There were early reports of necrotizing funisitis, such as case 2 of Perrin and Kahn-Vander bel (1). However, the seminal paper in English was published in 1974 by Navarro and Blanc (2). They called the lesion subacute necrotizing funisitis. They provided a detailed gross and microscopic description, demonstrated a clinical correlation with ascending fetal infection and with stillbirth, and speculated that the lesion resulted from the lack of capillary and lymphatic beds in the umbilical cord. The lesion was considered a variant of acute funisitis associated with chorioamnionitis, and as implied by the subacute designation, perhaps due to longer duration of infection, but with the same mix of pathogens (although two cases were associated with Candida krusei). Controversy about the lesion occurred with the publication of a paper suggesting that all cases were due to syphilis. Subsequent published series found syphilis to be a relatively uncommon etiology. Other publications have documented unusual organisms associated with the lesion. I favor the hypothesis that the lesion is not only due to the radial uniformity of the exodus of inflammatory cells from the umbilical vessles, but also due to the presence of microorganisms in the cord which halts the progress of the inflammatory cells out of the umbilical cord. The chemotactic gradient ends in the cord, not in the amniotic fluid.
Incidence:
1) 16 cases from 3000 placental examinations (0.5%) submitted from 35,000 deliveries (0.05%), and 50% of the cases were stillborn (2).
2) 18 cases from 18,848 deliveries (0.1%) and 13 of 45 total cases (28%) were stillborn (3).
3) 45 cases from 3,600 placental examinations (1.25%) submitted from 36,000 deliveries (0.12%) and 10 (22%) were stillborn (or 31% if previable fetuses are counted as stillbirth) (5).
Thus, the lesion is uncommon, approximately 1 per 1000 deliveries. However, there is a 22-50% chance that the infant will be a stillbirth.
Description:
Gross features:
The umbilical cords have chalky or yellow stripes that follow the vessels longitudinally along their helix, i.e. like a barber pole (Fig 1).
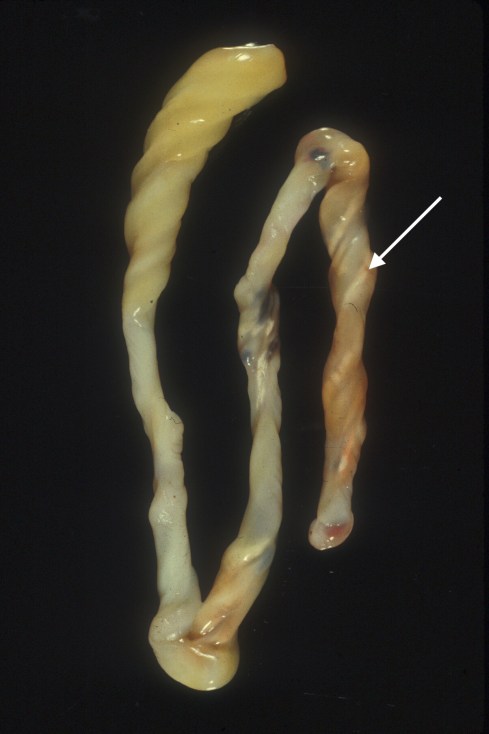
Fig 1) The arrow points to the typical white helix. One end shows the typical yellow, edema of Wharton’s Jelly.
The cord is often discolored yellow or brown, and may appear more translucent and edematous than normal (Fig 2).

