Umbilical cord hematoma
Beside direct compression there are other lesions that are accepted mechanisms of “cord accident”. One such mechanism is umbilical cord hematoma caused by hemorrhage into the umbilical cord. A hematoma inside the cord becomes a constrictive ring similar to the direct compression of the experimental ring occluder. It appears as a thick red swelling of the cord (Fig 2).

Figure 2: This shows a large hematoma that was clamped at near the umbilicus in a infant delivered with severe acute distress who died from the effects of intrauterine asphyxia at 10 hours of age. The entire vein in this cord showed some thinning of the media, but the hematoma was confined to the periumbilical segment.
Hematomas large enough to cause fetal asphyxia are infrequent, one in thousands of deliveries, but they have a high risk of causing fetal injury or death. Because the umbilical cord is a closed, tight space, the hematomas are usually tamponaded before reaching a size that would cause fetal exsanguination, unless the cord surface also ruptures. They also seldom appear to result from a torn normal umbilical vessel. Rather, the most common histologic lesion is a dilated thin walled vein often with a point of rupture, basically an aneurysmal rupture.
The published studies of umbilical cord hematoma are case reports or case series of very variable detail about the actual anatomy of the hematoma. When a description is included, the most frequent is that of a thinned, often aneurysmal vein[1-13]. The location is most commonly near the umbilicus[5, 7, 13-22]or near the cord insertion[1-3, 6, 9, 21, 23]. Yet, no regions are immune, and some hematomas are located toward the middle of the cord[4, 8, 9, 11, 12, 24]. Most are more than 3 cm in at least one dimension. The clinical histories include stillbirth, rescued fetal distress, and incidental findings in an uncomplicated pregnancy. A relatively recent report of three live born infants demonstrates the ambiguity in interpreting the anatomy of the hematoma after delivery. From their case 2: “ An umbilical cord hematoma that measured 4 x 2 cm was identified that was associated with the umbilical vein. The cord measured 52 cm in length and had three vessels. The arterial lumens were compressed but appeared patent.” As a pathologist I would want to know the orientation of the dimensions of the hematoma and the meaning of “associated” ? There was a normal length of the cord, but where was the hematoma? The arterial lumens should be compressed unless the infant is stillborn. The hematomas were not visualized prenatally. The focus of the report was on the details of the antenatal fetal heart rate tracings. The case reports reveal a significant knowledge gap about the formation and clinical hemodynamics of umbilical cord hematoma.
Beside the case reports, other insights into cord hematomas can be gleaned from hematomas produced accidentally by therapeutic interventions. In the course of giving the fetus a blood transfusion via the umbilical vein, blood may be accidentally injected into Wharton’s jelly. These relatively small amounts of blood induce bradycardia[25-27]. That Doppler ultrasound demonstrated decreased blood flow on the far side of a hematoma in one case, led the authors to hypothesize that the hematoma caused vasospasm[25].
Another unintended model of cord hematoma is laceration of a cord vessel following amniocentesis or cordocentesis to sample fetal blood (Fig 3).

Figure 3a: This cord from an infant undergoing percutaneous fetal blood sampling had persistent hemorrhage into the amniotic fluid seen by Doppler ultrasound. The surface of the cord is stained by blood in the amniotic fluid. The small red dot on surface proved to be the point of hemorrhage (arrow).
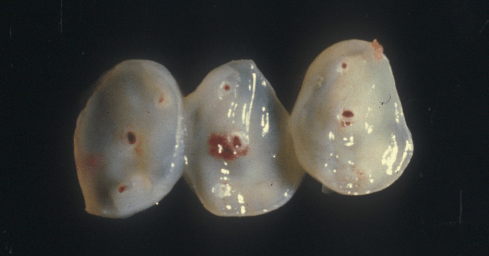
Figure 3b: The cross sections from the region of the red dot demonstrated a faint trail of hemorrhage from the artery to the surface.
There are case reports of such hematoma formation causing fetal death (2 were from fetuses with trisomy 18)[28]. Conversely, a 5×2 cm hematoma was found by ultrasound 24 hours after an amniocentesis but it was no longer detectable 10 days later[29]. A review of 50 cords from cordocentesis patients found evidence of brief bradycardia associated with bleeding. There was only one large cord hematoma that became apparent within 1 hour of the procedure. The infant died 6 days later but there were no reported details on the progression of the lesion over that time[30]. The variation in these iatrogenic reports suggests that in general a venous hemorrhage into the cord, or an injected hemorrhage results in transient fetal bradycardia, and with larger hematoma may lead to fetal death, but the precise relationships of size, timing and fetal death are still not clear.
The umbilical cord hematomas that I have examined have always demonstrated an abnormally widened and tortuous vein, a varix (Fig 4) within the hematoma.

Figure 4a: The umbilical cord demonstrates at least a doubling of diameter from a hematoma at the fetal end of the cord in this term gestation, stillborn infant.
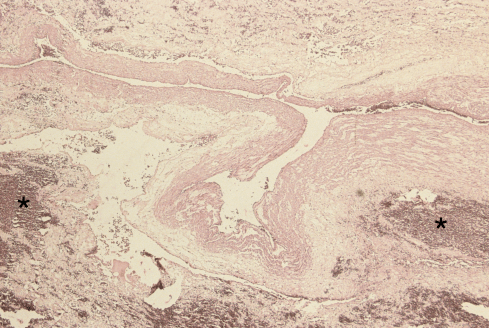
Figure 4b: From the same umbilical cord, the variceal umbilical vein in the center of the photomicrograph can be seen to be collapsed and there is surrounding hemorrhage in Wharton’s jelly (*). (H&E, 2x)
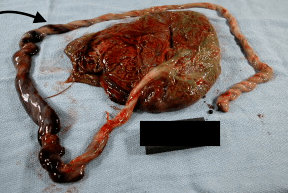
Figure 4c: This umbilical cord from a stillborn term infant demonstrates a mid-cord hematoma with a dilated vein extending from it toward the fetal end of the cord. (arrow)

Figure 4d: This photomicrograph of the cord from figure 2c demonstrates the hematoma surrounding the dilated umbilical vein. (H&E, 2x)
Just looking with the naked eye at the umbilical cord, a large hematoma and a large varix often appear similar. They can be distinguished on cross sectioning the cord and confirmed by microscopic evaluation. Even very large varices may not cause injury to the infant (Fig 5) [31-34].
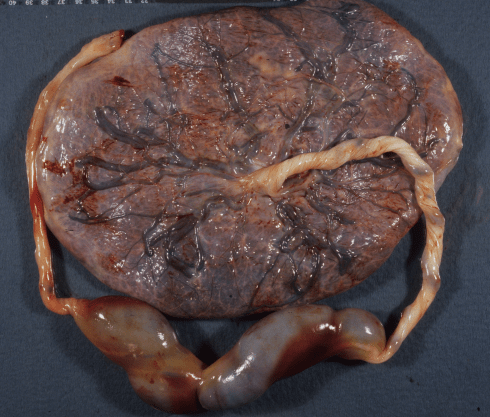
Figure 5a: This large midcord aneurysm remained intact with a small layer of laminated thrombus along part of the wall. The infant had variable fetal heart rate decelerations and bradycardia. The Apgar scores were 4 and 7 at 1 and 5 minutes.
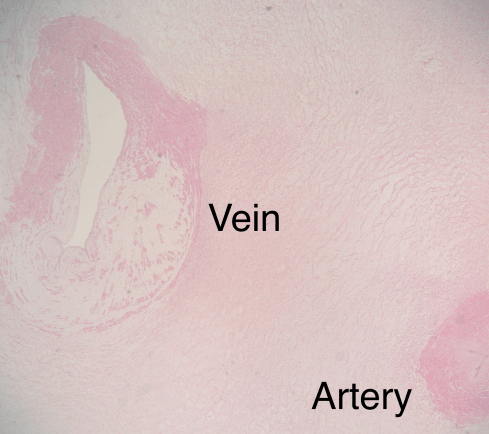
Figures 5b: This photomicrograph through the vein away from the aneurysm shows the loss of architecture of the vein wall replaced by loose connective tissue and scattered muscle fibers. The etiology of this change in the media is unknown.
