The heart
Gross lesions:
Pericardial and pleural effusions:
After opening the chest, the volume of pericardial and pleural effusions can be measured or estimated. A 20 ml Toomey syringe will be successful. The pericardial fluid can be removed using a sharp needle on the syringe. Prenatal ultrasound has identified some normal fetal pericardial fluid1,2, but an abnormal effusion leaves visibly distends the pericardial sac (Fig 1a,1b).
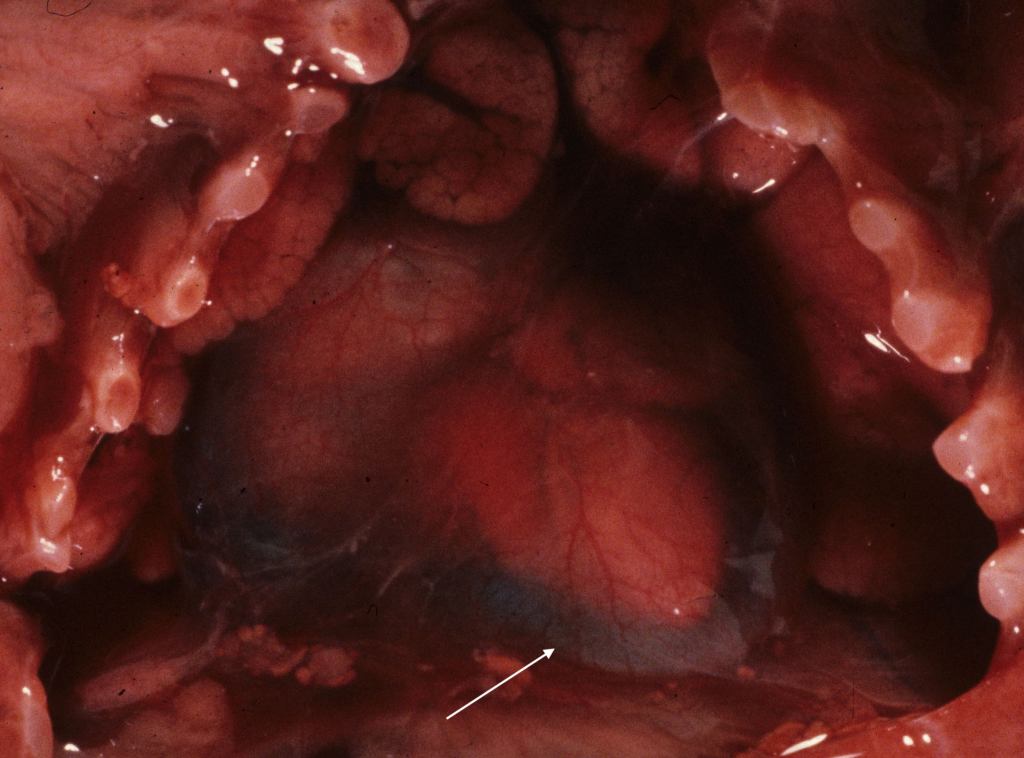
The stillborn infant had heart failure with nonimmune fetal hydrops from familial polyvalvular dysplasia.
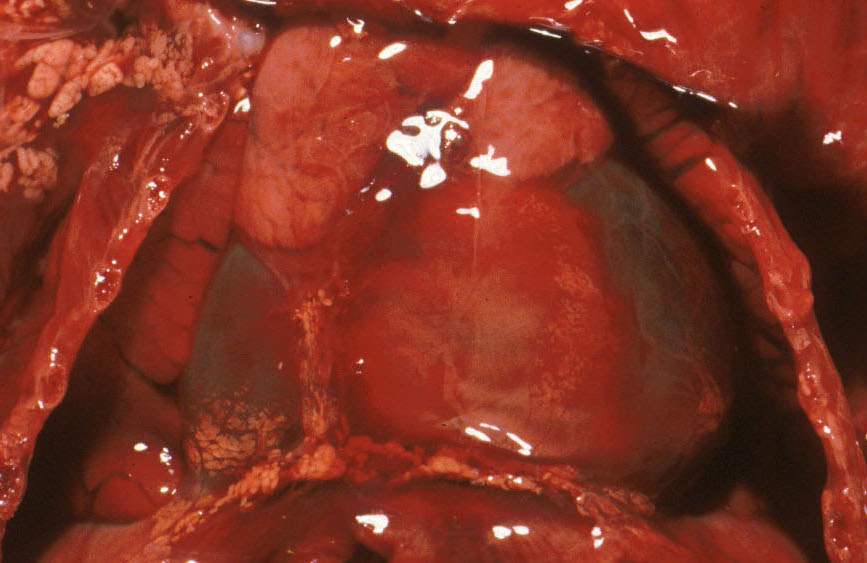
The fluid may be clear, or with the leakage of hemoglobin into tissue as part of autolysis, the fluid may be deep red, but still without solid components. This is still a serous effusion, not a hemorrhage. Old blood or pus is murkier from protein and dead cells.
Heart size:
The size of the heart can be judged to some degree by how much it fills the chest cavity. (Fig 3).

The diagnosis of cardiomegaly is ultimately determined by the heart weight. The mean heart: brain weight ratio is .053. To avoid adding the weight of lumen blood, the heart is weighed after the complete dissection which will have removed any luminal blood. Heavy, but normally formed, hearts are a common finding in infants of diabetic mothers (Fig 4). Such infants typically are macrosomic and show increased subcutaneous adipose tissue. The heart: body weight ratio in these macrosomic infants may exceed .005.

The “size” of the heart on inspection is also a function of the dilation of the chambers. The degree of distention of the atria and of the ventricles can be documented by a digital image best taken after the thymus has been removed. Normally a small portion of the left ventricle can be seen when the chest is opened. With ventricular dilatation the left ventricle appears to bulge beyond the usual contour (Fig 5a-5d). With atrial dilatation, the atrial appendage is often distended with blood.


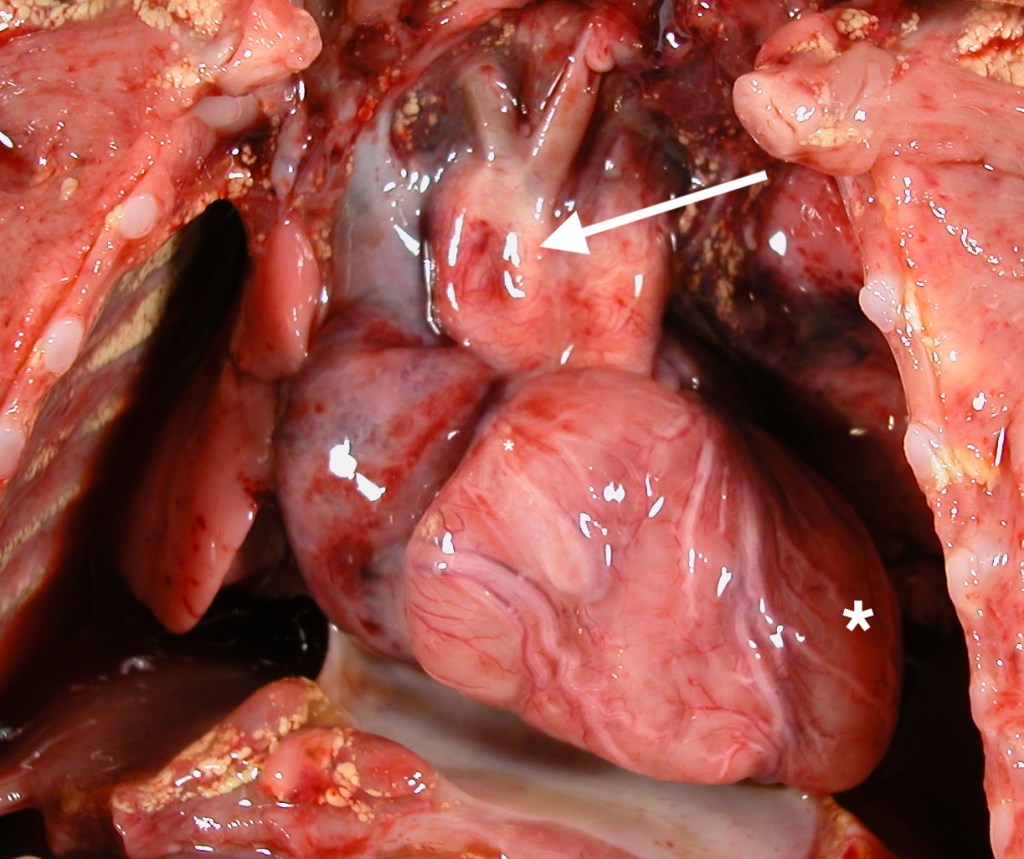
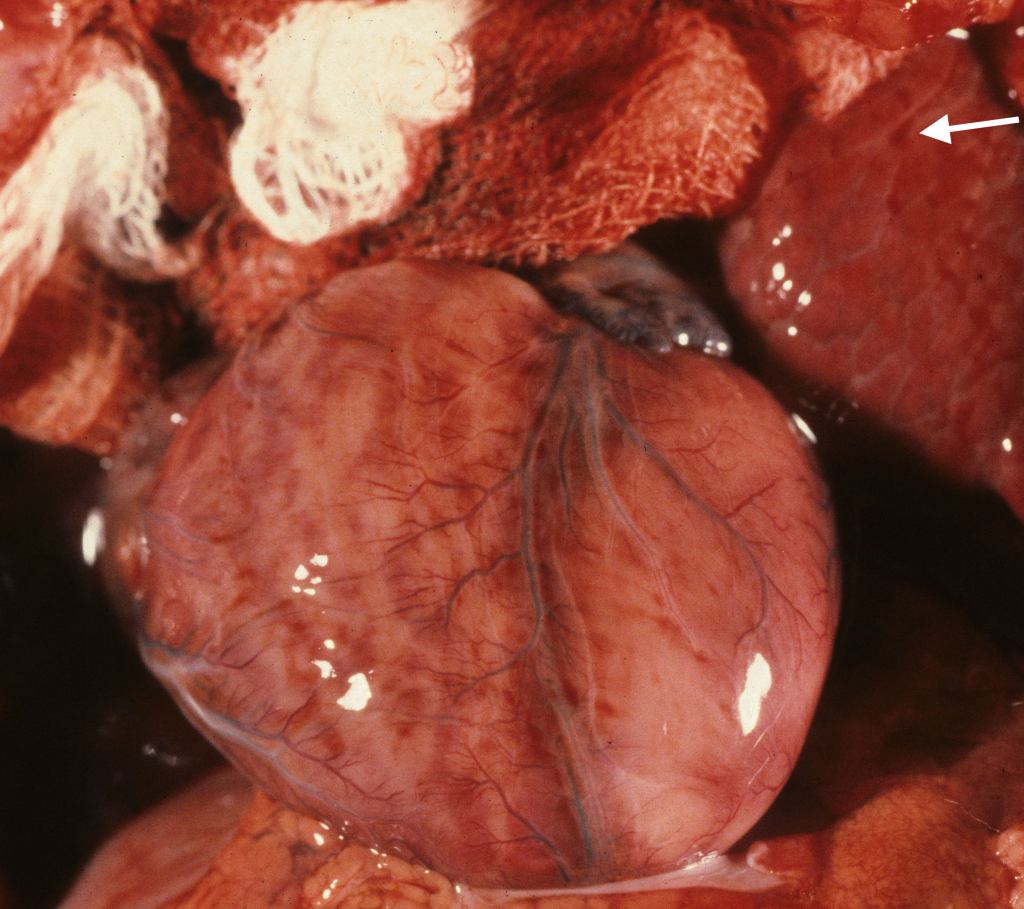
External examination of the heart:
Of heart malformations, stillbirth is most often associated with lesions that elevate right sided heart resistance, and lead to right sided heart failure and anasarca (non-immune fetal hydrops). The cause underlying this association is likely due to the elevated pressure on umbilical venous return. Some outflow lesions may also be associated with disruption of nodal pathways that can lead to tachyarrhythmias and high out put cardiac failure. Many congenital heart malformations, such as hypoplastic left heart, VSD, or transposition, do not compromise cardiac function until after birth and transition from the fetal to the permanent circulatory pattern. Other cardiac lesions may be associated with syndromes that have an increased risk of stillbirth, such as common atrio-ventricular canal in trisomy 21. A systematic examination of the heart during the autopsy of a stillborn infant is critical to detecting the wide array of congenital heart anomalies. The pathologist can note the normal and document the abnormal, often with digital images. The intent of this manual is to guide the autopsy of fetal and peripartum death, and it cannot cover these malformations in depth. There are many excellent textbooks and monographs that present cardiac malformations.
The external heart examination begins with noting the situs and direction that the apex points. The removal of the thymus exposes the innominate vein crossing from the left subclavian vein to the right superior vena cava. If this is absent, then there is likley a left superior vena cava entering the coronary sinus that may warn of possible other venous or lymphatic abnormalities. (Fig 6a, 6b).
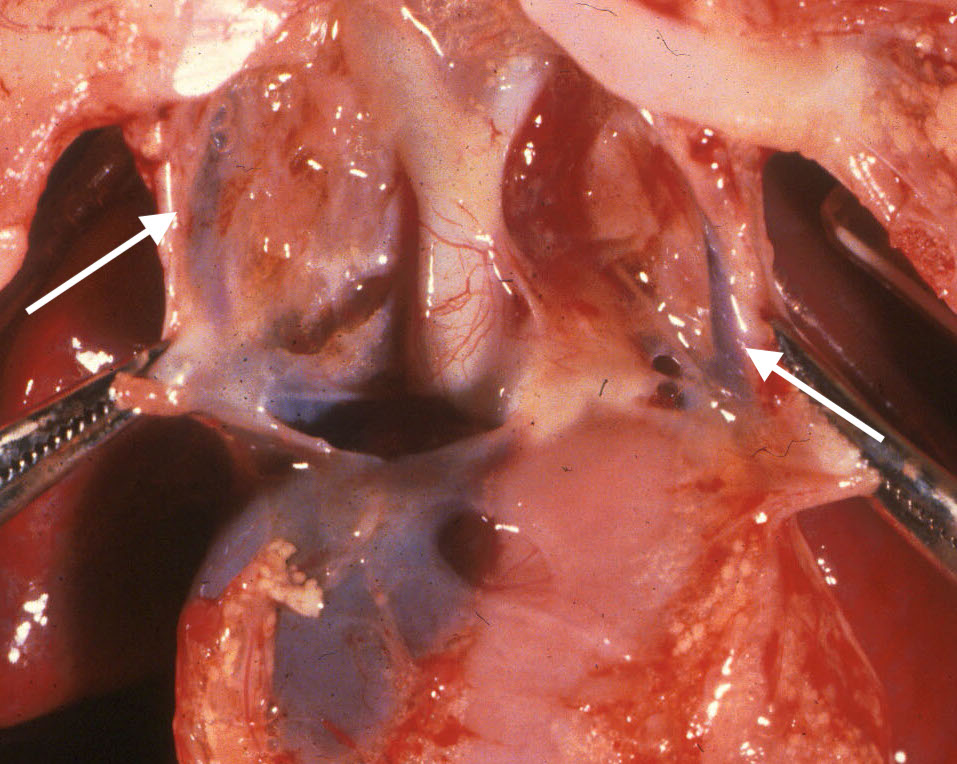
After the pericardium is completely removed, the color of the heart should be noted. It may be pale from anemia, red stained from hemolysis of postmortem retention, mottled from lipid deposition, or dotted with petechiae (Fig7a, 7b).
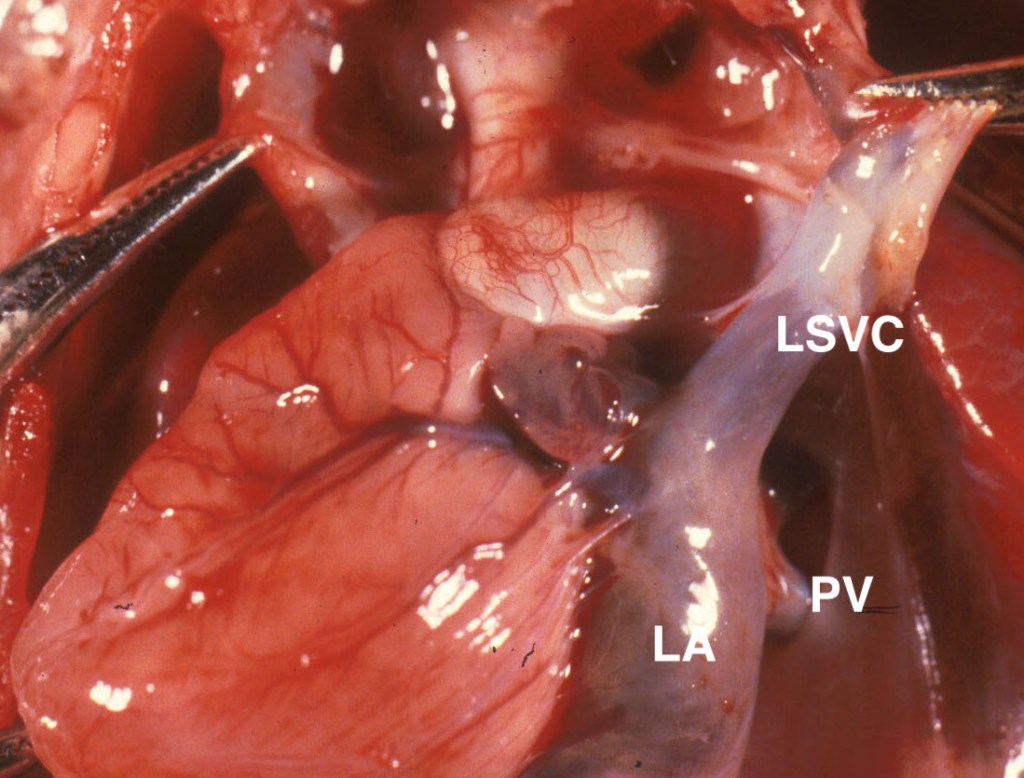
The position of the atria can be determined by identification of the pyramidal right atrial appendage and the chicken wing like left atrial appendage. The right atrium normally lies superficial to the left. The anterior surface of the heart facing the pathologist should normally show the right ventricle with a conus leading up to the pulmonary artery. The pulmonary artery should be superficial to the aorta and directed to the left, while the aorta should be deep, and directed to the right of the infant. These two great arteries should be of equal diameter. If one is narrower, it is evidence of an underlying malformation interfering with cardiac outflow (Fig 8).