F)ig 2a. A dichorionic twin with clouding of the lower placental surface from chorioamionitis, and SNF in that cord only.
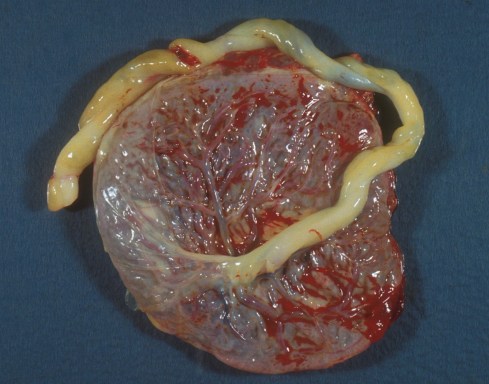
Fig 2b) This cord with SNF shows only the very prominent yellow discoloration.
A portion or all of the cord may be grossly involved. In cross section, the stripe is located between the vessels and the cord surface, usually forming an arc or circle paralleling the vessel surface (Fig 3).
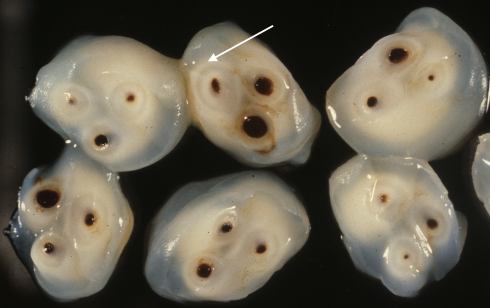
Fig 3a) The arrow points to a rim of opacity (?calcification) between the vessel and the surface characteristic of SNF

Fig 3b) This cross section shows that the white opaque rim tends to be between the surface and the vessels.
The calcification may be demonstrable radiographically (4) (Fig 4).

Fig 5) The presumed calcification of the SNF lesion can be seen in the umbilical cord stump (arrow).
Microscopic features:
The most characteristic feature is the crescent of necrosis, often calcified, that is interposed between the umbilical surface and the vessel walls. Both arteries and vein are usually involved. Commonly there is acute funisitis, often with a gradient of viable neutrophils near the vessel leading to an accumulation of distal necrotic neutrophils (Fig 5).

Fig 5a) The basophilic rim of neutrophils and necrosis at low power.
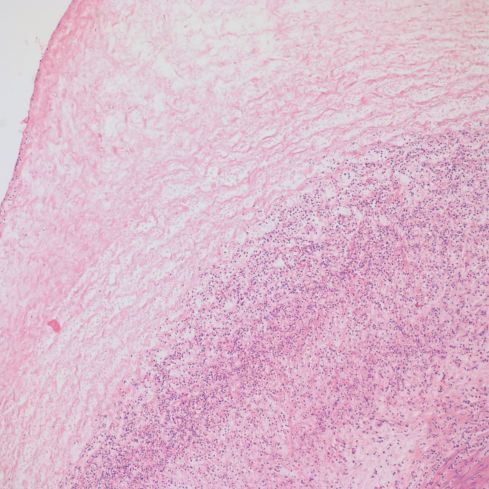
Fig 5b) A medium power showing the inner basophilic band and an outer band of coagulation necrosis.

Fig 5c) A high power image showing basophilic neutrophils and debris, and then more superficial coagulation necrosis including neutrophils necrosis.
The microorganism causing the inflammation may result in different histologic pictures including those with chronic inflammation and different patterns of necrosis (see below)
Almost all reported cases are associated with chorioamnionitis. In the rare case of SNF and no membrane inflammation, there are two logical possibilities. Either the mother cannot respond to an amniotic infection or the organisms are reaching the cord via the fetal circulation. Jacques and Qureshi found no correlation between the grade of funisitis and the grade of chorioamnionitis5. They graded the funisitis as mild, moderate or severe based on subjective evaluation of the extent of inflammation necrosis and calcification.
The major differential diagnosis is routine funisitis, which may demonstrate neutrophils surrounding the vessel in Wharton’s jelly, but does not show a band of necrosis. Calcification in the necrotic areas is common, but not necessary for the diagnosis of SNF. Thrombosis of an umbilical artery may produce a pattern of necrotizing inflammation that mimics SNF but the inflammation is oriented toward the necrotic artery, not toward the periphery of the cord (Fig 6).

Fig 6a) The cord is stained red in a liveborn infant due to hemoglobin diffusion from an umbilical artery thrombus. The arrow points to a white streak running with the vessel.