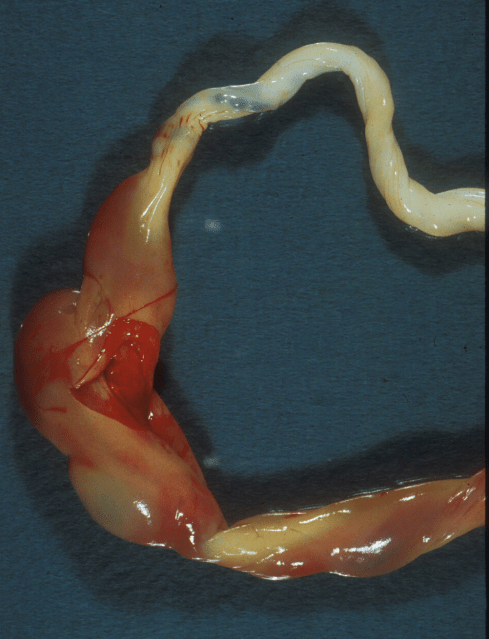
Figures 5c: This large umbilical vein varix was from an infant with a normal delivery and Apgar scores of 9 and 9 at 1 and 5 minutes. Some hemorrhage can be seen in Wharton’s jelly but not a compressing hematoma.

Figure 5d: The cross section shows the patency of the vein filled with blood.
The varices I have seen appear to involve only a part of the umbilical cord length. They usually have uneven thinning of the media circumferentially. An aneurysm is a very small area of weakened wall in a vessel that expands outward like a similar weak point in a tire. I do not see any value in most cases between distinguishing between varices and aneurysms in the umbilical vein. However, I do diagnosis as an aneurysm a focal, eccentric dilatation of the umbilical vein arising at the insertion into the placenta (Fig 6).
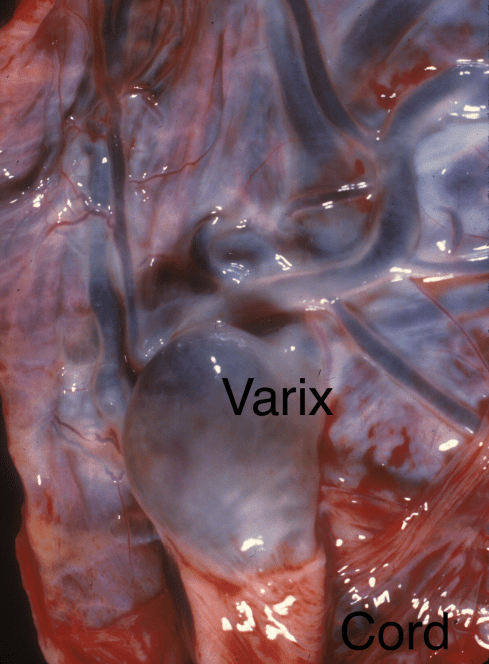
Figure 6 a: An incidentally present aneurysmal dilatation of the umbilical vein is present at the placental insertion.
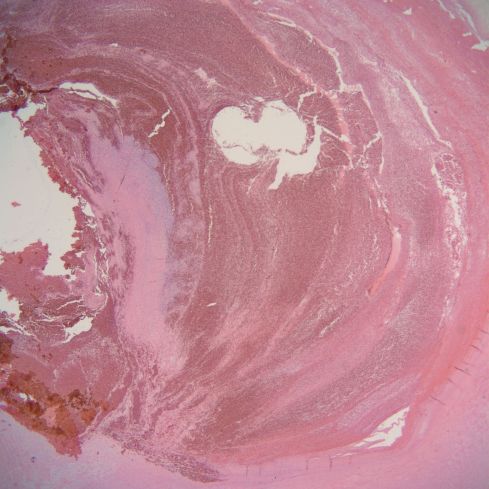
Figure 6 b: This photomicrograph shows a laminated thrombus in a similar incidentally found umbilical vein aneurysm.
This lesion is relatively common and may have a developmental origin different from the larger varices in the cord. This lesion has not caused a cord hematoma in the cases I have examined, but may be filled with laminated thrombus. These are just my observations on a small number of cases, and an understanding as to the causes of these dilated sections of umbilical vein and the events that lead to rupture and hematoma formation are not understood.
The initiating etiology of umbilical vein varices and aneurysms could be an acquired injury or a developmental anomaly. The umbilical vein may be vulnerable to injury in part because it has no source of oxygen for its own metabolic needs except the oxygen in the blood in its own lumen. Other large veins in the body have surrounding tissue with oxygenated capillary beds. Further risk of injury occurs because the length of the flexible umbilical cord subjects the vein to torsion, wrapping and compression.
In uncomplicated pregnancies microscopic abnormalities of the umbilical vein are not uncommon. In one study 17 such areas were found in 1100 placentas submitted for pathological examination from 9,600 deliveries[35]. This prevalence is much higher than that of umbilical cord varices and hematomas so many may not progress. In my own experience such microscopic lesions are at least that frequent as in the published study (Fig 7).
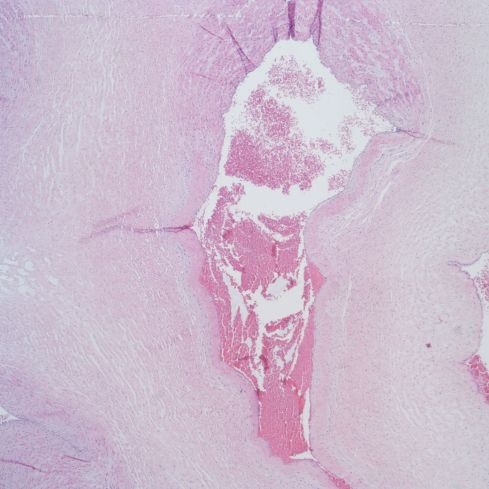
Figure 7a: This photomicrograph shows necrosis and aneurysmal dilatation of the lower half of the umbilical vein. This infant was small for gestational age with severe lympho-histiocytic villitis without villous evidence of utero-placental ischemia. (H&E, 4x)
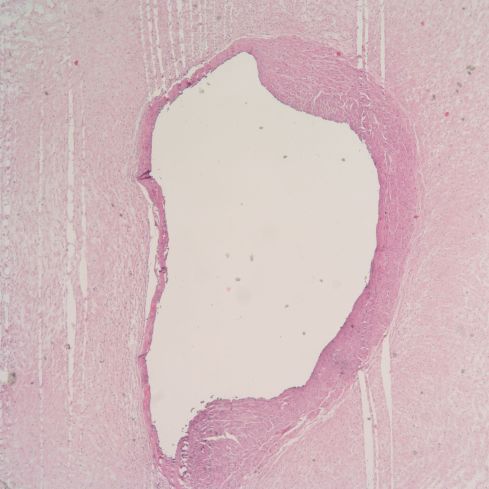
Figure 7b: This photomicrograph shows thinning and necrosis of approximately half of the circumference of the vein. This infant had a non-reassuring fetal rate that motivated an emergency Cesarean section. (H&E, 4x)
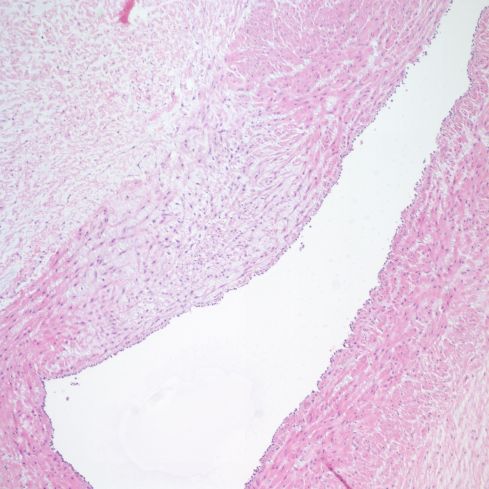
Figure 7d: This photomicrograph shows a focal thinning of the muscle cells with edema between fibers in the umbilical vein of a normal pregnancy and delivery. (H&E, 10x)
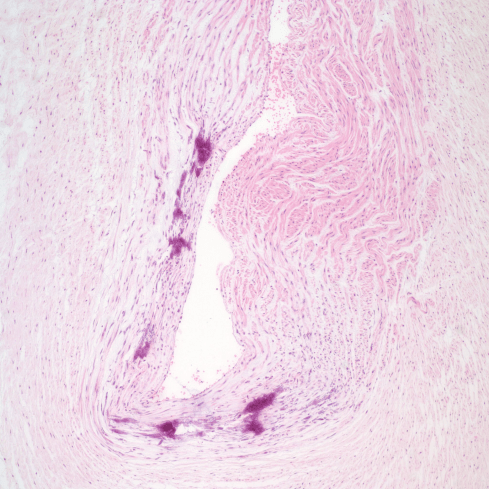
Figure 7c: This photomicrograph demonstrates necrosis and thinning of half of the venous circumference with dark blue calcification in the necrotic vascular wall. (H&E, 10x)
The thinned area of the vein in cross section is not always bulging outward, perhaps because the regions involved are small and venous pressure is low. However, the time course of development of larger varices and aneurysms, and of their rupture is not well described. I have seen intermediate lesions that are small but show some hemorrhage (Fig 7).