The left anterior descending artery defines the ventricular septum. The right ventricle is positioned more anteriorly, but both ventricles are normally of approximately the same size. The aorta normally arises from the left ventricle and sweep first right, then left to follow left of the vertebral column to the diaphragm. An aorta that runs along the right of the vertebral column may form a vascular ring that the junction with the ductus arteriosus can compress the esophagus and impede fetal swallowing (Fig 9). This can result in polyhydramnios. The heart next is lifted outward and the four pulmonary veins visualized.
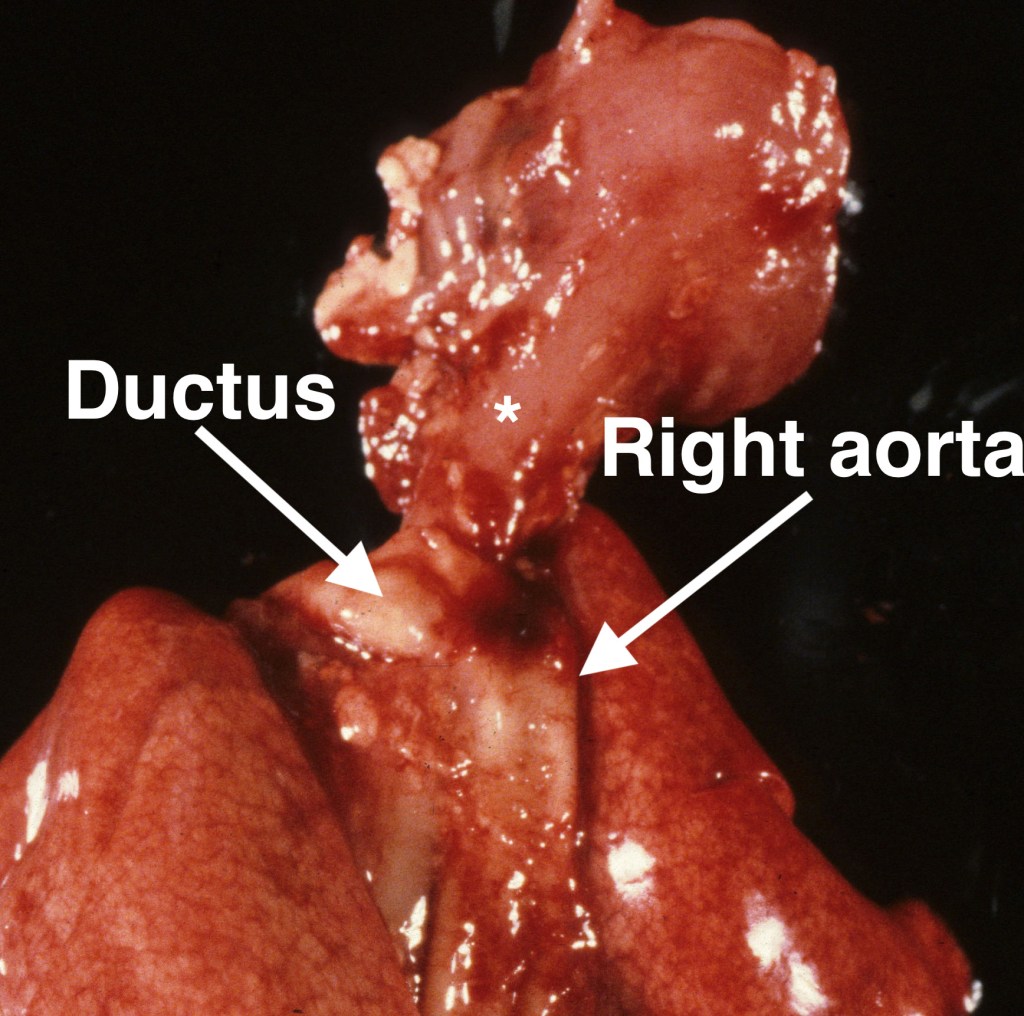
The ductus arteriosus and aortic isthmus
The left lung can be lifted out of the chest (a piece of gauze may make gripping the lung easier). Then, peeling back the pleura over the descending aorta, will provide a clear view of the ductus arteriosus and aortic isthmus. The ductus is a continuation of the pulmonary artery between takeoff of the right and left pulmonary arteries to the junction with the aorta. The aortic isthmus starts at the left subclavian artery and continues until the junction of the ductus with the descending aorta. As over the anterior heart, the relative diameters of these two vessels reflects relative flow. The aortic isthmus is often narrower than the ductus. If it is more than 50% narrower, this is likely to reflect an abnormality. The narrow isthmus is a common finding in some cases of hydrops particularly with early jugular lymphatic obstruction sequence (Fig 10a, Fig 10b).