Fig 6b) The layer of necrosis (arrow) is between the vein and the thrombosed artery, not toward the surface as expected in SNF.
Clinical and diagnostic implications:
In Navarro and Blanc’s original report, the major clinical associations were stillbirth and newborn infection (2). They also suggested recurrent infection, but this was poorly documented. Craver and Baldwin in 60 cases found histologic and microbiologic features similar to Navarro and Blanc (3). They compared a subset of their necrotizing funisitis cases with controls having acute funisitis. The incidence of stillbirth and neonatal infections were significantly increased (P < .05). There was also an association with prematurity and possibly with growth retardation, although many infants were normal weight. The authors suggested that calcification and neovascularization (essentially granulation) associated with progressively longer duration of membrane rupture. Jacques and Qureshi reviewed 45 cases of necrotizing funisitis and demonstrated a high incidence of stillbirth [10], but a lesser incidence of neonatal infection (5). Of the 18 infants with medical records available that were discharged, one had positive E. coli from urine and the eye, and one grew Haemophilis from the conjunctiva. However, the majority did not have significant positive cultures despite evaluation for sepsis. Thirteen mothers had symptomatic chorioamnionitis. One mother had an intrauterine device. 28 mothers had prolonged rupture of membranes (defined as > 24 hr. between rupture of membranes and delivery). One case was a twin with the other twin not demonstrating the lesion.
Fojaco et. al. found 16 cases with necrotizing funisitis, all with congenital syphilis, from a database of 465 perinatal autopsies (6). The database had 35 cases of congenital syphilis (32 stillbirths, 3 neonatal deaths), which results in 46% with necrotizing funisitis, but no cases of necrotizing funisitis without syphilis .Their autopsy database is weighted with a high incidence of syphilis, but the results still seem anomalous. In Creaver and Baldwin’s study, clinically, three of their patients had candida, one acute genital herpes, but none syphilis (3) In Jacques and Qureshi’s study, five mothers had syphilis of which 3 infants were stillborn, and 2 died on the first day of life (5). Together those two studies had 30 cases of stillbirth with necrotizing funisitis without syphilis. Also of note is that herpes virus and actinomyces meyeri have been identified in cords with necrotizing funisitis (7, 8). In my experience, toxoplamosis can also produce a lesion that appears to be SNF (Fig 7).

Fig 7a) The mineralization follows the vessel but appears to reach the surface from this cord with toxoplasmosis.
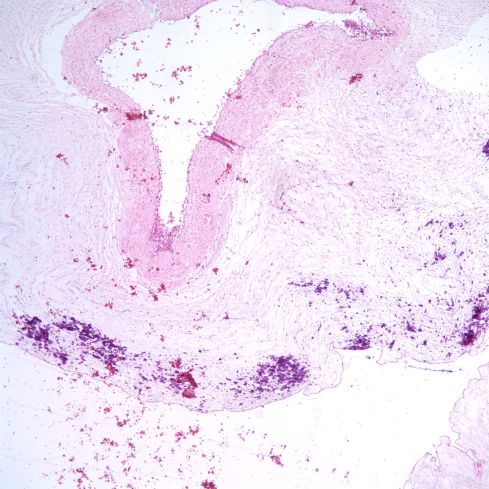
Fig 7b) In this low power image of the same cord, the mineralization can be seen to superficial but otherwise similar to SNF.
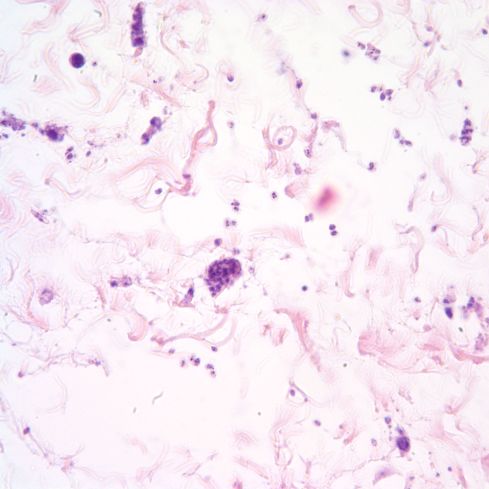
Fig 7c) Toxoplasma are within all of Wharton’s Jelly.