Figure 7e: The cord demonstrates a small variceal dilatation of the umbilical vein and focal bulges in the arterial wall.
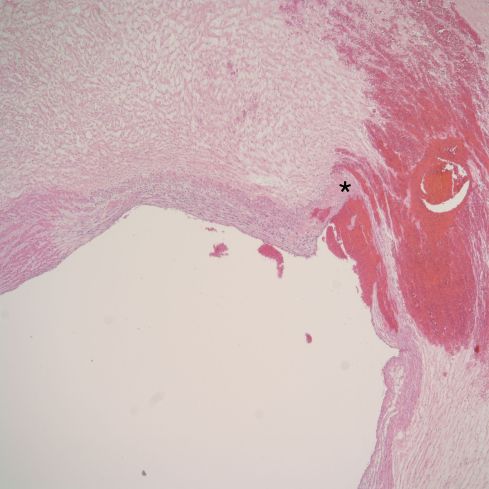
Figure 7f: This photomicrograph demonstrates thinning of the vein wall with very focal necrosis (pale pink area at the “*”) and a small hemorrhage into Wharton’s jelly. (H&E, 2x)
Whatever the cause, if a portion of the umbilical vein wall weakens, like aneurysms elsewhere in the body, the circumferential wall tension will increase according to Laplace’s law (wall tension in a hollow cylinder is directly proportional to the radius) and the lesion will expand. Less clear is how any of these umbilical vein aneurysms rupture in the low pressure venous system. The initiation and time course of expansion of varices of the umbilical vein remains to be elucidated.
One likely developmental/genetic example of varix formation in the fetal circulation is in the context of mesenchymal dysplasia. In a Doppler study of a varix in a placenta with mesenchymal dysplasia, umbilical venous flow was uncompromised despite the observation of mural thrombi in the vessel after delivery[36]. The large surface vessels in mesenchymal dysplasia arise from a markedly hyperplastic vascular network in the vili. In one placenta that I received the initial impression on prenatal ultrasound examination was that of dilated loops of bowel on the placental surface from gastroschisis (Fig 8).

Figure 8a: The fetal surface of this placenta demonstrates large sausage like dilations of chorionic veins starting at the insertion of the umbilical cord.
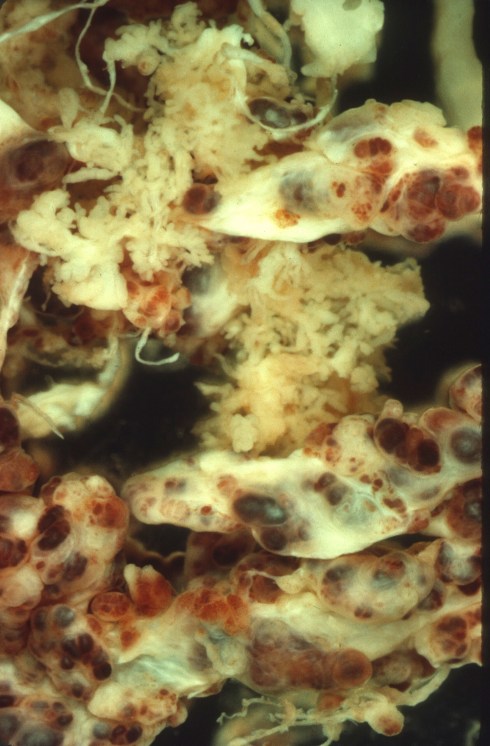
Figure 8b: A dissecting microscope demonstrates the large tortuous vessels within the villi of the placenta in the same case as Figure 8a.
The reason for the massive dilation of the vessels on the surface of the placenta and at the junction with the umbilical cord must reflect some weakness in these vessels, as the umbilical vein was not massively dilated.
In reports of fetal death with umbilical cord varices, the mechanism of death is often attributed to a fresh thrombus[37, 38]. Logically, a thrombus that severely narrowed the diameter of umbilical blood flow or completely occluded it could be lethal, but reports of acute thrombi need careful pathologic correlation. Distinguishing a fresh thrombus from a postmortem clot can be very difficult even with microscopy. A review of thrombi in normal umbilical veins found either that the infant had normal Apgar scores, or the infants were stillborn, which causes some concern about the accuracy of diagnosis.[39]The association of vessel thrombosis with poor outcome was as likely related to other obstetrical complications in most of the cases. This topic will be expanded in a subsequent page.
The umbilical arteries despite being subject to higher blood pressures and less oxygenated blood have fewer aneurysms reported than umbilical veins[40-42]. The limited case reports demonstrate a probable association with trisomy 18 (one with an arterial aneurysm to vein fistula in the cord)[43, 44]. Others have been associated with sudden fetal death[40, 42, 45, 46]. In the case reported by our group, we speculated that the large calcified aneurysm kinked or bent the other vessels causing fetal asphyxia (Fig 9)[45].

Figure 9a: The umbilical cord has an enlargement due to an aneurysmal dilatation of an umbilical artery. The area shows edema of Wharton’s jelly at both ends creating an ovoid appearance, and is stained with hemoglobin in the jelly.

Figure 9b: The umbilical artery has been injected with barium, and a radiograph shows that the barium enters the dilated aneurysm.
Intra-abdominal varices of the umbilical vein are no longer within the umbilical cord and logically cannot contribute to umbilical cord hematomas. In theory however, continuity with the intra-abdominal cord portion could occur. These varices generally have a good prognosis, and in at least one case report may have been due to the umbilical vein entering the portal system more proximally than normal[34, 47-50]. There are cases reported with anomalies. There is a case report of a varix in a persistent right umbilical vein[51]. This right vein in humans normally regresses with the left carrying the cord blood through the liver to the heart . Its persistence has been associated with malformations. Intra-abdominal umbilical vein aneurysms may have turbulent flow and even thrombosis. I have never seen a case in my stillborn autopsies.
Umbilical cord hematomas are not a major contributor to the incidence of stillbirth, but a better understanding of the development of umbilical vein varices and subsequent hematomas could not only prevent stillbirth, but also prevent unnecessary therapy for non-progressive lesions. Some hematomas occur during labor and it possible that stretching or temporary occlusion of the vein might play role. Unfortunately experimental models in lambs would be difficult and expensive not to mention that lambs have 2 umbilical veins in the cord. At the least a central repository of case data might be useful that included the size, location and outcome.
Umbilical cord wrapping, entanglement, encirclement
Anyone who has used a garden hose knows that water flow can be stopped by physical changes in the length of the hose unrelated to direct external compression. Problems related to the flexible length of the cord, analogous to the garden hose, present other mechanisms for umbilical cord “accident.” We will consider these next.
The most common form of umbilical cord wrapping is around the neck (a nuchal cord), or perhaps it just seems more common because the loop does not slip off with fetal movement or delivery. Even nuchal cords come and go during gestation, although they become more frequent towards term with a frequency usually just above 25% at delivery1, 2. A posterior location of the placenta has been associated with nuchal cords, but how this location favors nuchal wrapping is unclear 3, 4. Nuchal cords correlate with complications such as low 1 minute Apgar, meconium passage, lower cord pH, or lower birth weight, but the effects are of low magnitude in the pooled population of infants5-10. Some infants may have significant complications of nuchal cord due to particular circumstances. Nuchal cords can be sub-classified by the number of nuchal wrappings (nine times around the neck in one case report11), by complex wrapping of other body parts, and by the type of loop. Type “A” the result of a simple 360˚ rotation that must end with the placental end of cord on top, versus type “B” in which the fetus rotates through the loop creating an incipient knot with the fetal end of the cord on top12, 13. Other studies have sub-classified tight nuchal cords (those cut to enable delivery) versus loose cords14. As will be discussed, tightening of the cord is usually due to fetal movement causing traction on a short segment of cord between the placental insertion and the wrapping. In general the more complicated the nuchal wrapping, the more likely that there will be fetal complications15-18.
