I. Thymus
A. Gross findings
1. Small size:
The size of the thymus needs to be compared to that expected for gestation. The ratio of thymic to brain weight is .027 average based on the Perinatal Collaborative Study[1]. My median value from 84 cases is .014 (range .001 to .053) Earlier in gestation the thymus appears relatively small overlying the heart compared to later gestation. (Figs 1,2) Comparing photographs and weights based on gestation permits a reasonable judgement as to relative size. The size of the thymus in living neonates can be measured by the ratio of the thymic shadow to the chest size[2]. The most common cause of a small thymus is involution. (Fig 3)The lobules will appear smaller and the thymus less compact. A small thymus may also be part of a neural crest anomaly such as DiGeorge syndrome may be compact and of abnormal shape.



.
2. Abnormal location:
The normal thymus is bilaterally symmetrical with a narrow cervical portion and a larger, pyramidal thoracic portion. It lies anterior to the innominate vein and pericardium. Abnormal locations can be asymmetrical between lobes. The thymus may be more cervical, posterior to the great veins or even in the submandibular gland[3] These ectopic locations can be associated with anencephaly and other neural crest abnormalities, especially cono-truncal abnormalities of the heart. (Fig 4)

3. Thymic hemorrhages:
Thymic petechiae are a commonly encountered lesion in stillbirth. They appear as small pinpoint dots of red, or as larger confluent pattern of small hemorrhages. If due to gasping, they should not be in the cervical portions of the gland. (Fig 5, 6) Larger hemorrhages occur only rarely. One such case was associated with massive thymic and intracerebral hemorrhage [4]. The authors did not comment on any possible platelet PL1 antigen sensitization.

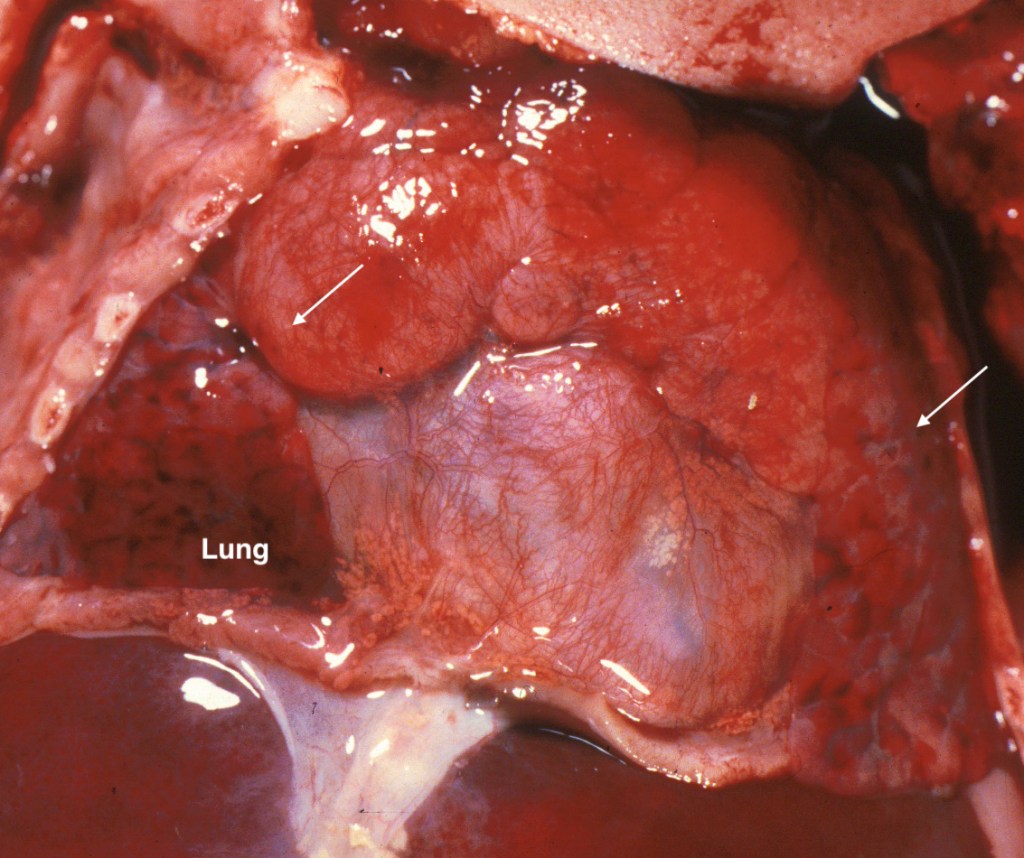
B. Histologic sampling:
In early gestation the entire thymus or one half of a bilaterally symmetrical thymus can be fit into a cassette. In older gestation, a longitudinal section can be cut and oriented on edge.
