrated Doppler evidence of increased umbilical flow resistance and mild growth retardation (29, 30). In one of these reports another patient with a similar cyst did not have growth retardation(30). None of the studies reported detailed placental pathology that could exclude other causes of increased placental vascular resistance, and no Doppler measurements were reported for the vessels overlying the cysts, but rather the values were for umbilical arteries. One reported antenatal case series did not find an association with fetal growth retardation of either the location of the cyst nor the size of the cyst (27). There is no direct evidence of harm from rupture of the cyst although this has been recorded to occur with delivery. Cysts sometimes appear to shrink with serial ultrasound examinations perhaps due to leakage of fluid perhaps into the maternal blood flow of the intervillous space.
(Footnote: A 4 cm prenatally diagnosed chorionic cyst was sampled biochemically and compared to the patient’s amniotic fluid, maternal serum and fetal serum using a basic metabolic profile screen(31). The fluid was slightly green, suggesting hemolysis which would account for the elevated iron and bilirubin found in the cyst. Small molecules such as glucose, urea, and carbon dioxide were the same in all fluids consistent with an ability to diffuse between the compartments. Other molecules distributed differently in the different fluids.
Despite the ease of sampling the fluid, only one component that might be a specific cytotrophoblast secretion has been identified. Using immunohistochemistry , one study identified immunologic reactivity for eosinophil major basic protein in the cyst fluid(32). This was confirmed by in situ identification of the protein in placental X cells, which includes the cytotrophoblast surrounding the cysts(33). It is a somewhat frightening notion that these cysts might be filled with such a noxious substance, but the proMBP protein is reportedly bound to pregnancy associated plasma protein-A which might make the substance harmless (34, 35).)
(Footnote 2: The cytotrophoblast cells on the fetal intervillous surface, like those in the fetal membranes, sometimes show colloid microcysts. Protein colloid accumulation initiates by an unknown stimulus but appears to occur in thicker epithelial layers. In the membranes, this accumulation is the end of the process. Against the intervillous space, something happens that creates macroscopic cysts. Since in both locations, the chorion has the same anatomic relationship to the amnion, the amniotic fluid is not the source of enlarging cyst fluid. That exclusion leaves the maternal blood and serum as the source of fluid to expand the cyst. A fetal surface microcyst is separated from the intervillous blood only by a floor of fibrinous fibrinoid. This floor is likely permeable to some solute, water and perhaps other molecules in the blood. The one biochemical study of a cyst is consistent with this compartmentalization of cyst fluid and blood(31). Since the protein of the colloid is too large to diffuse out of the microcyst, it would provide the osmotic force to enlarge the cyst. Another way the cyst could enlarge is by rupture of the floor with direct hemorrhage into the cyst and then thrombotic resealing of the gap. Some cysts have clearly suffered such hemorrhages (25, 26). An increase in pressure in the cyst increases wall tension in the overlying connective tissue of the chorion that then expands to relieve that tension. This explanation of the formation of chorionic macrocysts is hypothetical but fits the observed anatomy and physical laws.)
References
- Windrim C, Athaide G, Gerster T, Kingdom JC. Sonographic findings and clinical outcomes in women with massive subchorionic hematoma detected in the second trimester. J Obstet Gynaecol Can. 2011;33(5):475-9.
- Seki H, Kuromaki K, Takeda S, Kinoshita K. Persistent subchorionic hematoma with clinical symptoms until delivery. Int J Gynaecol Obstet. 1998;63(2):123-8.
- Ball R, Ade C, Schoenborn J, Crane J. The clinical significance of ultrasonographically detected subchorionic hemorrhages. Am J Obstet Gynecol. 1996;174:996-1002.
- Kim DT, Riddell DC, Welch JP, Scott H, Fraser RB, Wright JR, Jr. Association between Breus’ mole and partial hydatidiform mole: chance or can hydropic villi precipitate placental massive subchorionic thrombosis? Pediatr Pathol Mol Med. 2002;21(5):451-9.
- Hart D. The nature of the tuberose fleshy mole. J Obstet Gynecol Br Emp. 1902;1:479-87.
- Shanklin D, Scott J. Massive subchorial thrombohematoma (Breus’ mole). Br J Obstet Gynecol. 1975;82:476-87.
- Linthwaite R. Subchororial Hematoma Mole (Breus’ mole). J Am Med Assoc. 1963;186:867869.