Often implied, but not always explicit, is the notion that cord wrapping can cause a functional short umbilical cord, and that labor or descent of the head can put traction on the cord. Such traction is probably the mechanism of a tight nuchal cord. I did not find studies that measured the distance from the end of the wrapping to the placenta, but this would be a crucial measure to evaluate functional cord shortening. Such a functional short cord may be a reasonable explanation for acute fetal hypoxia with delivery or descent in the minority of infants with nuchal cord wrapping. One observational study noted that some stillborn infants had tension on the cord from the umbilicus to the neck, but this must be an uncommon cause of death if it is directly related[52].
Any anatomic variation of wrapping the cord around the fetus (body, limbs, neck) that causes a functionally short cord could cause intrapartum stillbirth, and some prepartum stillbirths during descent of the head into the pelvis (Fig 10).
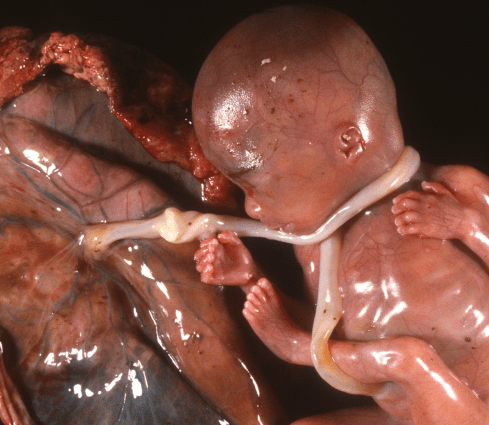
Figure 10: This fetus received en caul (still in the amniotic fluid sac) demonstrates how a wrapped cord creates a short cord between the fetus and the placenta, and perhaps how a second wrapping around the leg could tighten the cord wrapping in place. This was from an early preterm labor, and the cord wrapping had no known influence on the outcome for this fetus.
There are multiple potential mechanisms for a short cord to cause fetal hypoxia or death. These include cord traction pulling on the placenta and causing premature separation or even cord rupture at the placental insertion (manifested as subamnion hemorrhage). Fortunately, these are rarely observed complications. Plausibly, a wrapped and hence less mobile cord might become trapped and compressed against the pelvis. In theory an extreme stretch of the cord could produce vascular injury and stimulate arterial constriction or thrombosis[53, 54]. A medical student, and I stumbled on another mechanism of umbilical cord blood flow occlusion directly related to a short cord. In the course of a simple experiment with in vitro umbilical cord perfusion, we found that the shorter the free cord was after wrapping it around a PVC pipe, the less twist (perpendicular rotation) was needed to stop umbilical vein flow[55](Fig 11).

Figure 11a: This is the apparatus that we used to measure in vitro umbilical blood flow in umbilical cords wrapped around pipes of different diameter and subject to torsion of the outflow end of the cord. Not shown in the photograph is the tank of green saline that was attached to the inflow end of the cords. The outflow was measured as it flowed into a glass cylinder.

Figure 11b: This was an example of a rotation of the umbilical cord with a long segment. There is minimal change.
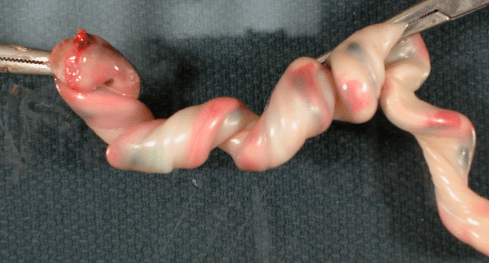
Figure 11c: This is an example in a short segment of umbilical cord that shows marked kinking for the same degree of rotation as in figure 11b.
This is actually simple geometry. A given twist in a short cord produces a much greater pitch (turns per length) than in a long cord. At a critical pitch, the vessel lumen collapses. This has not been reported to occur in clinically, but has not been searched for in living infants.
The indentations of the neck with nuchal cord can be deep, but there is little evidence that the cord causes significant tracheal obstruction. A few case reports suggest that the cord interfered with fetal swallowing and caused polyhydramnios[56]. There may be some jugular venous obstruction especially if the nuchal cord tightens[57, 58](Fig 12).
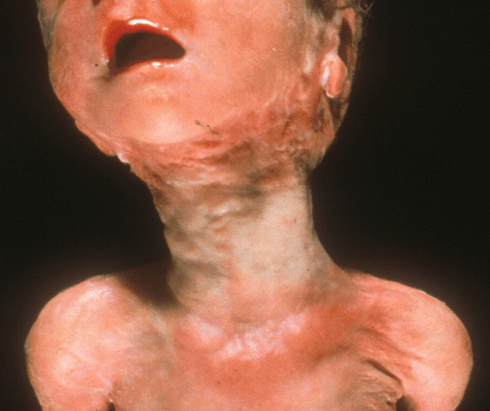
Figure 12: The neck of this stillborn infant demonstrates the compression marks and elongation from a tight triple wrapped nuchal cord. As with the more common single nuchal cord wrapping, this is the result of chance motions of the fetus. The cord likely causes asphyxia because it is a short fixed cord.
The presence of a nuchal cord alone is not a cause of stillbirth[59]. In an otherwise unexplained fetal death, assigning cord wrapping as the cause of a stillbirth is supported by finding a complicated wrapping or a very tight cord as evidence of functional short cord [52, 60-63]. Demonstrating a short length of the free cord from the neck to placenta or from the neck to the umbilicus would support cord traction or torsion as a mechanism of death. If the cord is still attached to the infant, the type A or B of wrapping can be determined simply by which segment of cord is on top (cephalad). Type B may be more likely to result in cord occlusion.
Umbilical cord length
We have proposed a theory as to how cord wrapping might lead to fetal asphyxia by twisting the short end. We have implied, but not shown, that normal fetal movement accidentally wraps the cord around the infant. Perhaps this wrapping is not just chance. There is evidence that cord wrapping is significantly associated with long umbilical cords[64-66]. Why does this association occur? We could propose that long umbilical cords are just part of the random distribution of cord lengths around a mean length, and that they are just more likely to get in the way of the fetus and therefore entangled and wrapped. I think that a more insightful hypothesis can be developed by considering the basic mechanisms that control umbilical cord length, and that this hypothesis will also lead to better insight into another association of long cords linking them to fetal asphyxia, and will lead back to our hypothesis that wrapping produces a functionally short cord, even if the cord is long at the time of birth. The onset of wrapping would still be an accident of fetal movement.
If we consider the development of cord length as an engineering problem, we want to create a cord that is the minimal length needed to assure that the infant can be delivered without pulling the placenta off the uterine wall. Since this needed distance has some variability due to the site of placental implantation and the fetal presentation, the final length must be long enough to account for these variables. As the fetus grows, the uterus will also expand, and the umbilical cord will need to lengthen in compensation. Studies have shown the length of the cord does increase with fetal gestation and size, and that the normal range is approximately 40 to 70 cm in length with the mean varying with gestational age, weight, and sex of the infant[67-71].
The clue to the control of the umbilical cord length comes from clinical experience with very short cords. The common factor associated with a short cord is a lack of fetal movement. The most extreme condition is fetal paralysis from a number of conditions that then result in rigid joints, small lungs (from lack of respiratory movement) and a short umbilical cord[72, 73]. Most such paralysis is due to intrinsic fetal neuro-muscular abnormality, but unusual causes are maternal myasthenia gravis with transfer of an anti-neuromuscular junction IgG, connective tissue restriction in Neu Laxova syndrome and maternal treatment with curare for tetanus[74-76]. The cord is also likely to be short with any restriction of fetal movement including a lack of amniotic fluid, amniotic bands, and even very short limbs as occurs in severe osteogenesis imperfecta and thanatophoric dysplasia[77, 78]. Experimentally the administration of curare to the mother rat produces the same fetal findings as paralysis in human fetuses[79]. In the terminology introduced by Dr. David Smith and colleagues, the condition is called fetal akinesia/hypokinesia sequence.
There is even a plausible hypothesis for the mechanism of stretch induced growth. The internal collagen coiling of the Whartons’ jelly is attached to charged glycoproteins that bind growth factors keeping then inactive[80]. With stretching the factors are released and growth of collagen and vessels is initiated[81]. This proposed mechanism would need to function despite the fact that the cord is long enough to usually be slack in utero. The mechanism could be triggered if incidental entanglement with normal fetal movement provided the necessary tug. The clinically important implication of this hypothesis is that a wrapped functionally short cord increases fetal tugging on the cord and results over time in elongation of the cord segment between the placenta and wrapping. Given enough time that cord segment will return to the mean length of a normal cord. The total cord length at delivery will be the elongated segment attached to the placental insertion plus the cord that is wrapped around the infant. Thus, a long cord implies a period in which the cord was short due to wrapping or entanglement.