C. Microscopic findings:
1. Petechial hemorrhages:
Petechial hemorrhages are usually scattered within the parenchyma, not just on the surface. (Fig 7,8)
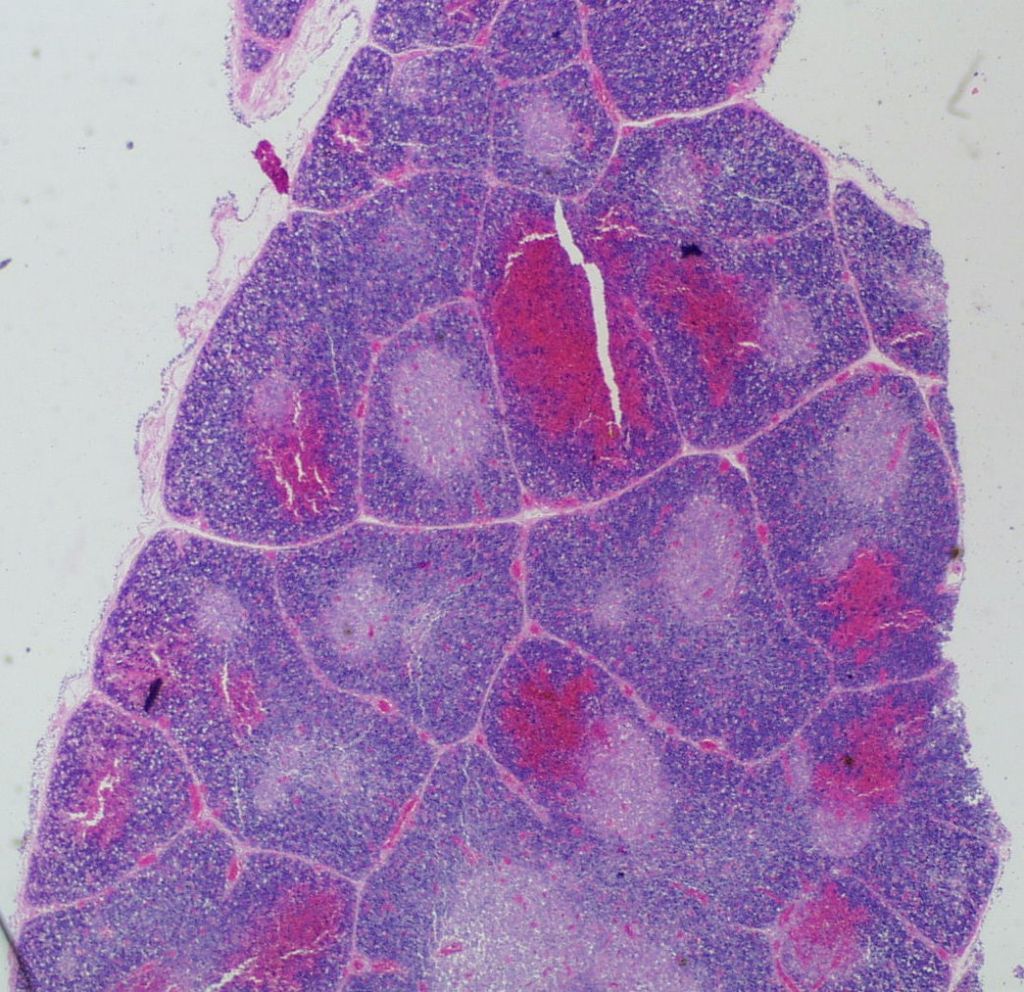

They may be difficult to perceive in autolyzed tissue. Thymic petechiae have been associated with acute asphyxia in stillborn infants, such as with premature separation of the placenta[5, 6]. In this scenario, the petechiae are usually sparse, superficial, and as is also the case with thymic petechiae in SIDS, not on the cervical thymus. Larger, confluent hemorrhages in the thymus, often suggest a more systemic coagulation disorder. Thymic petechiae associated with asphyxia are often accompanied with other intrathoracic petechiae including on the visceral pleura and the epicardium. To interpret the meaning of these petechiae requires understanding the mechanism of petechial hemorrhage formation and then to relate that to the events that occur with acute intrauterine asphyxia.
In general, the mechanism of the formation of capillary hemorrhage (petechiae) is multi-factorial [7]. Hemorrhage from capillary rupture will occur when the pressure difference across the wall of the capillary produces a stress in the wall greater than its tensile strength. The increased pressure difference could be caused by a surge in arterial pressure, as in the experimental administration of norepinephrine [8]. Elevations of venous pressure, as occur with the tourniquet test for scurvy, or with the Valsalva maneuver during pushing in labor, also produce petechiae. The chance of hemorrhage is favored by weak capillaries (scurvy), local anoxia, and clotting abnormalities (DIC, thrombocytopenia). The pressure difference depends not only on blood pressure, but also on the external pressure around the capillary. In deep inspiration, the high negative intrathoracic pressure favors the rupture of surface capillaries in the chest. The negative pressure is maximized if inspiration starts with obstruction of the glottis and the lungs at end expiration[9]. In taking the first breath, the fetal lungs are at the equivalent of end expiration, and initially there is a glottic stop [10, 11] Thus, the relationship between superficial intrathoracic petechia, including thymic petechia, and acute asphyxia is most likely the result of a fetal attempt to take the first breath. The role of negative intrathoracic negative pressure explains the absence of the petechiae in the cervical thymus.
Fetal gasping with acute asphyxia is analogous to the first breath, since the first breath typically is initiated by the unavoidable asphyxia of cessation of the umbilical circulation at birth. The onset of in utero gasping to acute asphyxia was identified experimentally very early in the history of experimental fetal physiology[12]. This deep inspiration with a glottic stop is different from the shallow intrauterine respirations that typically involve shallow, glottic-open respirations. Absent intrathoracic petechiae imply that the fetus was apneic at the time of death, in effect anesthetized, usually by the chronic development of hypoxia/ acidosis. If petechiae are present in other parts of the body, then causes such as DIC, sepsis, shock, thrombocytopenia, no just gasping may be present. In infants born alive, there are few “control” cases to test whether normal initiation of respiration results in petechiae.
Animal studies demonstrate the multi-factorial nature of the formation of intrathoracic petechiae. For example, one study of adult rats sacrificed by tracheal occlusion at end expiration, found that norepinephrine increased the number and alpha-adrenergic blockade decreased the number of petechia [8]. These drugs were given 5 minutes before death, and they increased and decreased blood pressure respectively. A study of tracheostomized rabbits demonstrated collapse of the pharyngeal airway (which most likely closed at end expiration) resulted in pulmonary, but not abdominal, petechiae [13]. A very complex study in rabbits used different modes of death and evaluated pulmonary petechiae [14]. They found that a single fatal episode of apnea from pentobarbitone injection did not produce petechia. Tracheal occlusion at end expiration did produce petechiae and also was associated with multiple episodes of gasping and wide shifts in intrathoracic pressure. Hypotensive deaths from cardiac electrocution and hypertensive deaths from norepinephrine, even without gasping, also produced pleural and abdominal petechiae. The highest numbers of petechiae were in the hypertensive animals, both those with obstructive airways and those with norepinephrine. In a study of end expiratory airway obstruction in adult rabbits, the elevation of pulmonary wedge pressure compared to endotracheal pressure correlated directly with petechiae [15]. In utero, with a patent ductus arteriosus and low pulmonary blood flows, terminal left heart failure may not necessarily lead to elevated capillary wedge pressure, making studies in adult animals not comparable to that in fetuses.
Bottom line: The bottom line is that thymic petechia if alone or with pleural and epicardial petechiae are probable evidence of intrauterine gasping due to asphyxia. A lack of thymic petechiae suggests a fetus too obtunded to respond or not stimulated to gasp in response to the conditions of death. Finding petechiae also in non-thoracic locations makes the diagnosis of gasping equivocal.