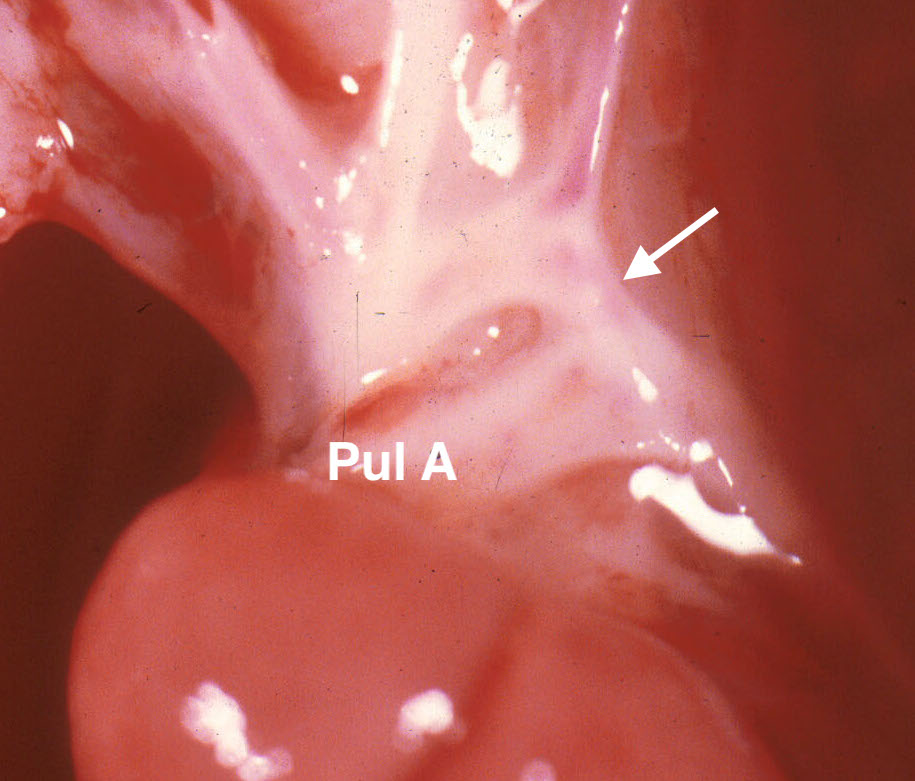
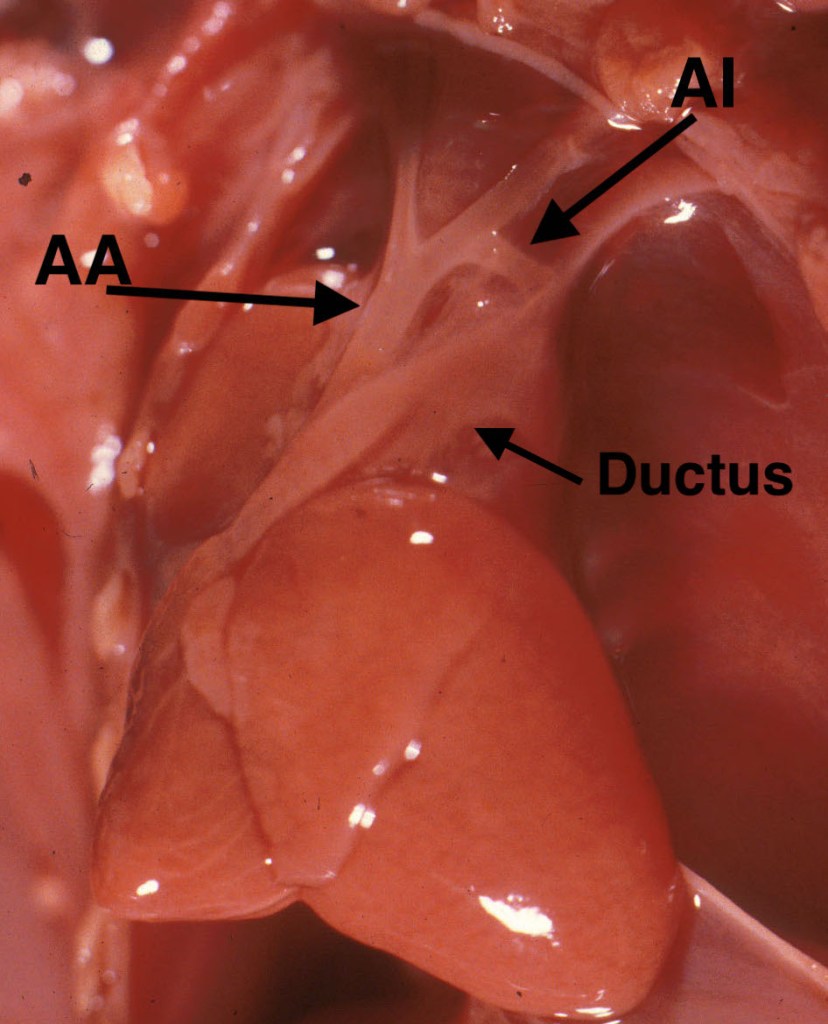
The dynamics of such disturbed fetal blood flow and its relationship to the development of aortic coarctation is unclear. If the ductus arteriosus is narrow, this may be from decreased flow usually in early gestation (Fig 11a, 11b)

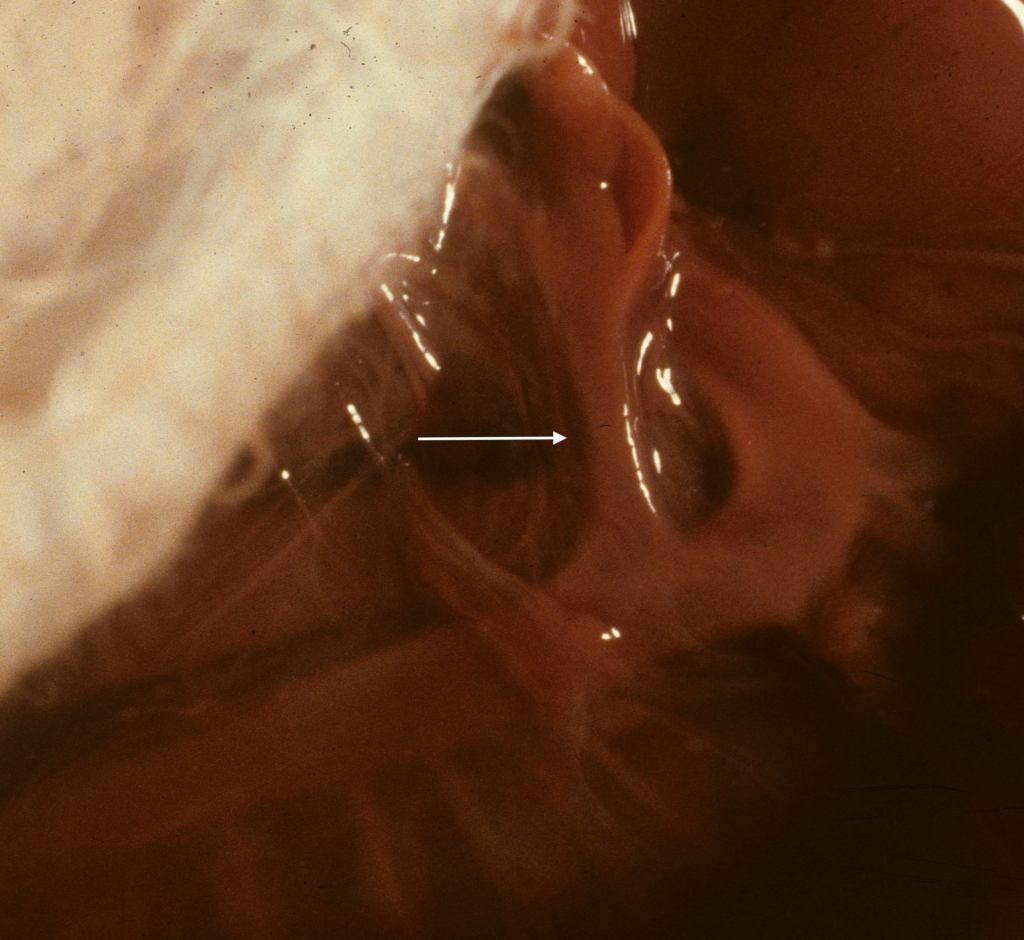
or from ductal contraction in later gestation (Fig 12a-c).


As with a narrow isthmus, the hemodynamics creating a narrow, uncontracted ductus in younger gestation fetuses is not understood. In older gestation fetuses the ductus responds to oxygen and prostaglandins as it must after birth to close the fetal circulation. Indomethacin is commonly used to close a patent ductus arteriosus in neonates. Indomethacin used to treat preterm labor has resulted in fetal ductal constriction/closure3,4. Whether other NSAIDS can contract the ductus in utero is unclear. The consequences of ductal closure in utero would block outflow from the right ventricle5,6.
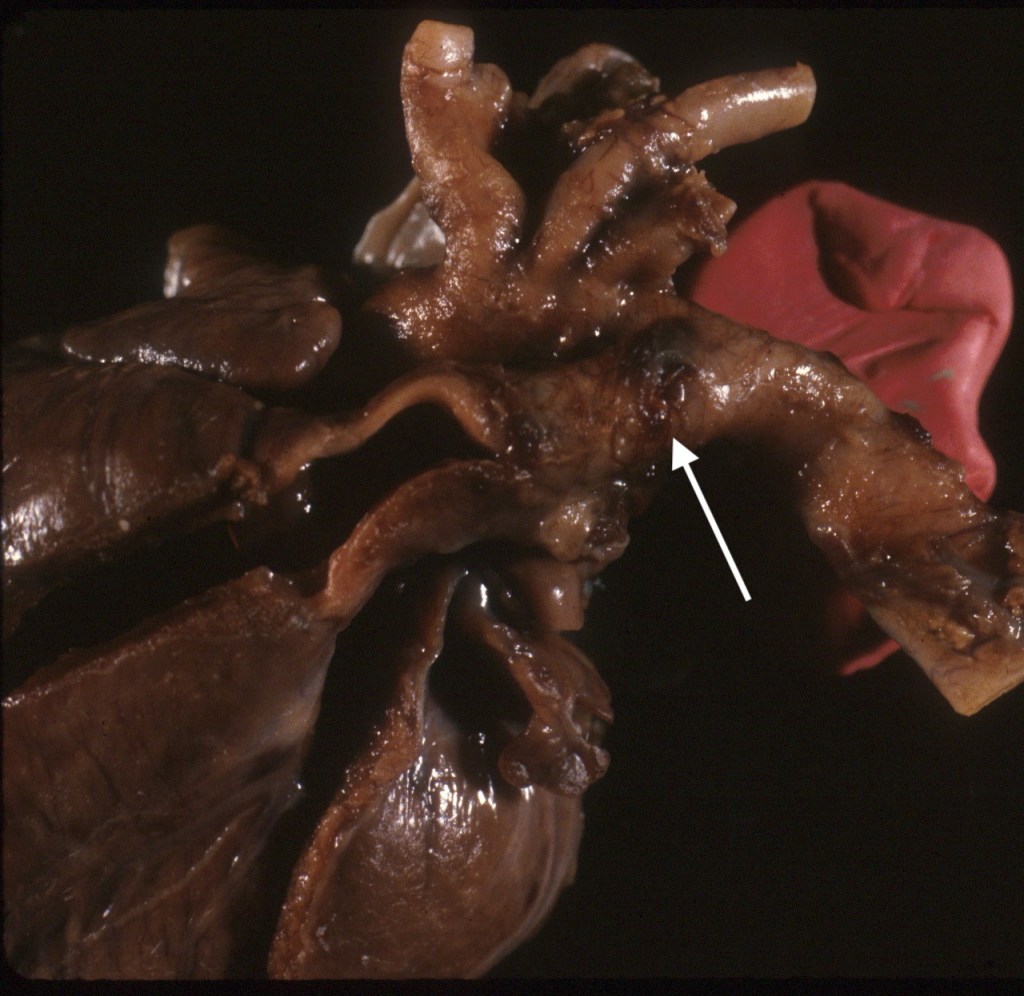
An unexplained but common autopsy finding in stillborn infants is a small adventitial hemorrhage of the ductus arteriosus. This may be an effect of ductal contraction (Fig 13a-c).