I do not have direct proof of this theory, and there are missing molecular steps to be understood. However, the theory is consistent with known facts, and would explain the curious association of long cords with evidence of fetal asphyxia in some studies[65, 66]. These studies found a correlation of longer cords with non-reassuring fetal status in one and lower cord blood gas pH in another. They also confirmed the correlation with cord entanglement and deep variable decelerations of the fetal heart rate. One case was singled out for description in which the fetus developed bradycardia, meconium passage, and had an umbilical cord blood gas of pH 7.11 and a base deficit of 19.9. The umbilical cord was 131 cm long and encircled the fetal neck 4 times[66].
As a practical matter for the pathologist, the length of the cord attached to the placenta is usually not the full length, and in emergency procedures and with tight nuchal cord, the cord may not be cut close to the infant, making the cord attached to the placenta much less than the true length. Of course, a cord that is longer than 70 cm attached to the placenta, is a long cord. To really understand the role of wrapped cord in causing fetal asphyxia, there is a need to document the in vivo effects. The hypothesis that very short distances between the placental insertion and the begin the wrapping of the cord may make the cord vulnerable to torsion could be directly tested without needing to trace the whole cord. The possibility that wrapped cords become trapped between fetal parts and pelvic bones might also be testable in vivo even during labor. The use of Doppler velocimetry in living infants is an advantage over studying stillborn infants in utero, but the lack of urgency to decide on emergency therapy may be an advantage. If any cord wrapping anatomy proved to be an important cause of fetal asphyxia, the next goal would be to determine if the cord could be manipulated into a less dangerous arrangement by instrumentation, either via the cervical os or even transabdominally.
Umbilical Cord Knot
True umbilical cord knots are found in approximately 6 of every thousand deliveries based on an average of multiple studies (1198/201,612)[82-91]. If the fetus swims through a loop of umbilical cord, this act creates a simple overhand knot. The fetus can also perform more complex knots that have been diagrammed (Fig 13) [92, 93].
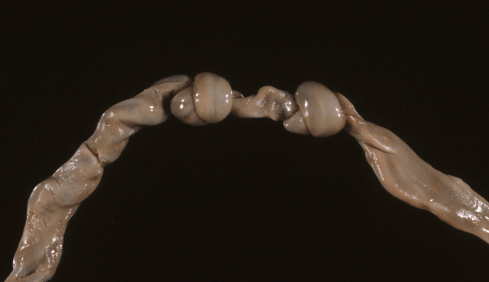
Figure 13a: This umbilical cord has two tight knots but without a clear difference in the appearance of the vessels on either side of the knots. The infant was severely growth restricted secondary to maternal floor infarction (MFI). The knots may have tightened after death, but have no known relationship to MFI.
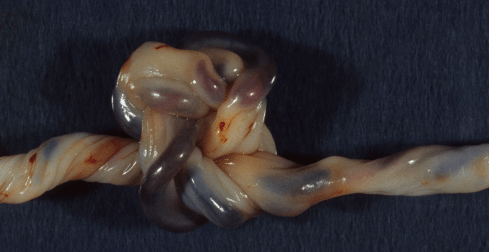
Figure 13b: This umbilical cord demonstrates a complex double knot as an incidental finding.
The existence of umbilical cord knots demonstrates the flexibility of fetal swimming and adequacy of the intrauterine space for such complex maneuvers. Some fetuses even manage to form more than one knot in the cord, perhaps by swimming through a complex snarl of cord created by rotation of the fetus [94]. Knots should occur more frequently if the ratio of amniotic fluid to fetus is large which is true in early gestation and with polyhydramnios, a risk factor for knots[95]. If the fetus remains stuck in the middle of the loop, the knot may only be a potential knot until the infant slips through the loop. The fetus being caught in the loop is likely frequent given the association of the umbilical cord knots and long cords. Except for the subtle twist of one cord portion around the other, the knot would appear to be a simple wrapped cord. Even if the knotted cord is free of the fetus, the fetus cannot easily tighten it. The umbilical cord is longer than the uterine dimensions, so normal movement should not put continuous traction on the knot. However, if the cord subsequently wraps around a fetal part, the free length of the cord may shorten sufficiently to tighten the knot[96, 97]. Some knots may not tighten until the delivery of the infant puts tension on the cord.
A tightened knot could compress the umbilical vessels and occlude blood flow. An early review in 1925 suggested most of the ideas that have been reported with umbilical cord knots and stillbirth: increased movement before fetal death, wrapping around the body or part is a way to tighten the knot, some knots are around the baby as loops until pulled through at delivery, older knots have compressed areas of Wharton’s jelly and return to form untied, in death there is swelling of the cord on at least one side[97].In addition to apparent swelling, stillborn infants with very tight knots may show distinct differences in color on either side of the knot consistent with stopping arterial blood flow (Fig 14).
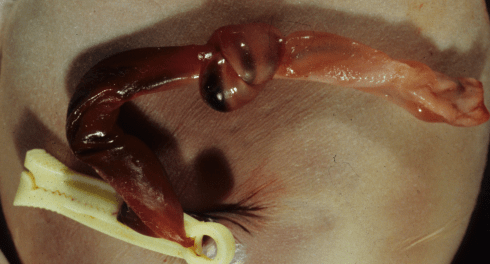
Figure 14 a: This umbilical cord has a tight knot near the umbilicus and the cord on the fetal side of the knot appears redder than the placental side due to vessels distended with blood.
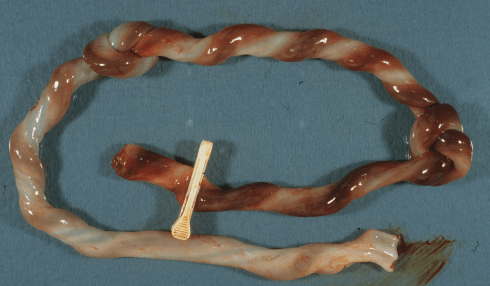
Figure 14b: This umbilical cord has two overhand knots with some pallor on the placental side after the second knot. The infant was stillborn with thrombi in the stem vessels of the placental villi.
If blood flow were obstructed in the umbilical vein, both sides of the vein would be expected to dilate due to backpressure from the vena cava and from placental flow into the placenta. If the artery is obstructed, the vein is also, and placental backpressure would be less likely to distend the umbilical vessels on the placental side of the knot. This is speculation based on observing the knots, but very little is known of the hemodynamics of umbilical vascular occlusion in the cord.
Second, the prevalence of knots in the cord in stillborn infants in various studies averages 3% (16/559) [52, 86, 89]. This is 5 times the average incidence of umbilical cord knots in all infants (0.6%). Also studies show that in aggregate if the umbilical cord has a knot, 4% (52/1209) [82, 83, 85, 86, 88, 89, 91, 98, 99]of those infants will be stillborn which is an order of magnitude higher than most published stillbirth rates. Finally, in vitro evidence shows that tying a mass to a knotted cord and letting gravity pull one end will stop umbilical vein flow[100]. The masses required varied from 50 mg to 300 grams with thicker cords requiring more force. The values in vivo may not be the same, but the principle is established. A reasonable conclusion is that umbilical cord knots can be a cause of stillbirth if conditions allow them to tighten sufficiently to stop umbilical blood flow.
A surprising finding given the above conclusion is that umbilical cord knots do not appear to be associated with neurologic injury. The Collaborative Study of Cerebral Palsy study (17,190 deliveries) found a very significant association of umbilical cord knot with stillbirth, but did not show a statistical association between umbilical cord knot and neurologic abnormality at seven years of age[83]. There is a single case report of venous distention behind the first knot of three in an infant with cerebral palsy[101]. Multiple studies often confirming the association with stillbirth have not found clinically significant compromise in the neonate. Some have found an increased incidence of abnormal heart rate patterns or even increased Cesarean section due to abnormal fetal heart rate pattern, but the numbers are small[84, 85, 91]. One small study showed no difference in arterial blood gases with cord knots[102]. One of the studies demonstrated a lower 1 minute Apgar score which was attributed to tightening of the knot with delivery[103]. A study that showed no difference in Apgar scores nor in Cesarean delivery, did find an association with cord wrapping and longer umbilical cords[91]. A relatively large study of umbilical cord knots (N=841) also confirmed the association with various measures of fetal distress (meconium, non-reassuring fetal heart rate, Cesarean section), as well as stillbirth[95]. The consensus is that an umbilical cord knot per se is not a major risk to neonatal well being unless it causes death. This conclusion begs to be understood. The logic would seem to be that the knot can cause distress or death, but does not produce the conditions of brain injury. As discussed, the most severe patterns of experimental brain injury occur with partial or recurrent intermittent asphyxia, while complete asphyxia has a narrower window between onset of neuronal necrosis and death. It is unlikely that birth would just happen to occur in this window of brain injury if the knot tightened and stopped all blood flow.