2. Thymic Involution
The thymus normally involutes as the infant ages. However, in some stillborn infants, usually with a chronic stress such as infection, fetal hydrops or growth restriction, there is accelerated involution, that is loss of the lymphocytes in the cortex of the thymus. An early phase of this process is often seen in response to chorioamnionitis. In the acute cases, the prominent abnormality is a “starry sky” appearance due to the lymphorrhagia (phagocytosis) of cortical lymphocytes. Low magnification microscopy demonstrates open spaces in the cortex reminding some observers of a “starry sky”. (Fig 9,10) At higher magnification, these apparent spaces can be seen to be the cytoplasm of a large cell often with small, fading nuclei of small lymphocytes within.

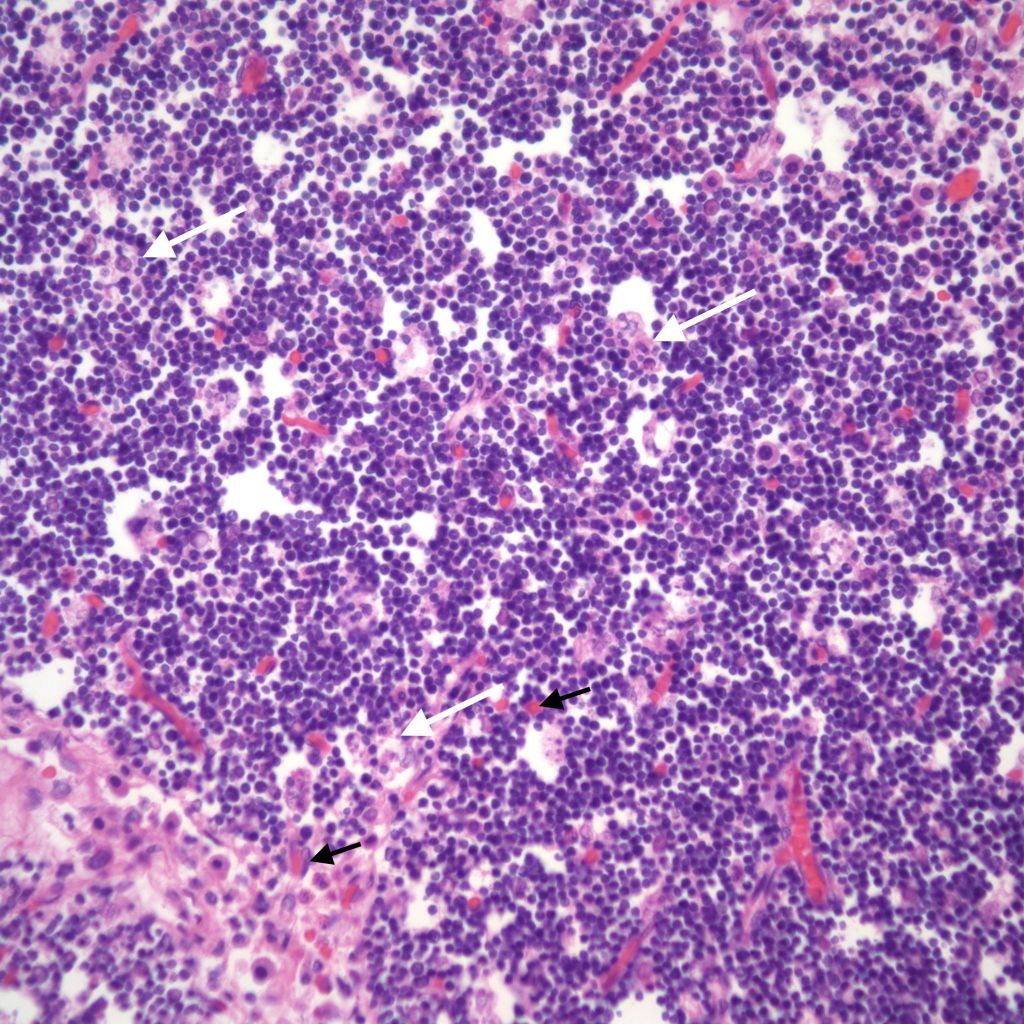
As involution progresses, there will be a progressive loss of cortical lymphocytes. The cortex will appear notably thinned compared to the medulla with thinner lobules and more connective tissue between lobules (Figs 11a, 11b, 12a, 12b, 13a, 13b).

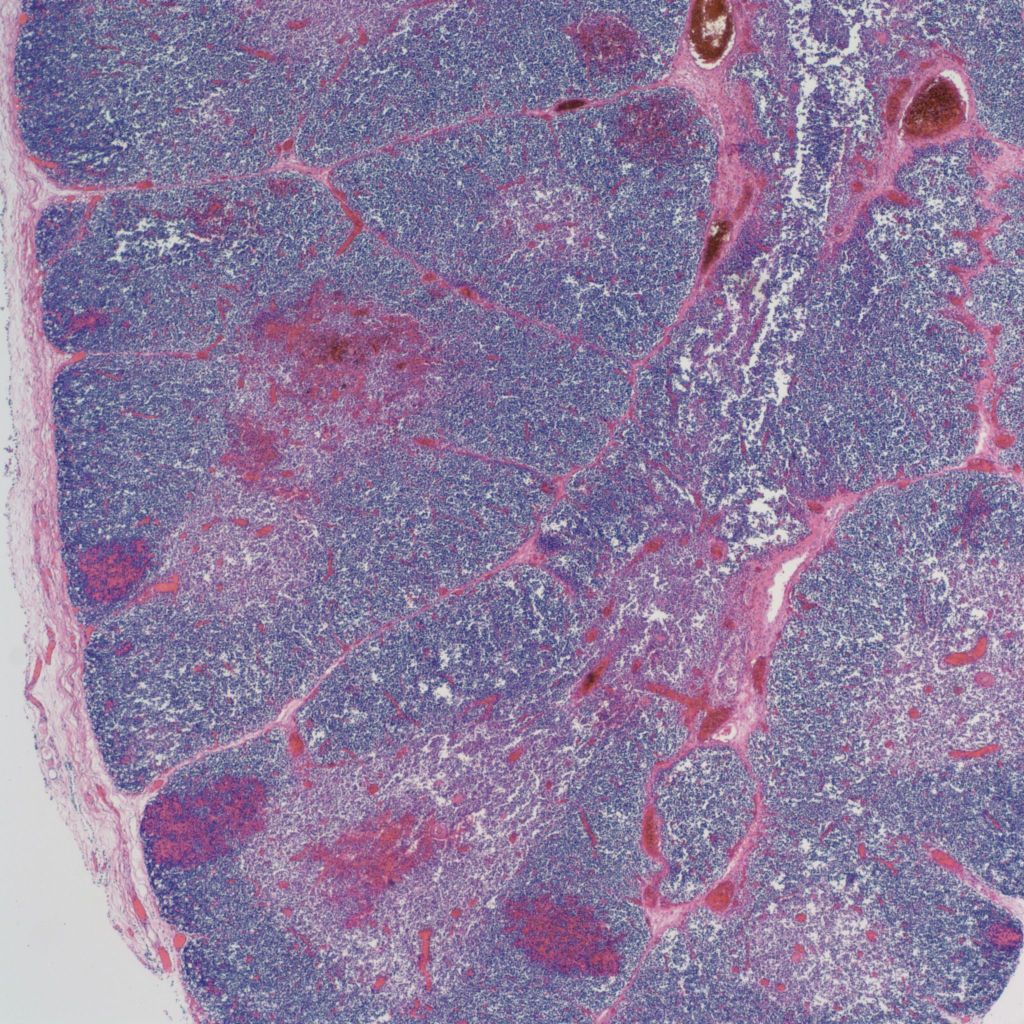
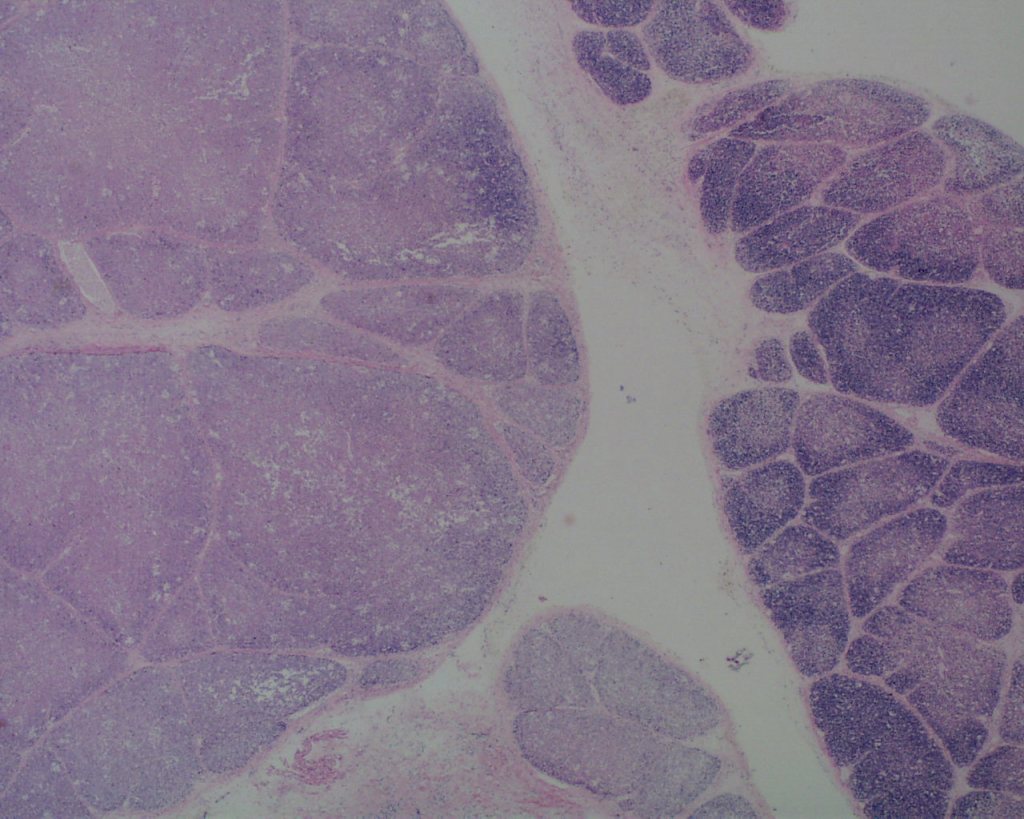

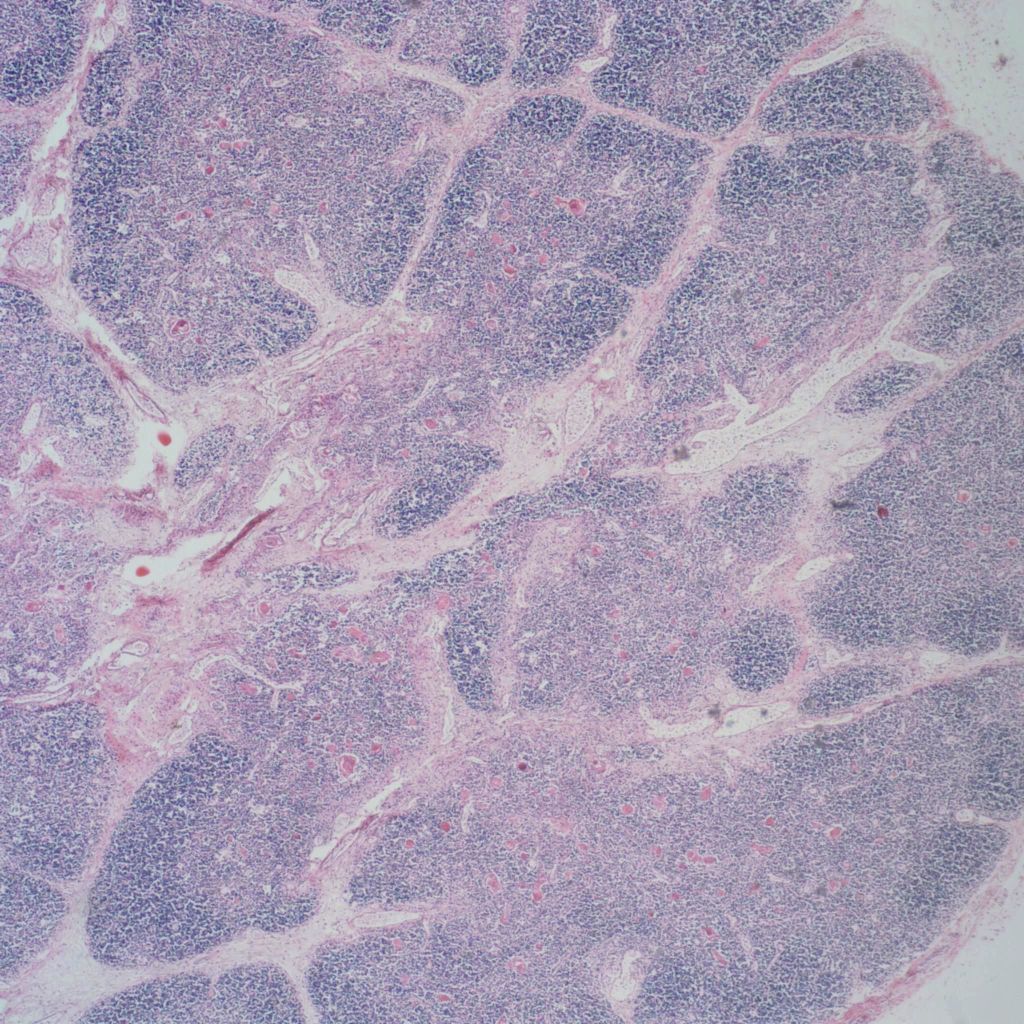

At the extreme, the medulla will appear to have the only small lymphocytes in the thymus, giving an inside out appearance at low magnification (fig 14, 15).