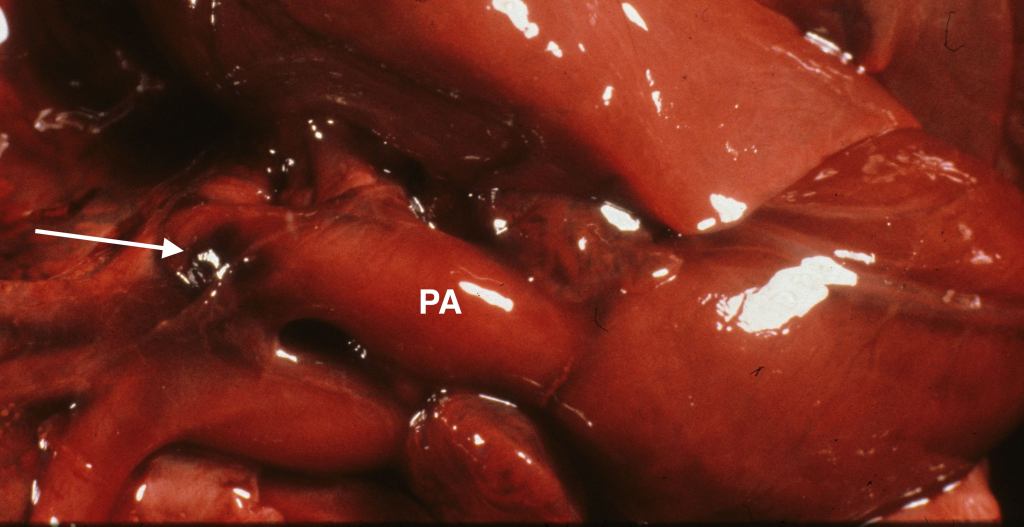
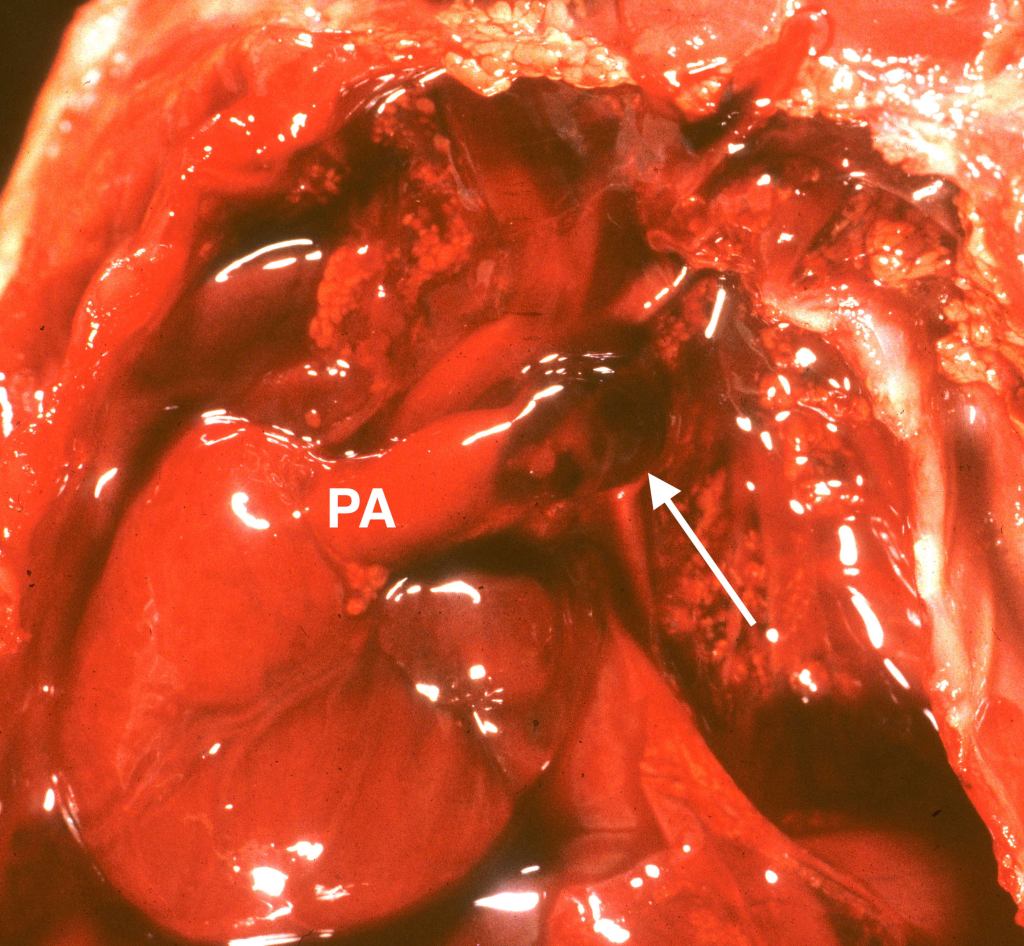
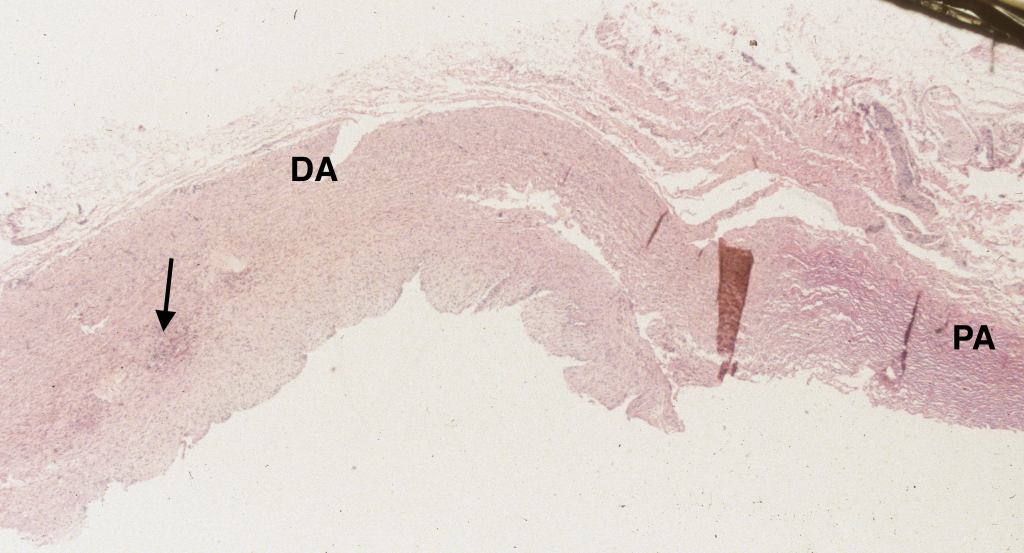
The heart dissection:
Dissecting the small hearts of stillborn infants in situ maintains the relationships to vessels and other organs prior to removal of the heart. One technique is to open the right atrium from the superior vena cava into the inferior vena cava in the plane parallel to the chest. This will expose both the atrial septum and the tricuspid valve. The extent of coverage of the septum secundum over the foramen ovale can be observed, and normal right to left probe patency can be demonstrated. The tricuspid valve can be observed for its position, normal valve leaflets, and possible vegetations. Then under direct observation, the lateral margin of the right ventricle can be opened, then scissors turned toward the base and cut following the ventricular septum into the anterior surface of the pulmonary artery. After the conal bands and the semilunar valves have been observed, the cut can continue through the ductus arteriosus into the descending aorta. With this opening, the entry of the isthmus is exposed and can be probed to identify coarctation. Then the left atrium can be entered from the appendage and a cut made along the lateral atrial and ventricular margins which will visualize the mitral valve. The cut can then be continued into the left ventricle, and as on the right side, the scissors can be turned toward the cardiac base and by cutting parallel to the septum and slipping the top blade of the scissor beneath the pulmonary artery the aorta can be opened, likely through the non-coronary cusp. The semilunar valve leaflets and coronary ostia can be seen, and the main arterial branches identified.
Cardiac malformations, if present, need to be accurately described. Some lesions can lead to right heart failure and result in non-immune fetal hydrops (anasarca). These lesions include absent or dysmorphic tricuspid valves (Fig 14), pulmonic stenosis (Fig 15), or lack of right ventricular volume such as Eisenmenger anomaly.

A vascular ring of the great vessels can be a cause of polyhydramnios if it interferes with the swallowing of amniotic fluid. An unusual cause of fetal hydrops in a cardiac rhabdomyoma, usually associated with tuberous sclerosis, protruding into the right ventricle (Fig 16).