However observations of infants that have died with a cord knot do not seem to confirm this hypothesis. There are reports of a sudden increase in fetal movement and then a decrease, or just a decrease in movement, or a non stress test with loss of variability[52, 97, 104]prior to fetal death. One stillbirth with 3 days of decreased fetal motion, and then loss of variability on an NST five hours before the fetal heart rate was no longer detectable[104]. Another infant demonstrated bradycardia, followed by a sinusoidal pattern and ending in stillbirth. The autopsy demonstrated no other cause of death, but the case report did not describe the knot[105]. These observations suggest that the studies may not have had the statistical power to capture the rare infant with a knot delivered in a compromised interval who then developed brain injury.
Prenatal observations of blood flow in umbilical cord knots have not been very helpful in understanding knots. Three-dimensional power Doppler can confirm knots often first suspected on two-dimensional ultrasound, but this poses a dilemma over how to manage a prenatally discovered umbilical cord knot[106-110]. In a study of umbilical artery waveform notching, one case had a true knot but this was not statistically significant[111]. Ideally, it may be possible to look at the conditions of the knot. One in a free segment of normal length cord should be low risk. A knot associated with a wrapped umbilical cord creating a short cord that could potentially tighten would be a high risk. The problem is that we have no current therapy except close observation for abnormal fetal heart tones and delivery. At the least one could be prepared that if the fetus did not slip through the wrapping with delivery, the knot would tighten and be a potential problem with fetal descent. In the extreme case of a class three heart rate tracing, as the cases of stillbirth with bradycardia, loss of beat–to-beat variability, and sinusoidal pattern, at least the caregiver would know that the situation was not going to relent.
Umbilical cord knots would logically be an added risk of asphyxia in fetuses with umbilical cord wrapping. Unfortunately we still can’t unwrap or untie the umbilical cord in utero. Even if a prenatally discovered knot could be untied without risk to the fetus, the impact on the rate of stillbirth would be low because of the low frequency of this cause. The caution has been published that even an accurate prenatal diagnosis of a cord knot would not be of practical benefit[112]. However, a strong predictor of fetal death based on antenatal findings with a cord knot might still have diagnostic benefit, although none has yet been discovered.
Pseudoknots, an aside
Psuedoknots are just that, not really knots. They are hypercoiled umbilical vessels within the umbilical cord (Fig 15).
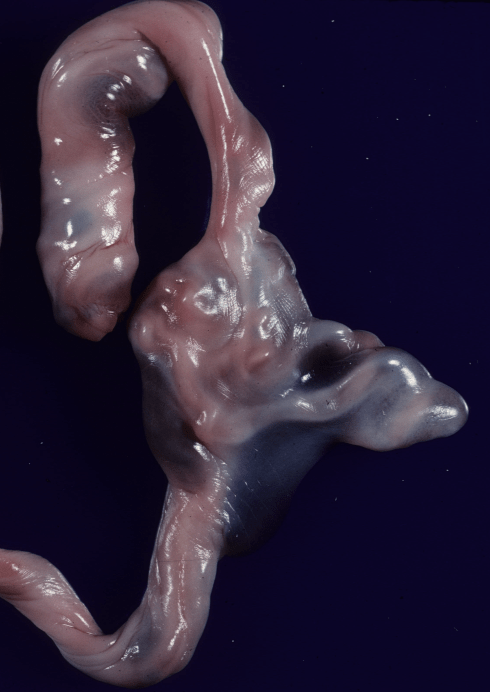
Figure 15: This cord has a prominent pseudoknot, which is a tangle of vessels within Wharton’s jelly.
A possible mechansim can be demonstrated by twisting a cord fixed at one end until hypercoils form, but unlike pseudo knots these involve the entire cord not the vessels. Another possible mechanism is overgrowth of the umbilical vessels within the cord. One study has suggested a correlation of maternal cigarette smoking with pseudoknots[113]. They are not considered clinically significant, but the understanding of them is incomplete.
Umbilical cord torsion
In thinking about the risk of cord occlusion with a short cord, the concept of twisting the cord to stop umbilical flow was introduced. There turns out to indeed be a twisted topic. Simple observation of the cord demonstrates a helical path of the umbilical cord vessels. These vessels are tightly embedded in the cord and twisting the cord manually will twist the vessels as well. One of the earlier papers to consider this vascular helix in the cord, started by noting the predominance of right handed twists (75%), and pondering this lack of randomness considered hypothesis that the fetus was turning in one direction, and the twist might reflect neurologic dominance, or handedness. The handedness of the cord vessel helix can be established by duplicating the direction of coil with the fingers of the hand and then noting the direction that the thumb points with the bottom of the hand toward the placenta.
One study has proposed that the number of cord twists and presumably the handedness is established very early in gestation, 9-13 weeks, when the collagen structure might allow free movement of the vessels in Wharton’s jelly[114]. This seemed more logical to me than the fetus actually rotating in one direction to establish the direction and number of vascular coiling. Intuitively, a flexible coil with fluid flowing through it, when confined by a rigid outer would hit the wall of the outer cylinder and form a helix. Rather than try to prove this mathematically, I took the simple approach of attaching a thin rubber tube to a garden hose and then putting it within a much larger curved clear plastic cylinder. When I turned the water on, the small tube did form a helix within the larger, and the handedness could be changed by the starting angle of water stream in the small tube (Video 1))
(I need to upgrade to a premium plan for the site to add the videos!)
. This result requires adequate pressure in the small tube, and the flow corresponds to that within the umbilical vessels in this simple model. Cords without a helix by ultrasound have been associated with an increased incidence of fetal death[114, 115]. In this simple physical model this would correspond to inadequate pressure/flow in the umbilical vessels to force them against the wall of the cord.
These intrinsic vascular coils do not exclude twisting of the fetal vessels after they are firmly fixed in Wharton’s jelly to cord surface. When I was a resident, local myth held that twisting the cord while waiting for the placental separation (third stage of labor) might hasten the process. This twisting of the umbilical cord after delivery of the infant causes a visible increase in the pitch (number of twists per length). This is just geometry. The pitch will increase in relation to the angle of rotation perpendicular to the length of the cord, but the numerical increase also depends on the length of cord being twisted. The clinical question is whether the fetus can and does significantly further twist the cord in utero. In effect does the fetus spin?
The answer appears to be yes, at least passively. A common finding in midtrimester stillborn infant who have had a prolonged period of retention in utero is that the umbilical cord is markedly twisted. In early and mid trimester fetuses there is relatively more amniotic fluid and room to move in utero. I suspected from the circumstances, and more importantly from the presence of this twisting even with a verifiable unrelated cause of death, that this twisting occurred postmortem. To test this idea, I suspended a smooth oval rock on a string inside a large jar filled with water. The string was fixed to the lid. Shaking the jar and then setting it down resulted in the rock beginning to rotate (Video 2).
In effect the random vectors of movement constrained by the jars cylindrical walls, summed up into a rotational force. Once the Wharton’s jelly is soft from postmortem retention, I think each of these rotations adds a permanent rotation to the cord, and these are summed over time. This interpretation means that the cord twists per se are unlikely to be the cause of death. However, it is conceivable that a fetus who was unable to resist the passive twisting forces of maternal movement might succumb. In vitro our study of cord wrapping, showed that eventually flow could be stopped with enough twists even in the longer portions of cord, although our experiment did not approach the full length of the cord.
In our in vitro experiments we measured the natural pitch of the cords, which varied widely (look up numbers), and demonstrated that it did not affect flow rate but our measurements were crude based on a stopwatch and graduated cylinder. The pitch is usually reported as a coiling index in pathology reports. One study correlated a high coiling index with fetal demise, fetal intolerance to labor, intrauterine growth retardation and chorioamnionitis [116]. This study measured the additive pitch of intrinsic helix and external torsion. An ultrasound study also emphasized the distinction between vascular helix and external torsion[117]. The distinction between the two mechanisms affecting the vascular pitch is important, but still not clear to me how to make the distinction in practice (Fig 16).

Figure 16a: This umbilical cord was used in our experiments that perfused the umbilical vein with green saline, and it demonstrates very clearly the vascular helix of the umbilical cord. The number of coils per length (pitch) can easily be measured.