A staging system has been proposed for this progression: stage 0 = no histopathologic changes, stage 2 = starry sky appearance, stage 3 = loss of distinction of the medulla and cortex, and stage 4 = loss of all cortical lymphocytes with an inverted appearance[16]. Based on the clinical histories, the stage has been correlated with duration of involution as follows: stage 0 = 0-12 hours, stage 1 = 12-24 hours, stage 2 24-48 hours, and stage 4 > 72 hours. A continuous stress keeps the thymus involuted, and results in decreased weight as demonstrated in human autopsy and in experimentally in the monkey with repetitive dexamethasone injections [17].The thymic to brain weight usually reflects the histological degree of involution when the small size is due to histological identified involution and not a failure of thymic development.
Accelerated thymic involution can be produced experimentally by corticosteroids (or ACTH), thyroid hormone and sex hormones [17, 18]. In the perinatal period, a stress response from ACTH is the most likely natural cause. Experimentally the lesion can be produced by lipopolysaccharides or foreign blood cells [19, 20]. Other possible external causes are radiation, exogenous steroids, or chemotoxic agents.
The complexity of thymic changes is demonstrated in a case of a 22 week gestation infant with a week of preterm premature rupture of membranes who was treated with betamethasone (a synthetic corticosteroid that can cross the placenta to the infant and used to promote lung maturation) and antibiotics. The infant survived for three quarters of an hour. The placenta demonstrated choroamnionitis with some fetal inflammation in the superficial chorionic vessels. The autopsy demonstrated complete involution of the thymus, follicular formation in the spleen, and extensive, intense acute pneumonitis with no growth of microorganisms on lung culture. The thymus demonstrated an inverted picture with no cortex and small lymphocytes in the medulla. The cortical rim demonstrated a compact epithelial layer. (Figs 16, 17) This infant had evidence of acute inflammation, and antibody stimulation, making it impossible to assess the changes due to the exogenous steroid.
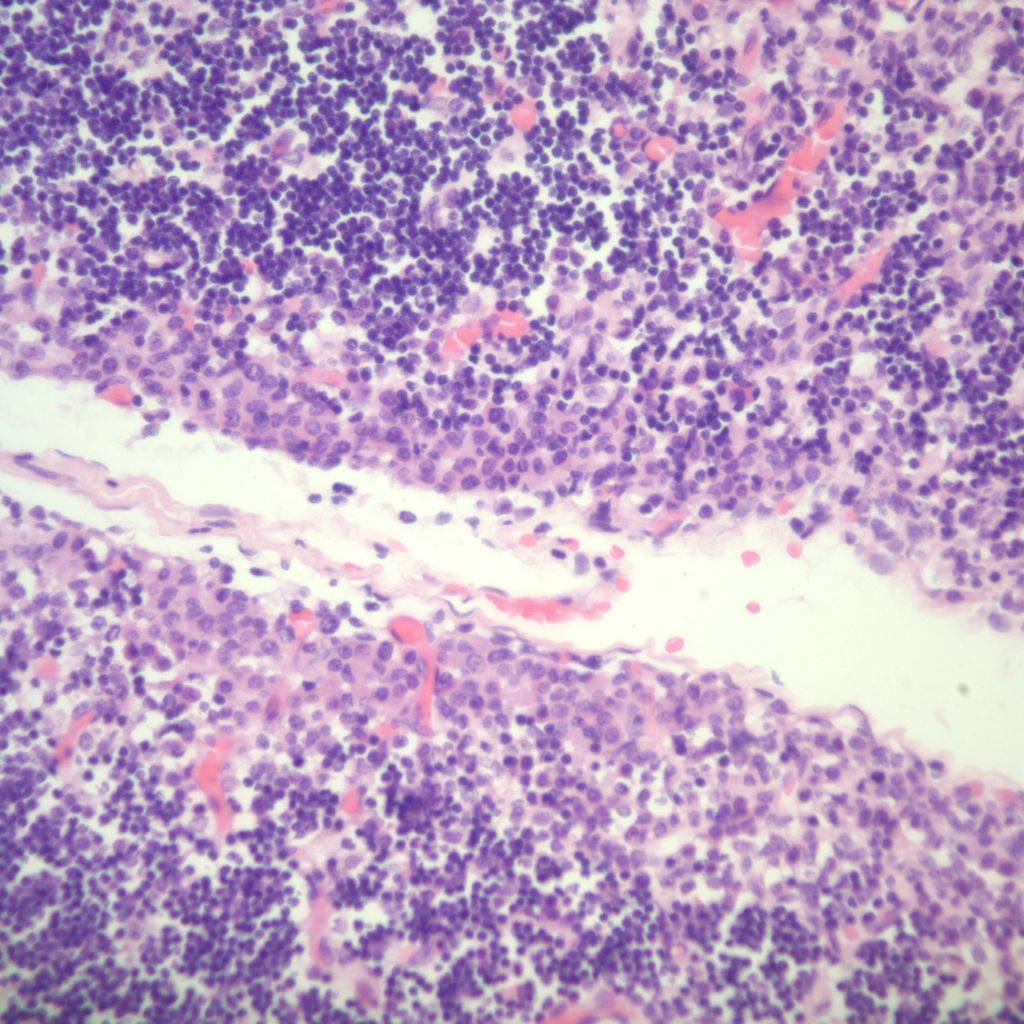
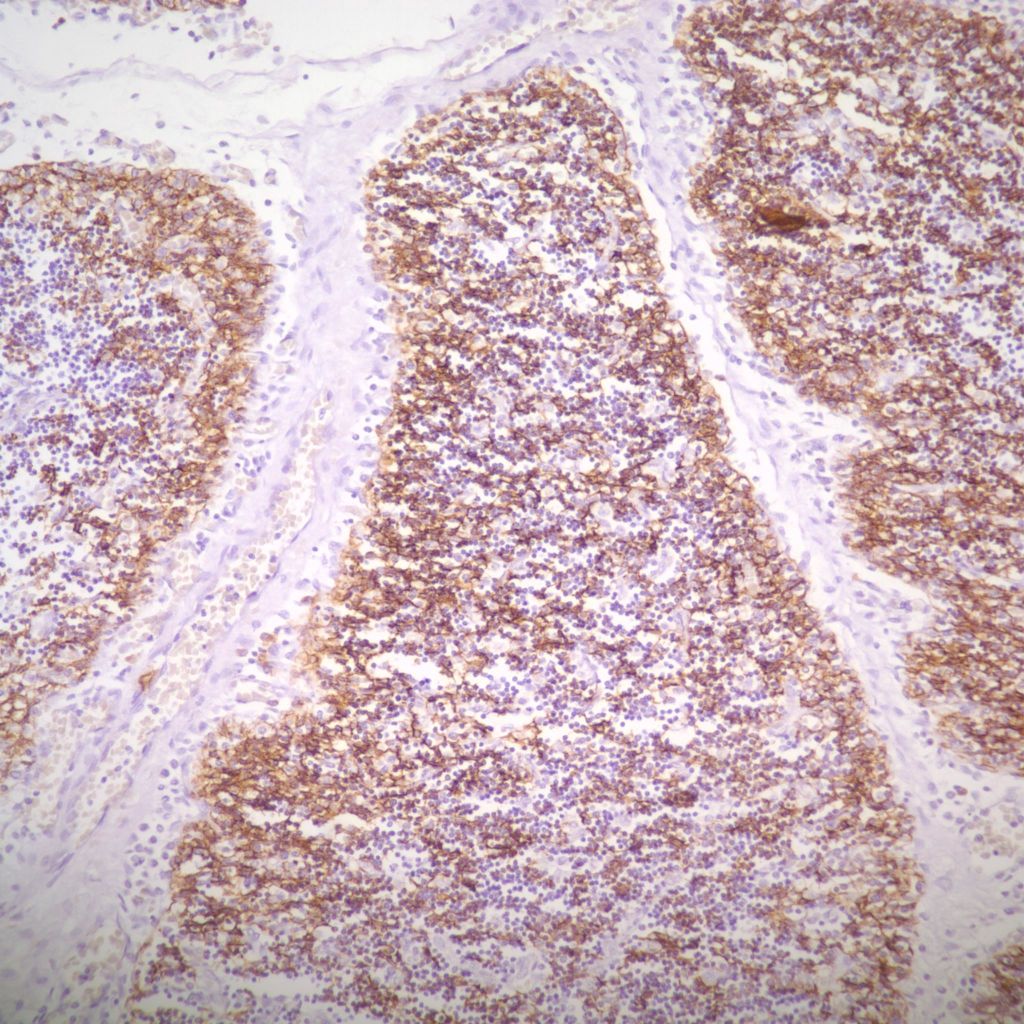
The fetal thymus is the site in which immature thymic lymphocytes enter the outer cortex, mature into double positive CD4+ and 8+ cells, and then as single positive lymphocytes in the medulla are released into the blood as single effector or helper phenotypes. This process is guided by thymic epithelial cells and the chemokines. Immature lymphocytes can be negatively selected, for example, unable to bind to MHC antigens, and die. Presumably these cells are phagocytosed by macrophages and dendritic cells in the cortex and medulla. In the medulla, specialized cells present self-antigens. Cells that bind strongly to these antigens undergo apoptosis. There is normally a high proliferation and turnover of lymphocytes. The appearance of loss of cortical lymphocytes in accelerated involution could be some