- Heller DS, Rush D, Baergen RN. Subchorionic hematoma associated with thrombophilia: report of three cases. Pediatr Dev Pathol. 2003;6(3):261-4.
- Gupta R, Sharma R, Jain T, Vashisht S. Antenatal MRI diagnosis of massive subchorionic hematoma: a case report. Fetal Diagn Ther. 2007;22(6):405-8.
- Kocak M, Kandemir O, Sen S, Baskan B, Demir OF. A rare form of abruptio placenta and clinical presentation in a preterm labor case: Breus’ mole. Fetal Diagn Ther. 2006;21(6):540-3.
- Fung TY, To KF, Sahota DS, Chan LW, Leung TY, Lau TK. Massive subchorionic thrombohematoma: a series of 10 cases. Acta Obstet Gynecol Scand. 2010;89(10):1357-61.
- Giri V, Cheema Q, Mulay A. Early-onset severe growth restriction and placental abruption associated with Breus mole. Int J Gynaecol Obstet. 2011;113(1):81-2.
- Nishida N, Suzuki S, Hamamura Y, Igarashi K, Hayashi Z, Sawa R, et al. Massive subchorionic hematoma (Breus’ mole) complicated by intrauterine growth retardation. J Nihon Med Sch. 2001;68(1):54-7.
- Richards DS, Bennett BB. Prenatal ultrasound diagnosis of massive subchorionic thrombohematoma. Ultrasound Obstet Gynecol. 1998;11(5):364-6.
- Nishijima K, Shukunami K, Tsuyoshi H, Orisaka M, Tajima K, Kurokawa T, et al. Massive subchorionic hematoma: peculiar prenatal images and review of the literature. Fetal Diagn Ther. 2005;20(1):23-6.
- Thomas D, Makhoul J, Muller C. Fetal growth retardation due to massive subchorionic thrombohematoma: report of two cases. J Ultrasound Med. 1992;11(5):245-7.
- Kojima K, Suzuki Y, Makino A, Murakami I, Suzumori K. A case of massive subchorionic thrombohematoma diagnosed by ultrasonography and magnetic resonance imaging. Fetal Diagn Ther. 2001;16(1):57-60.
- Steininger H. [On the origin of islands and septa of the human placenta (author’s transl)]. Arch Gynecol. 1978;226(3):261-75.
- Paddock R, Greer ED. Origin of common cystic structure of human placenta. Am J Obstet Gynecol. 1927;13(164-173).
- Stanek J. Acute and chronic placental membrane hypoxic lesions. Virchows Arch. 2009;455(4):315-22.
- Stanek J. Diagnosing placental membrane hypoxic lesions increases the sensitivity of placental examination. Arch Pathol Lab Med. 2010;134(7):989-95.
- Stanek J, Weng E. Microscopic chorionic pseudocysts in placental membranes: a histologic lesion of in utero hypoxia. Pediatr Dev Pathol. 2007;10(3):192-8.
- Stanek J. Placental membrane and placental disc microscopic chorionic cysts share similar clinicopathologic associations. Pediatr Dev Pathol. 2011;14(1):1-9.
- Katz VL, Blanchard GF, Jr., Watson WJ, Miller RC, Chescheir NC, Thorp JM, Jr. The clinical implications of subchorionic placental lucencies. Am J Obstet Gynecol. 1991;164(1 Pt 1):99-100.
- Hong SC, Yoo SW, Kim T, Yeom BW, Kim YT, Lee KW, et al. Prenatal diagnosis of a large subchorionic placental cyst with intracystic hematomas. A case report. Fetal Diagn Ther. 2007;22(4):259-63.
- De Leon-Luis J, Oneson RH, Santolaya-Forgas J. Placental surface cyst with contents less echogenic than amniotic fluid on a second-trimester ultrasonographic evaluation. Ultrasound Obstet Gynecol. 2004;23(6):627-8.
- Brown DL, DiSalvo DN, Frates MC, Davidson KM, Genest DR. Placental surface cysts detected on sonography: histologic and clinical correlation. J Ultrasound Med. 2002;21(6):641-6; quiz 7-8.
- Ferrara N, Menditto C, Di Marino MP, Ciccarelli A, Gerosolima G, Menditto V. Subchorionic placental cyst: histopathological and clinical aspects in two cases. Pathologica. 1996;88(5):439-43.