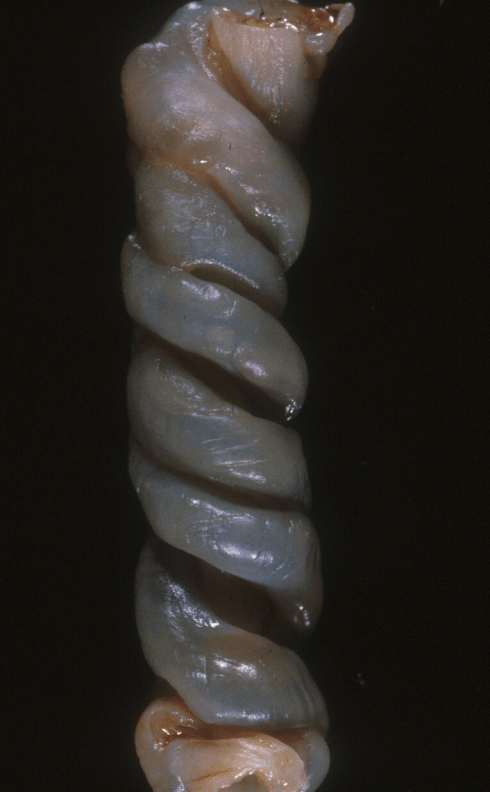
Figures 16b: This umbilical cord has a high pitch, or hypercoiled vessels in a twin that was perfusing an acardiac twin. In many cases, there is no direct association with increased blood pressure or other clinical correlation to account for the coiling.
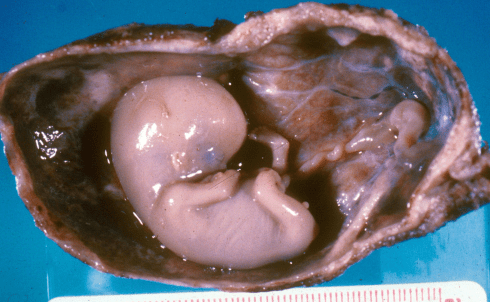
Figure 16c: In this early gestation fetus delivered within a decidual cast, there appears to be superimposed twists on the umbilical vascular coil likely due to postmortem rotation of the fetus.
Kinking
Anyone who has struggled with a garden hose is likely to be aware of the problem of a hose kinking. The hose starts out on the ground coiled, and as it is pulled on to lengthen it, a small loop forms in the hose instead of becoming straight. There is a clear torsion on this loop perpendicular to the length of the hose, and eventually this collapses the hose lumen at the top of the small loop. In order to quickly stop flow in a hose, it is far more effective to simultaneously twist and bend the hose until a segment of lumen collapses than to try to stop the flow with direct pressure.
Since some hoses are advertised as non-kinking, I emailed one of the firms, and an engineer replied that they avoided kinking by strengthening the helical fabric of the hose wall. Then if the stronger wall prevents collapse of the lumen, you can pull the small loop straight. Clinically, does kinking occur in the umbilical cord? I don’t know, but I suspect at the attachment of the vessels to the umbilicus, a kink could occur spontaneously. If such a mechanism did cause fetal asphyxia, any lesion in the cord would be very subtle.
Umbilical cord insertion:
The umbilical vessels may be more susceptible to occlusion at the points that that they join the fetal body (the umbilicus) or the placenta. The body stalk of the fetus has to join the vessels forming on the placenta and generally does this on the surface of the placental disk. Approximately 1% of the time (and more frequently in twins), the cord misses and inserts on the fetal membranes, and the arteries and vein devoid of Wharton’s jelly travel along the chorion membrane to reach the placenta. This type of insertion is referred to as velamentous (vela referring to the membrane similar to the root of veil, not villamentous as I sometimes have heard residents refer to it!). These exposed vessels can be subject to rupture, and will be further discussed in the chapter on fetal exsanguination. The vessels may also be vulnerable to compression by a fetal part as they often splay near to each other, and conceivably with labor or rupture of membranes all be compressed within that confined area. Even more frequently the cord attaches to the margin of the placenta, sometimes called eponymously a Battledore insertion. This less secure attachment often appears as if it could be kinked or compressed (Fig 17).
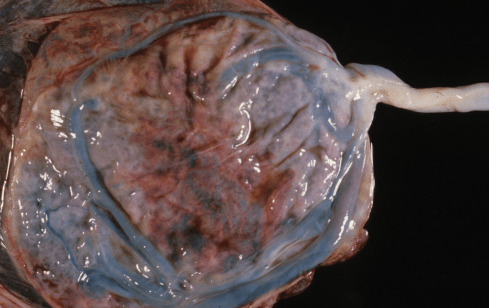
Figure 17a: The umbilical cord is inserted into the margin of the placenta and did not cause any known complications.
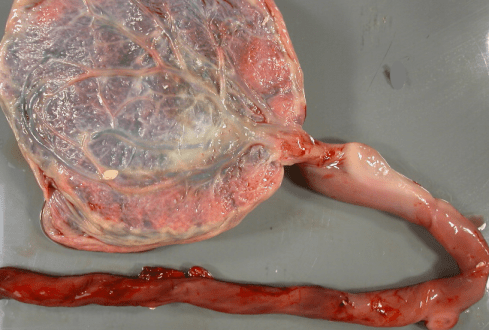
Figure 17b: This marginal insertion of the cord in a stillborn infant appeared narrowed and eroded.
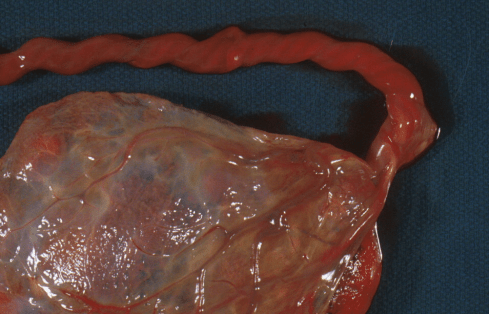
Figure 17c: This marginal insertion of the cord in a stillborn infant appeared narrowed and vulnerable to twisting.
A third abnormal insertion of the cord is the furcate or forked insertion in which there is a loss of Wharton’s jelly before the cord vessels contact the placental surface. These vessels may be vulnerable to not only compression but to torsion. This appears again to be a problem with a short cord in that the bare area takes all of the turn over the short distance and at least in vitro the vessels appears to twist and collapse (Fig 18).
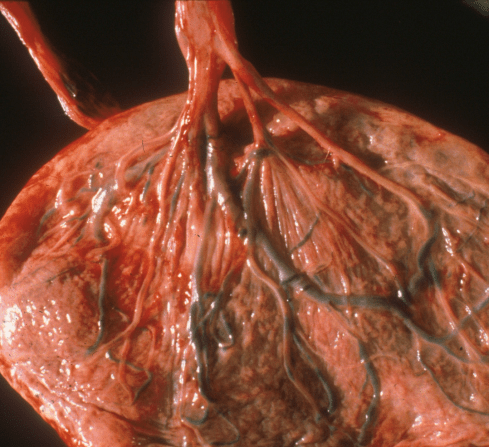
Figure 18: This furcate (fork like) umbilical cord insertion demonstrates the loss of Wharton’s jelly that could make the vessels subject to collapse with the application of fetal turning. The infant was stillborn without other explanation.
An early review of the pathology of spontaneous abortion noted that there was a high incidence of velamentous and Battledore (marginal) insertion of the umbilical cord in mid-trimester abortions[118]. However large-scale studies have not shown a high risk of fetal asphyxia with abnormal cord insertion. A prospective study of 1000 placentas failed to demonstrate a significant association of the location of the umbilical cord insertion with low birth weight, intrauterine fetal death, threatened abortion, premature delivery, fetal malformation or intrauterine fetal hypoxia (not further defined)[119]. They lumped marginal and velamentous cords into a single “peripheral insertion” category that had 72 placentas, approximately 7%, while only 1% would have been expected to be velamentous. A clinical study of velamentous insertion in 12,750 high risk patients found 216 cases, excluding twins and malformations or aneuploidy[120]. In 48 cases with fetal Doppler studies, all abnormalities were believed due to preeclampsia. A review of 15,865 singleton deliveries, using the clinical record of the obstetrician’s observation of the delivered placenta, found 77 (0.5%) velamentous cord insertions[121]. There were 268 twins with 5 velamentous insertions (2%). there was a significant increase in meconium stained fluid, blood stained fluid, abruptio placenta, variable decelerations, and intervention with forceps or vacuum extractor. This latter study suggests some risk of fetal asphyxia with velamentous cord insertion. The conditions in which abnormal cord insertion causes asphyxia may be much less frequent than the incidence of the insertion abnormalities, making detection of the risk in a population difficult. These studies do not exclude the value of looking for examples of umbilical blood flow occlusion in individual cases with abnormal cord insertion.