Fig 7d) This is an anti-toxoplasma immuno-stain of the same region of Wharton’s Jelly.
The microscopic features described by Fojaco et. al. differ from typical cases of necrotizing funisitis. Viable neutrophils were not usually present, and necrosis and calcification directly involved the umbilical vessels. Both grossly and microscopically, the necrosis appeared to lack a clear zone between the vessel and the necrotic band. The band appeared more circumferential, than crescentic. Similar findings appeared in the study of Schwartz et. al. of the umbilical cord in congenital syphilis (9). In that study, there was a subtle shift of the definition of necrotizing funisitis: “Necrotizing funisitis was diagnosed when a predominantly mononuclear population of inflammatory cells, usually composed of lymphocytes and macrophage-like cells having “smudged” nuclear features, was present in the umbilical vessels and mesenchyme, accompanied by necrosis and disruption of blood vessel walls” (9). This shift of definition is repeated in the discussion citing Navarro and Blanc’s paper as funisitis characterized by “lymphoplasmacellular inflammation” (9) This is somewhat of a misquote as the referenced paper states: “The predominant inflammatory cell was the neutrophilic leukocyte; there were also relatively large numbers of lymphocytes and monocytes, particularly at sites of endovasculitis” (2). Thus, this shift of the definition suggests that absence of neutrophils and of vascular wall necrosis may be more common in the syphilitic versus the non-syphilitic cases of necrotizing funisitis. However, Jacques and Qureshi did not find distinctive features in their syphilitic cases (5). A spirochete stain will often demonstrate organisms in Wharton’s jelly with syphilis even without necrotizing funisitis 9(although prenatal maternal antibiotic therapy may eliminate organisms).
Thus, one can conclude that SNF1) is almost always associated with chorioamnionitis, and with a wide range of cultured organisms including bacteria, fungi, and herpes virus, 2) is possibly associated with chronicity of chorioamnionitis, 3) may occur with congenital syphilis, 4) is only weakly associated with neonatal infection, and 5) is associated with stillbirth (by an undefined mechanism).
Etiology and pathogenesis:
The unusual histologic picture is a clue to pathogenesis, and has often been likened to an ochterlany plate. In such a plate, antibody is in one well and antigen is in a distant well. Both antigen and antibody diffuse radially in media surrounding the wells. Where they meet in sufficient concentration, a crescent of precipitation occurs. In this analogy, the neutrophils “diffuse” from the vessel until they meet something diffusing from the umbilical surface. At this junction, there is a reaction that leads to a zone of necrosis analogous to the antibody-antigen precipitation in a ochterlany plate. This distinct geometric appearance is possible because there is no complex capillary bed anatomy. Why does “precipitation” occur in this situation, rather than free migration of neutrophils to the surface of the cord?. One plausible explanation is that the neutrophils are encountering their target, the organism. In the usual funisitis they are simply following a chemotactic stimulus to the amniotic fluid. However, if organisms are entering the cord, the ingestion of organisms or virally infected cells begins at the front between exiting neutrophils and entering organisms. Some of the organisms associated with necrotizing funisitis have been identified in the Wharton’s jelly including Treponema pallidum, Actinomyces meyeri, and unspecified bacteria. This theory cannot be directly proven since many cases to not demonstrate microorganisms with histological examination. However, some cases of SNF demonstrate neutrophils on the vascular side, the necrotizing zone in the middle, and numerous bacteria on the surface side of the umbilical cord (fig 8).
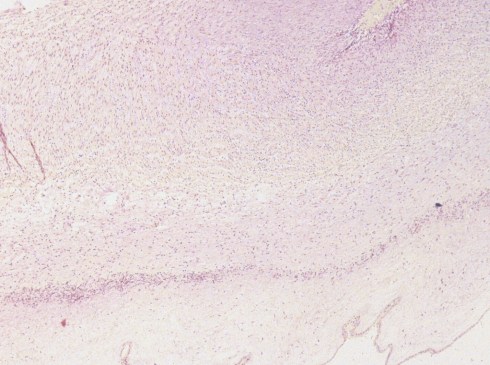
Fig 8a) Low power of an early/ mild, but typical. SNF lesion

Fig 8b) From the same cord, a higher power shows numerous short bacterial rods on the surface side of the band of necrosis.
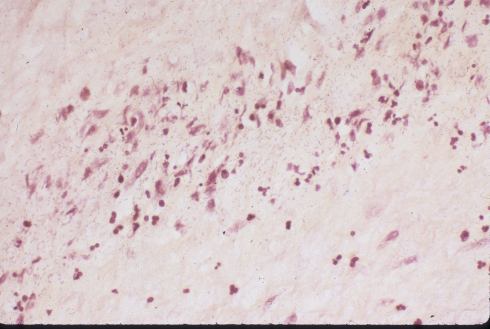
Fig 8c) A different case, but with similar findings. These were both gram negative rods, but other cases had gram positive cocci.
Candida is usually invasive at the surface, but its frequent inclusion as an organism in SNF could be interpreted as evidence of deeper invasion into Wharton’s Jelly (Fig 9).