- Raga F, Ballester MJ, Osborne NG, Bonilla-Musoles F. Subchorionic placental cyst: a cause of fetal growth retardation–ultrasound and color-flow Doppler diagnosis and follow-up. J Natl Med Assoc. 1996;88(5):285-8.
- Kirkinen P, Jouppila P. Intrauterine membranous cyst: a report of antenatal diagnosis and obstetric aspects in two cases. Obstet Gynecol. 1986;67(3 Suppl):26S-30S.
- Shipley CF, 3rd, Nelson GH. Prenatal diagnosis of a placental cyst: comparison of postnatal biochemical analyses of cyst fluid, amniotic fluid, cord serum, and maternal serum. Am J Obstet Gynecol. 1993;168(1 Pt 1):211-3.
- Wasmoen T, Coulam C, Benirschke K, Gleich G. Association of Immunoreactive Eosinophil Major Basic Protein with Placental Septa and Cysts. American Journal of Obstetrics and Gynecology. 1991;165:416-20.
- Bonno M, Kephart GM, Carlson CM, Loegering DA, Vernof KK, Gleich GJ. Expression of eosinophil-granule major basic protein messenger ribonucleic acid in placental X cells. Lab Invest. 1994;70(2):234-41.
- Overgaard MT, Oxvig C, Christiansen M, Lawrence JB, Conover CA, Gleich GJ, et al. Messenger ribonucleic acid levels of pregnancy-associated plasma protein-A and the proform of eosinophil major basic protein: expression in human reproductive and nonreproductive tissues. Biol Reprod. 1999;61(4):1083-9.
- Overgaard MT, Sorensen ES, Stachowiak D, Boldt HB, Kristensen L, Sottrup-Jensen L, et al. Complex of pregnancy-associated plasma protein-A and the proform of eosinophil major basic protein: Disulfide structure and carbohydrate attachment sites. J Biol Chem. 2002.
Figures
Fig 23a: This placenta from a 26-week gestation infant who died in the nursery shows a tumor like mass covering most of the fetal surface (within white arrows).
Fig 23b: The cut sections of the placenta in Fig 23a demonstrate a thrombohematoma (arrows) over much of the fetal surface. There is a pale, older blood layered over redder, more recent blood suggesting two separate events with the first occurring well prior to delivery.
Fig 24: This is a blown up image of the left corner of Fig 23b. The arrows show the placental stem villi being stretched through the thrombohematoma.
Fig 25: This very low magnification photomicrograph shows the fetal surface of the placenta with surface chorionic vessels on the left of the photograph with a thrombohematoma below. Lines of fibrin lamination can be seen in the thrombohematoma (H&E).
Fig 26: This is high magnification microphotograph of the blood in a thrombohematoma in which all of the red cells are sickled indicating that the blood is maternal as fetal cells do not sickle. (H&E)
Fig 27a: This is a very low magnification photograph with the thrombohematoma on the right of the picture and placental villi on the left. The tissue has been stained immunohistochemically to detect fetal hemoglobin as a dark brown precipitate. The light brown staining of the thrombohematoma does not demonstrate fetal hemoglobin in the red cells. (Anti-hemoglobin F)
Fig 27b: This image is a higher magnification of a villus in Fig 27a. The fetal blood within the vessels inside the villus shows the dark brown precipitate that identifies them as fetal cells. (Anti-hemoglobin F, 10x)
Fig 28a: The fetal surface of this placenta demonstrates a raised tan margin that appeared to be a circumvallate placenta with a large Breus mole. The margin however was not typically folded in but appeared simply raised.
Fig 28b: When the placenta in Fig 28a was sectioned, the margins could be seen to be a circummarginal old Breus mole with a second more acute Breus mole reaching from the margin to the center of the placenta (*).
Fig 28c: The placental and membrane surfaces are stained with brown hemosiderin. There is an old marginal hematoma from which hemoglobin must have diffused into the amniotic cavity. On the right side is an old (pale) Breus mole (*). The placenta was diagnosed prenatally as placenta previa in a 24 week-gestation fetus with 24 hours of postmortem intrauterine retention. The mother was delivered by Cesarean section.
Fig 28d: The cut sections of the placenta in Fig 28c demonstrate that a more acute Breus mole underlying the surface fibrin deposition created the raised mass (arrow). The fetal surface is on the left side of the slices. The relationship to the older marginal hemorrhage cannot be determined from the anatomy. There is an equally acute hematoma in the middle of the parenchyma (*) that appeared to be a dissection of the subchorionic thrombohematoma into the parenchyma.