Rarely, there is iatrogenic trauma. Some outflow lesions may also be associated with disruption of nodal pathways that can lead to tachyarrhythmias and high output cardiac failure. Some cardiac lesions may be associated with syndromes that have an increased risk of stillbirth, such as common atrio-ventricular canal in trisomy 21. Many congenital heart malformations, such as hypoplastic left heart, VSD, or transposition, do not compromise cardiac function until after birth and transition from the fetal to the permanent circulatory pattern. Knowing the clinical history and following a systematic examination of the heart during the autopsy is critical to detecting the wide array of congenital heart anomalies. The autopsy report can then note the normal and document the abnormal, often with digital images. Abnormalities may be photographed in situ and/or after heart removal including endocardial thickening and valve lesions, as well as malformations (Fig 17). There are many excellent textbooks and monographs that present cardiac malformations.
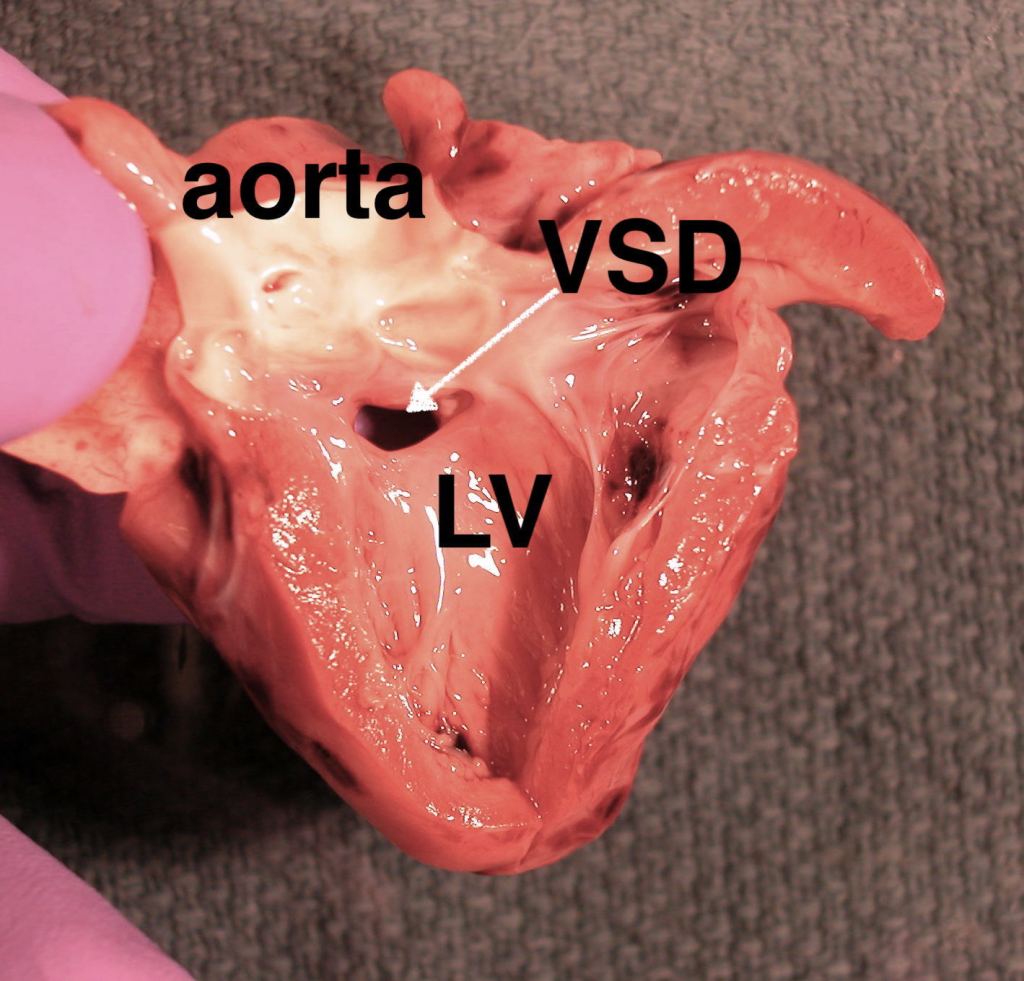
The heart, either after dissecting or after the removal of the organ block, can be separated at the great vessels. The heart weighed at this point in the dissection should have all luminal blood removed. The valve circumferences can be measured, as well the ventricular chamber diameters midway to the apex, and the thickness of free wall measured at the same location. One approach to histological sampling of a normally formed heart is to sample both ventricles with a longitudinal slice from near the apex, including papillary muscle, and extending through the atrio-ventricular valve. In very small hearts, appropriate sampling can document some malformations (Fig 18).

Microscopic findings
Postmortem retention:
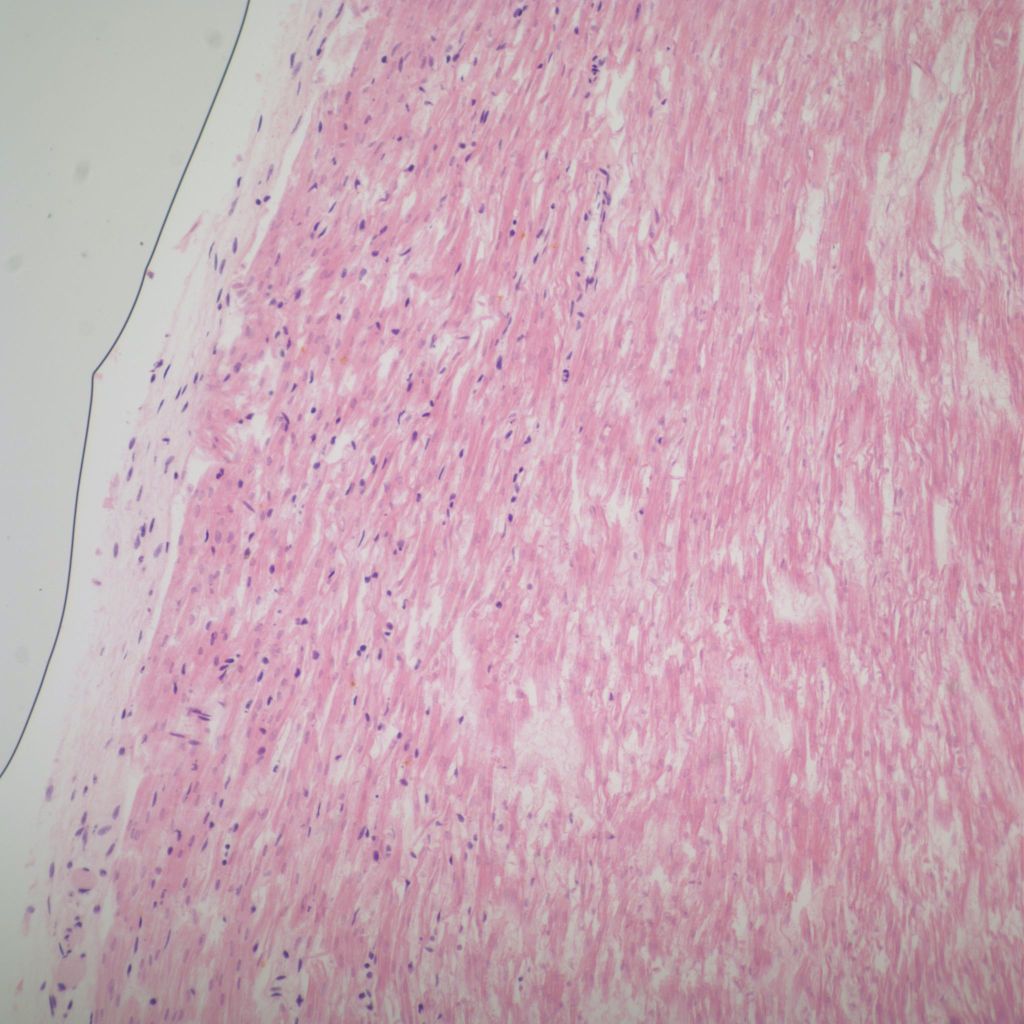
The findings of Dr. Genest and colleagues were that the inner third of the myocardium has lost basophilia after 24 hours and the outer third after 48 hours of postmortem retention7 (Fig 19). (Loss of basophilia is defined by at least 1% of nuclei entirely devoid of basophilic staining).