Fig 9a) A cord with both features of SNF and white plaques of candida on the surface.
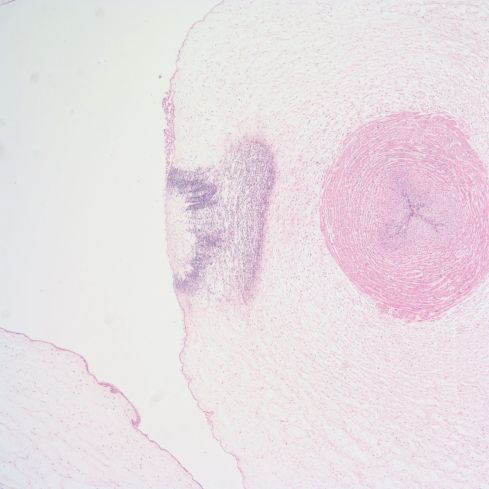
Fig 9 b) The low power show a focal area with the features of SNF.
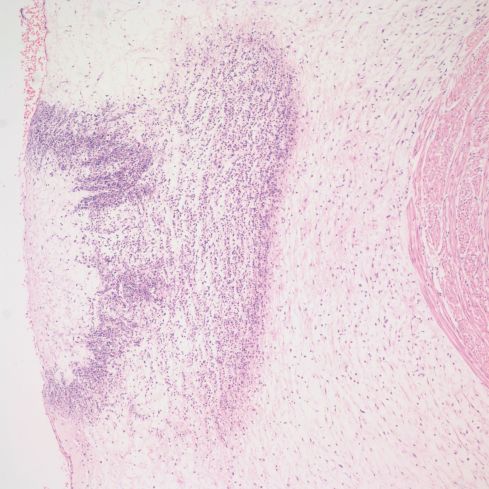
Fig 9 c) Medium power of the same lesion.
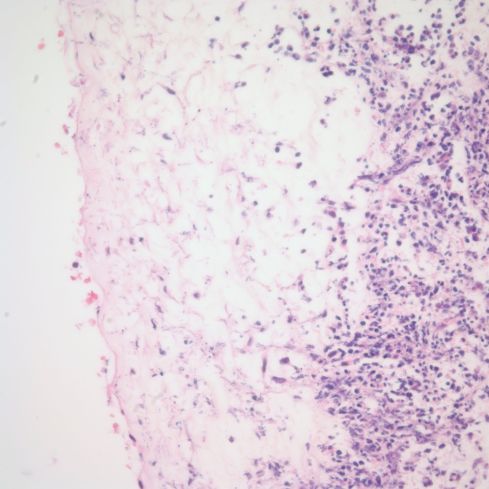
Fig 9d) High power shows neutrophils and necrotic debris on the right, and slightly basophilic hyphae on the left.

Fig 9e) Silver stain showing the invasive pseudohyphae of candida invading into Wharton’s Jelly.
Similarly, the occurences of SNF with HSV virus appears to reflect deeper invasion into Wharton’s jelly (Fig 10)

Fig 10a) The umbilical cord shows minimal findings of SNF.
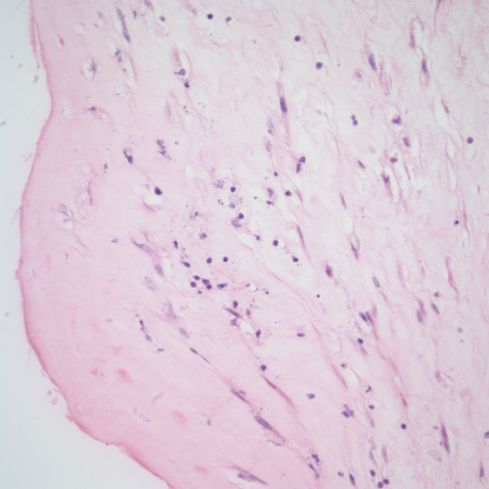
Fig 10b) Just beneath the surface there is a chronic inflammatory infiltration and necrotic cells.