combination of decreased influx of immature cells, more rapid negative selection (as suggested by phagocytosis of lymphocytes in the cortex and medulla), or even more rapid processing and efflux of mature cells. The consequences of continuous stress over acute stress on the future T-lymphocyte population is also unclear. An analysis of thymuses removed at less than one month of age at cardiac surgery from infants without exogenous cortisol, found that by one day of age there was a marked reduction in the level of double positive T cells. This suggests that some of the changes accelerated by corticosteroids may be the same as normal changes following delivery [21]. Interestingly, the thymus is commonly removed at the time of cardiac surgery, without apparent ill consequences. Similarly, adults usually have complete involution of the thymus without consequence.
The role of the thymus in preventing autoimmunity by eliminating clones that recognize self-antigens is complex. Immature thymic lymphocytes in order to survive must recognize MHC, class I and II antigen and have low affinity for some self-peptides. Some with medium affinity may become regulatory T cells. The consequences of accelerated, as opposed to normal, involution on elimination of harmful self-antigen clones is unclear. The experimental models of accelerated thymic involution have been short term. The long-term effect is unknown.
In previable births, it may be possible to obtain the thymus immediately after death for further study of its immune function.
3. Eosinophils in the thymus
..
The thymus of children invariably have some eosinophils (Fig 10 above). This has been known for over a century. They may be mature bi-lobed or immature mononuclear form. Some cell division and maturation may occur in the thymus, but experiments suggest that eosinophils can migrate rapidly into the thymus. The function of these eosinophils is unknown. Studies in chick embryos in 1979 demonstrated an increase in the oxidase positive cells (monocytes and eosinophils) in the thymus and spleen of chick embryos with injected substances including sheep red blood cells and lipopolysaccharide. [19, 20].
Throsby discovered eosinophils in the thymus while looking for dendritic cells. The eosinophils like dendritic cells were CD11c +, but were also CD11b+ [22]. They argued that eosinophils are antigen presenting cells, involved at the clonal selection in the thymus, based on their presence at the same time double CD 4+/CD8+ cells are being eliminated. The eosinophils in the thymus have a specific pattern of cytokine activation with IL-4. IL –13, a pattern also found in parasite and nasal antigen responses associated with eosinophil extravasation and survival. They note that eosinophils secrete free radicals that can diffuse through near-by cell membranes inducing apoptosis. They also discuss studies that eotaxin, an eosinophil-specific CCR3 ligand, is constitutively
expressed in thymus. These studies suggest that eosinophils are constituents of the thymus and may be involved in destruction of self-antigenic T-cells.