Fig 29a: The fetal surface of this placenta shows a large acute (red) Breus mole occupying the right two thirds of the placenta. This 33 week-gestation stillborn infant was delivered to a mother who had presented with acute abdominal pain. The fetal autopsy was consistent with acute asphyxia and a fetal death that occurred between 4 and 24 hours prior to delivery. A retroplacental hematoma would have been demonstrable after a minimum of 4 hours duration. The timing is consistent with the Breus mole causing acute asphyxia and death. The compressed placental parenchyma suggests that intravillous flow was impeded.
Fig 29b: The cut sections of the placenta in Fig 29c demonstrate the expansion of the Breus mole at both margins of the placenta, but there was no retroplacental hematoma.
Fig 29c: This placenta shows hemoglobin staining of the membranes and cord, and a large Breus mole (*). The placenta was from a 37 week AGA infant, with maternal distress thought to be an abruption, and with Apgars scores of 7/9.
Fig 29d: The maternal surface of the placenta showed no retroplacental hematoma, but the surface was redder beneath the hematoma.
Fig 30: This sliced placenta is from a therapeutic abortion showing the continuity of an acute marginal hematoma and an acute marginal Breus mole (white arrow). There is a central Breus mole that cannot be fully explained by the marginal lesion (pink arrow).
Fig 31a: The fetal surface of this placenta from a stillborn infant shows brown staining of the umbilical cord and membrane surfaces. There is a large, central, tan Breus mole. The staining is due to diffusion of hemoglobin both from the Breus mole and the umbilical cord blood. The image does not show well that there was a short furcate insertion of the cord.
Fig 31b: The cut section of the placenta in Fig 31a shows that the hematoma is brown tan consistent with the autopsy estimate of postmortem fetal retention of 48 hours. The autopsy also demonstrated a double outlet left ventricle. The surface thrombo-hematoma was subchorionic. A torn umbilical vessel would have created a subamnion hemorrhage or bled into the intervillous blood flow. The parenchyma beneath the Breus mole is not compromised. The timing of this Breus mole is consistent with occurring at the time of death, but the mechanism of death is unclear.
Fig 32a: This placenta is that noted in the text. The fetal surface shows a small subchorionic fibrin plaque (arrow).
Fig 32b: The maternal surface of the placenta has been pulled aside to reveal the smooth undersurface of the plaque seen in Fig 32a.
Fig 32c. The ultrasound demonstrates a lucency believed to be the plaque in Fig 32a.
Fig 32d: The microscopic sample of the plaque demonstrates tightly laminated fibrin strands. (H&E, 4x)
Fig 33a: The fetal surface of the placenta shows several small, clear chorionic cysts (arrows)
Fig 33b: The fetal surface shows a large chorionic cyst with some hemorrhage into it.
Fig 34a: This is a photomicrograph of the fetal surface of an immature placenta showing the thick layer of cytotrophoblastic epithelium lining the top of the intervillous space (arrow). (H&E, 4x)
Fig 34b: The fetal surface of the placenta is at the top of the photomicrograph with the pink subchorionic fibrinoid running diagonally above the intervillous space. The arrow points to some trapped residual cytotrophoblast. (H&E, 20x)
Fig 34c: This photomicrograph shows a larger volume of entrapped cytotrophoblast in the subchorionic fibrinoid that have formed a pseudo-lumen that is beginning to form a chorionic cyst (*). (H&E, 4x)
Fig 34d: This photomicrograph shows the expansion of a small surface chorionic cyst with fluid. (H&E, 4x)
Fig 35: The fetal surface of this placenta shows a large, multilobular chorionic cysts filled with dark blood stained fluid. There is also a partial circumvallate margin of the placenta (c).
Fig 36a: The fetal surface demonstrates a large, drooping, thin walled chorionic cyst (arrows) that has a base at a fibrin plaque (“fibrin”).
Fig 36b: The fetal surface of this placenta has a cyst that appears to arise from the partial circumvallate margin.
Fig 37a: This photomicrograph shows multiple chorionic microcysts (arrows) in the chorion layer of the fetal membranes. (H&E, 4x)
Fig 37b: This photomicrograph demonstrates a small cyst in a septum in the middle of the intervillous space. (H&E, 10x)