Epicardial surface:
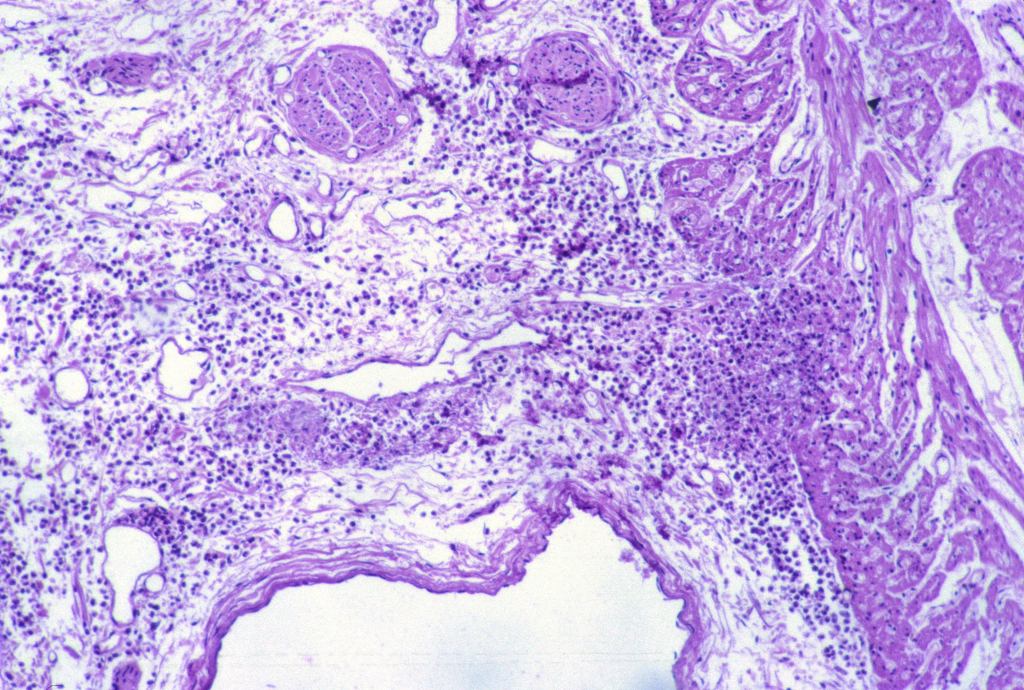
The microscopic sections should confirm the location of petechial hemorrhages and the size of confluence of hemorrhages. There is often congestion of venules and capillaries (Fig 20). A small hemorrhage is often deep to the surface coronary arteries (Fig 21a, b).


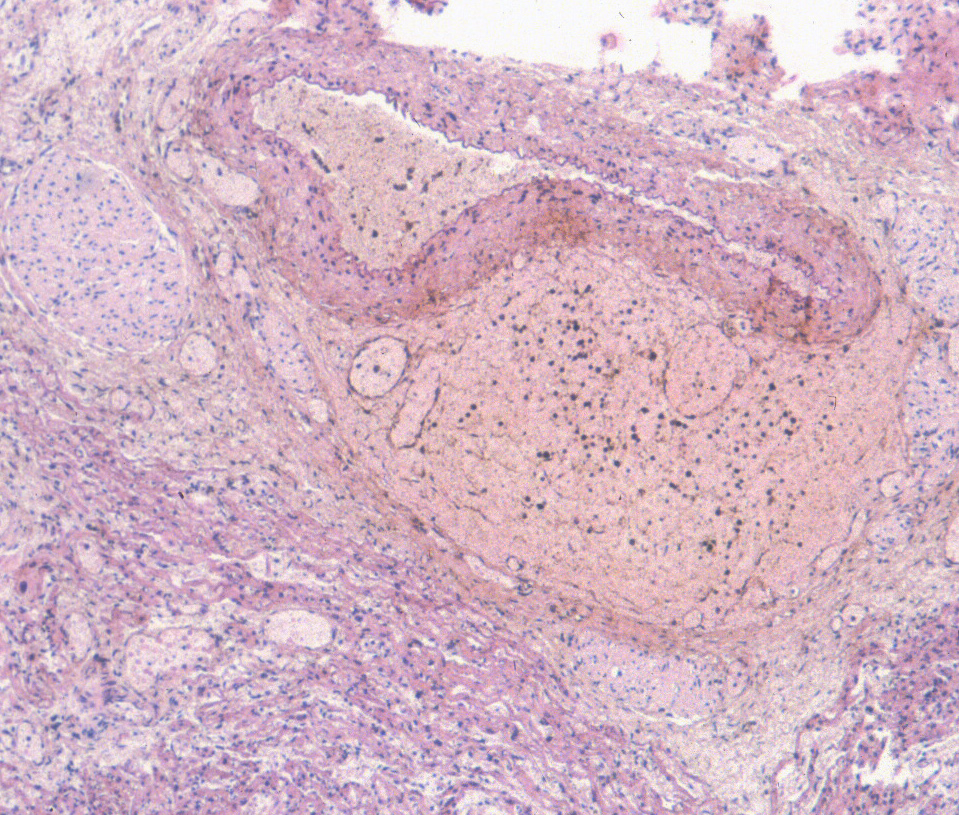
The significance of these petechiae is presented in the section on thymus. The parietal pericardium/epicardium may demonstrate acute inflammation from infection, although this inflammation is often difficult to distinguish from ectopic myelopoiesis (Fig 22a, b). The pericardium is a characteristic site of infection in horses with the blood borne bacterial infection from colonic ulcers (due to swallowed tent caterpillars) in Mare Reproductive Loss Syndrome8.
Endocardial surface:
Any notable subendocardial fibrosis is abnormal. This can be related to abnormal flow and pressure patterns in malformations, and also occurs in the recipient heart in twin-to-twin transfusion syndrome, presumably due to hypertension.
Heart valves:
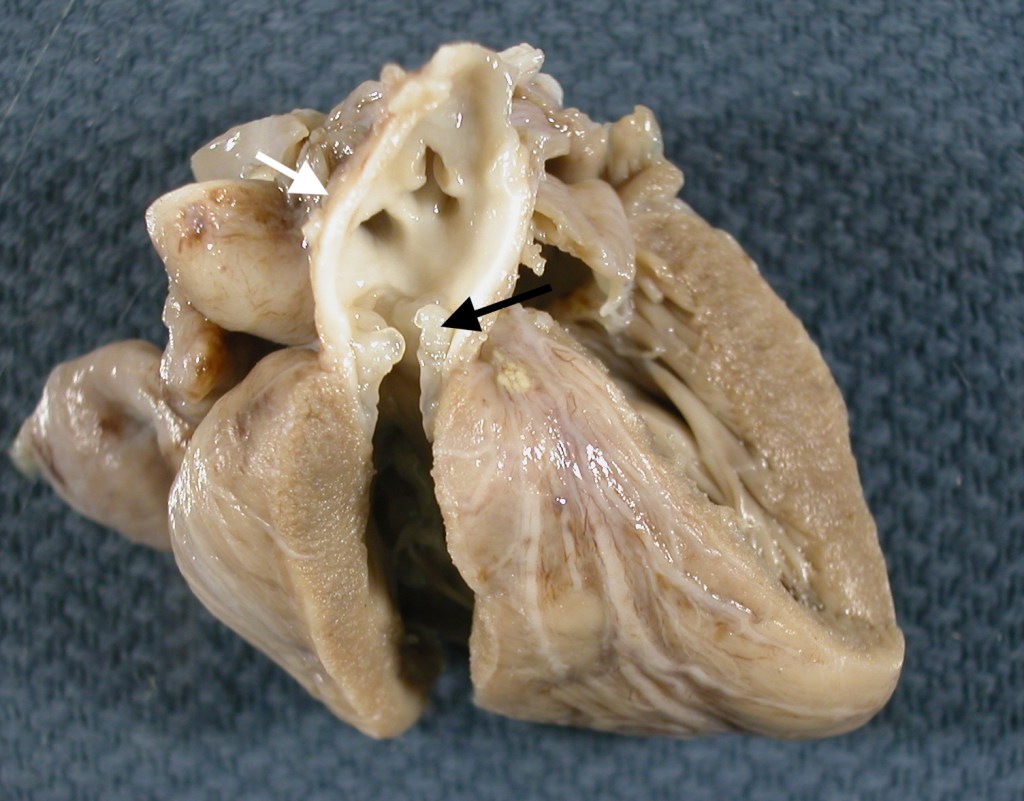
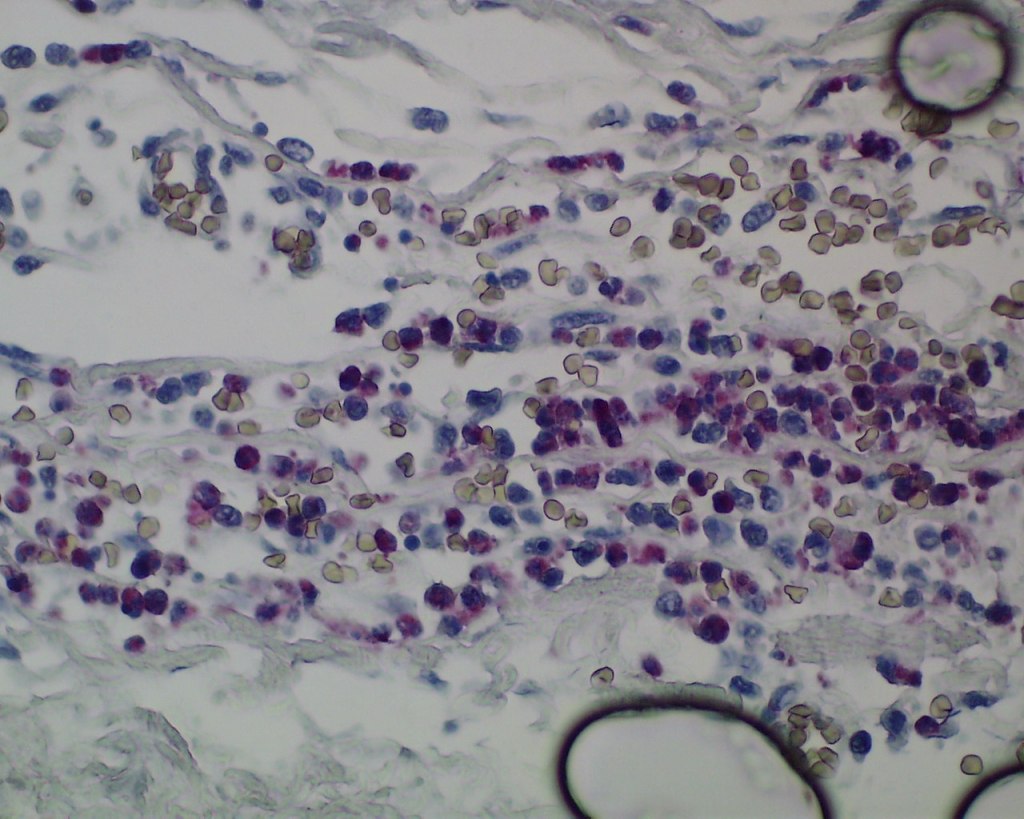
Vegetations on the tricuspid valve from marantic (non-bacterial) endocarditis occur rarely in infants of a diabetic mother (Fig 23). Another valve abnormality is formation of loose connective tissue forming globules in inherited polyvalvular dysplasia9 and in one form of a Noonan related syndrome10 (Fig 24).