Eosinophils may be increased with accelerated involution, although this is not well enough established to use as a diagnostic criterion. Such an increase could be part of the process of more rapid T-cell elimination.
4. Hassall corpuscles
In the thymus at younger gestation, Hassall corpuscles may not be evident although small clusters of epithelial cells may be found (Fig 18).

Hassall corpuscles become prominent by 28 weeks of gestation[23]. In trisomy 21, they may be quite large and cystic (Fig 19).
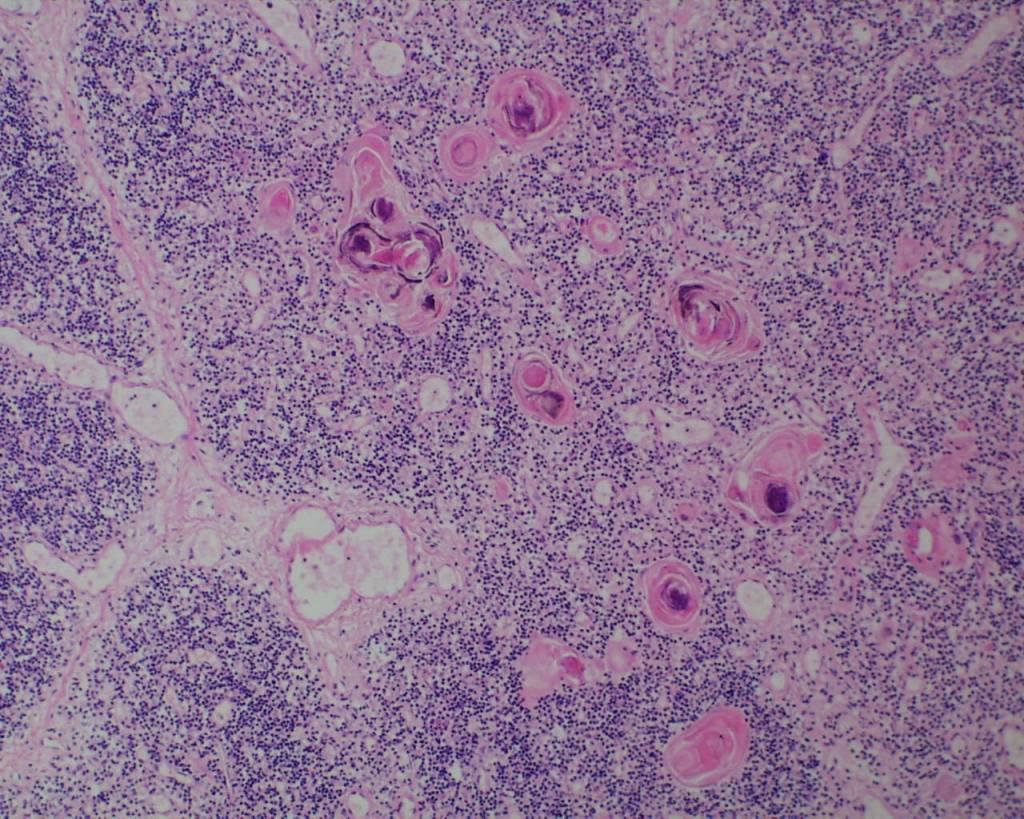
A complete lack of Hassall corpuscles is evidence of an inherited immune deficiency. The thymus most likely will be small. The lack of thymic Hassall corpuscles is associated with immune deficiency in postnatal life. Such a finding prenatally may be associated with a molecular diagnosis of a specific immune deficiency. Thymic dysplasia is diagnosed only if the medullary epithelial component is absent. Hypoplastic thymuses that are not involuted nor dysplastic may be part of a wider malformation syndrome such as Di George (deletion of 22q1.2) that have Hassall corpuscles may still be associated with a T-cell deficiency (Fig 20,21).
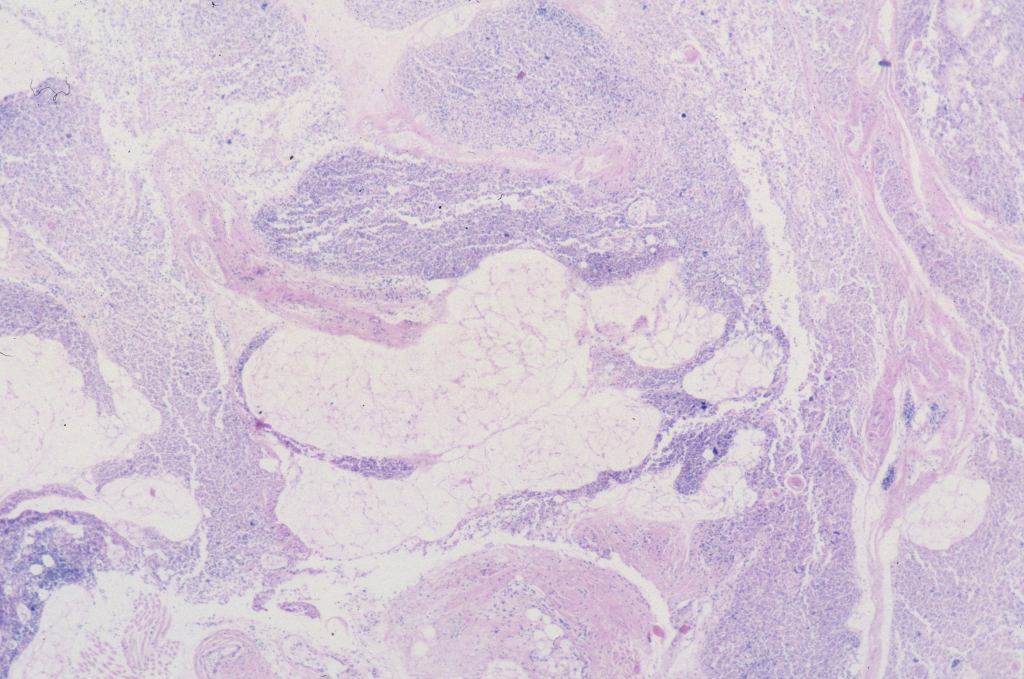

Thymic development has been tied clinically and experimentally to the neural crest. Deletion experiments in the chicken demonstrated that removing certain small areas of the neural crest stopped thymic as well as cardiac development[24]. These experiments mimic DiGeorge/22q1.2 deletion syndrome.