Fig 10c) In some areas there appear to be plasma cells.
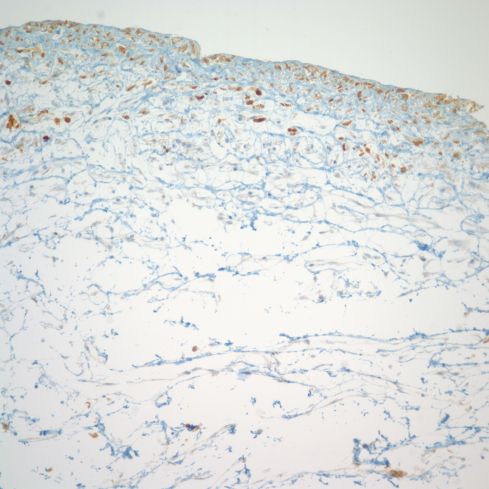
Fig 10d) An anti-HSV immuno-stain demonstrates that the cells in Wharton’s Jelly had become infected with HSV.
The yellow edematous appearance of the cord with SNF is likely due to the leakage of serum/plasma from the umbilical vessels in response to inflammation. Calcification is probably a secondary feature of the tissue necrosis and digestion that create charged molecular debris that binds serum calcium.
The variation in pattern is some cases could be the result of the response elicited by the organism. For example, syphilis might initiate a chronic response, which would not only favor chronic inflammatory cell migration, but also modify the types of chemical mediators, which could alter the pathology, perhaps favoring vascular necrosis. Silver stains frequently demonstrate great numbers of spirochetes in Wharton’s jelly that might stop many inflammatory cells from extending far from the umbilical vessel (Fig 11) (9).

Fig 11a) SNF lesion in a stillborn infant with syphilis.

Fig 11b) Higher power demonstrating chronic inflammation in the umbilical vein media.

Fig 11c) Silver stain demonstrating a spirochete (arrow). They were through out Wharton’s Jelly.
If the hypothesis that necrotizing funisitis results from direct microbial invasion of the umbilical cord were true, it would still not explain why some microorganisms become invasive. Is it a property of the organism, of the duration of infection, of the dose of the organism, or some other host factor?
References
- Perrin EVD, Kahn-VanderBel J. Degeneration and calcification of the umbilical cord. Obstet Gynecol 1965; 26:371-376.
- Navarro C, Blanc W. Subacute necrotizing funisitis. J Pediatr 1974; 85:689-697.
- Craver RD, Baldwin VJ. Necrotizing funisitis. Obstet Gynecol 1992; 79:64-70.
- Schiff I, Driscoll SG, Naftolin F. Calcification of the umbilical cord. Am J Obstet Gynecol 1976; 126:1046-1048.
- Jacques SM, Qureshi F. Necrotizing funisitis a study of 45 cases. Hum Pathol 1992; 23:1278-1283.
- Fojaco RM, Hensley GT, Moskowitz L. Congenital syphilis and necrotizing funisitis. JAMA 1989; 261:1788-1790.
- WrightJr JR, Stinson D, Wade A, Haldane D, Heifitz SA. Necrotizing funisitis associated with Actinomyces meyeri infection: a case report. Pediatr Pathol 1994; 14:927-34.
- Heifetz SA, Bauman M. Necrotizing funisitis and herpes simplex infection of placental and decidual tissues: a study of four cases. Hum Pathol 1994; 25:715-722.
- Schwartz DA, Larsen SA, Beck-Sague C, Fears M, Rice RJ. Pathology of the umbilical cord in congenital syphilis: analysis of 25 specimens using histochemistry and immunofluorescent antibody to Treponema pallidum. Hum Pathol 1995; 26:784-791.