Subendocardial and papillary muscle:
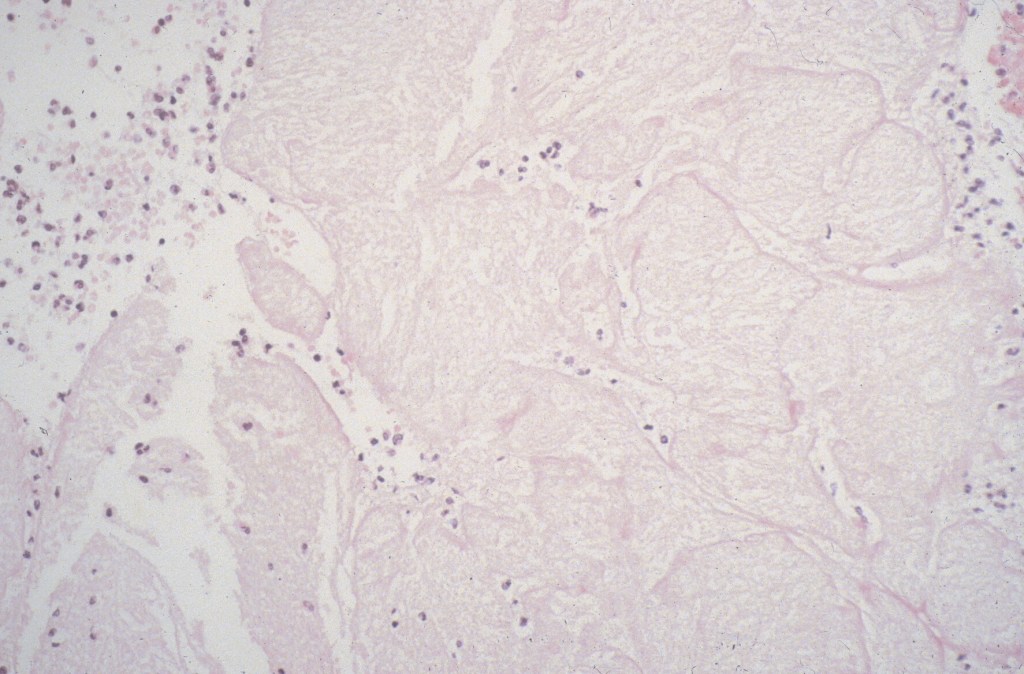
The papillary muscle and the subendocardial region have the most distant blood supply starting from the entrance of surface coronary arteries making these areas vulnerable to ischemia. Infants dying in cardiovascular shock from untreated hypoplastic left heart show infarctions in subendocardial tissue and papillary muscles11 (Fig 25a, b). The implication is not only that these areas became infarcted from lack of blood flow, but that the heart had continued to pump, and the infant survived long enough for the lesions to become histologically discernable , which in the classic Mallory White timing of adult myocardial infarctions is 3-5 hours12.


These infarctions can calcify and have been observed radiologically and histologically in neonates13. These subendocardial/papillary muscle infarctions can be seen in stillborn infants. In some cases, they are acute and are evidence consistent with hours of severe shock. They may also be scarred and calcified, indicating likely fetal recovery from a previous episode of shock (Fig 26).
Myocardium:
The fetal heart can suffer from viral myocarditis, particularly from Coxsackie B and echoviruses. The lesions may cause heart failure/hydrops. These lesions may also calcify, and this calcification be visible even is completely autolyzed hearts (Fig 27).

At least in theory, the heart muscle in a distended failing heart should have a different microscopic appearance from that of a heart that succumbs rapidly to asphyxia. There does sometimes appear to be more capillary congestion in an acute failing heart compared to sudden asphyxia seen in comparing stillbirth from large versus smaller extents of premature placental separation (Fig 28a, b). However, this observation is subjective and has not been tested rigorously. There is a study on wavy cardiac fibers in stillborn infants, but this has not been duplicated. As a research technique, staining for atrial natriuretic protein, or brain natriuretic protein in the ventricles, might show unique patterns between the brief heart failure from acute asphyxia and more prolonged heart failure, but such a study has not been reported in stillborn infants14,15.


The myocardium may demonstrate myocardiocyte hypertrophy or storage disease. There is a potential pitfall in the latter in that young gestation hearts tend to have increased glycogen and mitochondria creating a histologically clear myocyte cytoplasm in routine microscopic sections.
References:
1. Jeanty P, Romero R, Hobbins JC. Fetal pericardial fluid: a normal finding of the second half of gestation. Am J Obstet Gynecol 1984;149:529-32.
2. Dizon-Townson DS, Dildy GA, Clark SL. A prospective evaluation of fetal pericardial fluid in 506 second-trimester low-risk pregnancies. Obstet Gynecol 1997;90:958-61.
3. J R, P J. Fetal cardiac function and ductus arteriosus during indomethacin and sulindac therapy for threatened preterm labor: A randomized study. Am J Obstet Gynecol 1995;173:20-5.
4. Moise KJ, Jr., Huhta JC, Sharif DS, et al. Indomethacin in the treatment of premature labor. Effects on the fetal ductus arteriosus. N Engl J Med 1988;319:327-31.
5. Kohler HG. Premature closure of the ductus arteriosus (P.C.D.A.): a possible cause of intrauterine circulatory failure. Early Hum Develop 1978;2:15-23.
6. Heymann MA, Rudolph AM. Effects of acetylsalicylic acid on the ductus arteriosus and circulation in fetal lambs in utero. Circ Res 1976;38:418-22.
7. Genest DR, Williams MA, Greene MF. Estimating the time of death in stillborn fetuses: I. Histologic evaluation of fetal organs; an autopsy study of 150 stillborns. Obstet Gynecol 1992;80:575-84.
8. Bendon R, Sebastian M. Chorioamnionitis in the human with some comparison to the horse in mare reproductive loss syndrome. In: Powell DG, Furry D, Hale G, editors. Proceedings of a workshop on the equine placenta; 2003; Maxwell H. Gluck Equine Research Center: University of Kentucky. p. 116-8.
9. Bendon RW, Siddiqi T, de Courten-Myers G, Dignan P. Recurrent developmental anomalies: 1. Syndrome of hydranencephaly with renal aplastic dysplasia; 2. Polyvalvular developmental heart defect. Am J Med Genet Suppl 1987;3:357-65.
10. Bendon R, Asamoah A. Perinatal autopsy findings in three cases of jugular lymphatic obstruction sequence and cardiac polyvalvular dysplasia. Pediatr Dev Pathol 2007:1.
11. Coen R, McAdams AJ. Visceral manifestation of shock in congenital heart disease. Am J Dis Child 1970;119:383-9.
12. Mallory G, White P, Salcedo-Salgar J. The speed of healing of myocardial infarction A study of pathological anatomy in seventy-two cases. Am Heart J 1939;18:647-71.
13. Setzer E, Ermocilla R, Tonkin I, John E, Sansa M, Cassady G. Papillary muscle necrosis in a neonatal autopsy population: Incidence and associated clinical manifestations. J Pediatr 1980;96:289-94.
14. Das BB, Raj S, Solinger R. Natriuretic peptides in cardiovascular diseases of fetus, infants and children. Cardiovasc Hematol Agents Med Chem 2009;7:43-51.
15. Doyama K, Fukumoto M, Takemura G, et al. Expression and distribution of brain natriuretic peptide in human right atria. J Am Coll Cardiol 1998;32:1832-8.