One hypothesis of thymic development is that there are two distinct populations of thymic epithelium (Sheir). The first is the medullary tube which develops into Hassall corpuscles. This tissue is capable of forming a cortex in explants. Hassall corpuscles may have a role in inducing dendritic cells to produce CD4+ T-regulatory lymphocytes. The other thymic epithelial cells are the reticular cells that surround perivascular spaces. The loss of thymic function with multiple mutations associated with T-cell deficiencies is a topic to itself. An interesting knock out mouse demonstrated that loss of the estrogen alpha receptor reduces thymic medulla even though levels of testosterone, estradiol, and corticosteroids are similar to controls[25]. This evidence, along with the production of involution with sex steroids, suggests a thymic- sex hormone relationship.
5. Database:
Gross thymus:
Weight:
Relative size:
Maldescent/ malformation:
cervical only
not symmetrical
single lobed
irregular
other
Hemorrhages:
scattered petechial hemorrhages
thoracic portion
cervical portion
confluent petechial hemorrhages
large confluent hemorrhage
Mass lesions
none
cyst
mass replacing thymus
mass in the thymus
Microscopic:
Stage 0: Normal: Small lymphocytes in the cortex with a clear cortex: medullary junction.
Stage 1: Starry sky appearance:
Stage 2: Partial depletion of cortical thymocytes with blurring of medullary cortical junction
Stage 3; Inversion of the cortical medullary appearance
Eosinophils:
Scattered
Numerous
Petechiae in the thoracic parenchyma:
Medullary abnormalities:
Absent Hassall corpuscles
Cystic Hassall corpuscles
Other:
6. References:
1. Fujikura, T. and L. Froelich, Organ-weight/brain -weight ratios as a parameter of prenatal growth: A balanced growth theory of visceras. Am J Obstet Gynecol, 1972. 112: p. 896-902.
2. Chen, C.M., et al., Thymus size and its relationship to perinatal events. Acta Paediatr, 2000. 89(8): p. 975-8.
3. Bale, P.M. and C. Sotelo-Avila, Maldescent of the thymus: 34 necropsy and 10 surgical cases, including 7 thymuses medial to the mandible. Pediatr Pathol, 1993. 13(2): p. 181-90.
4. Saksenberg, V., B. Bauch, and S. Reznik, Massive acute thymic haemorrhage and cerebral haemorrhage in an intrauterine fetal death. J Clin Pathol, 2001. 54(10): p. 796-7.
5. Bendon, R.W., Review of autopsies of stillborn infants with retroplacental hematoma or hemorrhage. Pediatr Dev Pathol, 2011. 14(1): p. 10-5.
6. Jacques, S.M. and F. Qureshi, Adrenal Histologic Stress-related Changes in Third Trimester Stillbirth. Pediatr Dev Pathol, 2017. 20(2): p. 112-119.
7. Jaffe, F.A., Petechial hemorrhages A review of pathogenesis. Am J Foren Med Pathol, 1994. 15: p. 203-207.
8. Krous, H.F., A.C. Catron, and J.P. Farber, Norepinephrine-induced pulmonary petechiae in the rat: An experimental model with potential implications for sudden infant death syndrome. Pediatr Pathol, 1984. 2: p. 115-122.
9. Beckwith, J.B., Intrathoracic petechial hemorrhages: a clue to the mechanism of death in sudden infant death syndrome. Biol Psych, 1990. 28: p. 37-47.
10. Milner, A.D. and H. Vyas, Lung expansion at birth. J Pediatr, 1982. 101: p. 879-886.
11. Bosma, J.F., L. J, and G. N, Motions of the pharynx associated with the initial aeration ofthe lungs of the newborn infant. Acta Paediatr, 1959. 48, Suppl 117: p. 117-122.
12. Dawes, G., Foetal and Neonatal Physiology. 1968, Chicago, IL: Year Book Medical Publishers, Inc. 247.
13. Abu-Osba, Y.K., O.P. Mathew, and B.T. Thach, An animal model for airway sensory deprivation producing obstructive apnea with postmortem findings of sudden infant death syndrome. Pediatr, 1981. 68: p. 796-801.
14. Campbell, C.J. and D.J.C. Read, Circulatory and respiratory factors in the experimental production of lung petechiae and their possible significance in the sudden infant death syndrome. Pathol, 1980. 12: p. 181-8.
15. Farber, J.P., A.C. Catron, and H.F. Krous, Pumonary petechiae: ventilatory-circulatory interactions. Pediatr Res, 1983. 17: p. 230-233.
16. Toti, P., et al., Acute thymic involution in fetuses and neonates with chorioamnionitis. Hum Pathol, 2000. 31(9): p. 1121-8.
17. Novy, M.J. and S.W. Walsh, Dexamethasone and estradiol treatment in pregnant rhesus macaques: effects on gestational length, maternal plasma hormones, and fetal growth. Am J Obstet Gynecol, 1983. 145(8): p. 920-31.
18. Weaver, J.A., Changes induced in the thymus and lymph nodes of the rat by the administration of cortisone and sex hormones and by other procedures. J Pathol Bacteriol, 1955. 69(1-2): p. 133-9.
19. Moriya, O. and Y. Ichikawa, Accumulation of eosinophils and monocytes in lymphoid organs of chick-embryos. II. Effect of mitogenic stimulation. Microbiol Immunol, 1979. 23(6): p. 523-31.
20. Moriya, O. and Y. Ichikawa, Accumulation of eosinophils and monocytes in lymphoid organs of chick-embryos. I. Effect of antigenic stimulation. Microbiol Immunol, 1979. 23(6): p. 515-22.
21. Varas, A., et al., Analysis of the human neonatal thymus: evidence for a transient thymic involution. J Immunol, 2000. 164(12): p. 6260-7.
22. Throsby, M., et al., CD11c+ eosinophils in the murine thymus: developmental regulation and recruitment upon MHC class I-restricted thymocyte deletion. J Immunol, 2000. 165(4): p. 1965-75.
23. Asghar, A., Y.M. Syed, and F.A. Nafis, Polymorphism of Hassall’s corpuscles in thymus of human fetuses. Int J Appl Basic Med Res, 2012. 2(1): p. 7-10.
24. E, B.D. and K.M. L, Embryonic origins of defective heart development. Annals of the New York Academy of Sciences Vol 588, 1990. New york: New York Academy of Sciences.
25. Yellayi, S., et al., Normal development of thymus in male and female mice requires estrogen/estrogen receptor-alpha signaling pathway. Endocrine, 2000. 12(3): p. 207-13.