The cord insertion at the umbilicus generally appears firm and not vulnerable to occlusion. The exception is the in mid trimester fetuses with prolonged postmortem intrauterine retention with softening of the connective tissue. Typically the cord just prior to entering the body in these fetuses can become very narrow. If there is also torsion of the cord, this area can appear severely thinned and twisted (Fig 19).
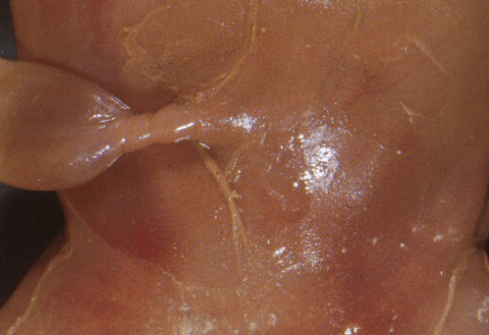
Figure 19: This close up of the umbilicus in a mid trimester stillborn infant shows the twisting and narrowing of the cord insertion often found with prolonged intrauterine postmortem retention in this gestational range.
I have not seen this condition in a stillborn without softening of the connective tissue. A reasonable explanation is that the thinning is a postmortem artifact.
Umbilical arterial constriction:
Not exactly an external compression, but clearly contraction of the arterial wall media can narrow the lumen and even stop umbilical blood flow. The normal umbilical cord has two arteries, each coming off an iliac artery. To completely stop flow both arteries would need to be constricted. If only one artery is occluded, almost all placentas have an anastomosis (fusion) of the two arteries as they enter the placenta. Of course, some placentas have a single umbilical artery and the wall contraction of one artery could cause asphyxia. A continuous arterial wall contraction of the arteries does occur normally after delivery of the infant. This peculiar muscular arterial contraction is necessary to stop flow of blood out of the infant’s umbilical arteries after delivery and severance of the umbilical cord. (Backward flow from the umbilical vein is stopped because of the pressure of the intrahepatic portion occludes back flow out of the infant.) In modern obstetrical practice, the umbilical cord is clamped to prevent blood loss, but for most animals or humans with unattended deliveries, no such precaution is available. If this umbilical artery contraction were to be triggered prior to delivery, the consequence could be lethal asphyxia, as the arteries appear designed to remain contracted and closed permanently until the umbilical stump is shed.
Arterial contraction is evident to the pathologist examining the cut cross section of the umbilical cord. In a live born delivery, on the microscopic slide, the constricted arteries appear closed giving the appearance of two eyes compared to the open “mouth” of the umbilical vein (Fig 20).

Figure 20a: This direct photograph of a microscope slide demonstrates the normal cross section of a delivered umbilical cord with two constricted arteries and open vein.
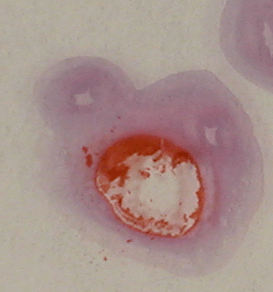
Figure 20b: This direct photograph of a microscope slide demonstrates a cross section of an umbilical cord from a stillborn infant demonstrating unconstricted umbilical arteries with open clear spaces in the lumens. The arteries may never have constricted normally or they may have dilated with death of the smooth muscle cells in the artery wall.
The umbilical artery has been observed to form constrictions in the artery with 20 seconds of birth that continue to increase in area over time. Some blood is trapped between these constrictions, called Hoboken nodes. The individual constrictions may not completely stop blood flow until 35 seconds or more after birth[122]. In an in vitro study of umbilical artery contraction it was noted that immediately after dissection of the artery from the umbilical cord it was often tightly constricted and that cannulation of the artery needed to be facilitated by either or both sodium nitrite or force with a blunt 18 gauge needle[54]. Once cannulated, “this constriction could last for 1-4 hr or more with no flow”. A study using thermodilution by injection into umbilical veins immediately following normal term deliveries found that flow dropped by more than 50% within the first 30 seconds after delivery[123]. There was wide variation, but initial flows averages approximately 200 ml/min, the flows decreasing from 100 to 25 ml/min. They did not use data if the umbilical vein showed visible contraction in response to the procedure. The same author also demonstrated a significant decreases in umbilical diameter starting 60 seconds after delivery and not associated with a significant increase in arterial oxygen content[124].
The ability of various chemicals to cause contraction of isolated umbilical artery smooth muscle or of rings of umbilical arteries with intact endothelium has been extensively studied[125]. These chemicals may control vascular resistance in response to pathological events such as blood loss or fetal hypoxia. Vaso-activity may also signal the growth of vessels and their smooth muscle thickness in response to long term changes in fetal growth or hypoxia. The effects at different levels of the umbilical placental circulation may be different for the same chemical. In the placental villi, vascular control may be needed for the equivalent of ventilation (maternal blood flow) perfusion matching which is not needed in the umbilical artery. Understanding chemical vaso-regulation is important to understanding placenta pathology of utero-placental ischemia and fetal growth. The complex interaction of control signals with physiologic events and anatomic changes in the fetus needs to be integrated with placental pathology in these respective areas. However, the post partum closure of the arteries may be an independent mechanism from the usual physiologic controls.
The closure at delivery must be triggered by a unique signal not present during intrauterine life, and it must be irreversible. Since the contraction occurs before the cord is severed, a change is fetal blood pressure is not a significant factor. Both cold and increased oxygenation of fetal blood following fetal respiration have been considered, but in vitro neither factor appears consistent nor intense enough to constrict the artery. The in vitro experiments generally use isolated artery or arterial muscle, a technique that interferes with discovering any translation of signal in Wharton’s jelly.
The search for a chemical signal for umbilical arterial contraction based on in vitro studies pointed to serotonin, or possibly bradykinin, as potential candidates. [126-129]. Serotonin can be secreted by platelet or mast cell granules. Umbilical cord microscopic sections stained with special stains show numerous periarterial mast cells not seen on routine H&E staining (Fig 21).
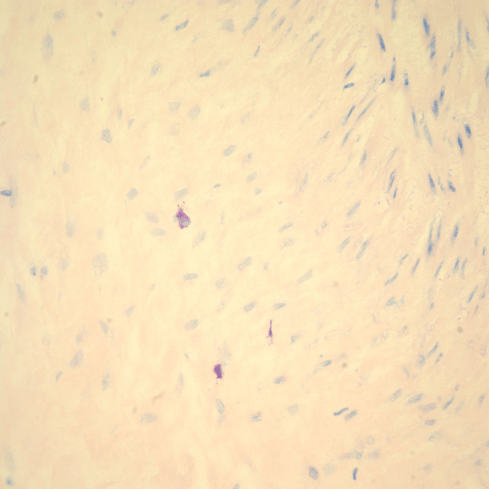
Figure 21a: This photomicrograph of Wharton’s jelly shows mast cells identifiable by the metachromasia that makes them appear purple with a blue dye. (Giemsa, 40x)
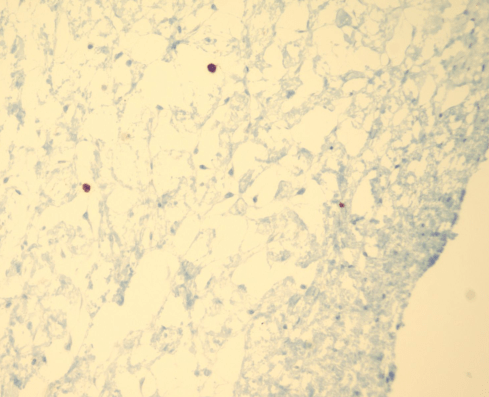
Figure 21b: This photomicrograph of Wharton’s jelly shows mast cells identifiable by immunological markers using that cause brown peroxidase precipitate. (CD117 immunohistochemistry, 40x)
Mast cell granules contain histamine, another potent constrictor of umbilical artery. Mast cells in the skin are responsible for hives (urticaria) with the release of their granules, but there are no small vessels in Wharton’s jelly to form the angioedema associated with hives. Umbilical arteries do not have an adventitia [130], and mast cell degranulation would be in direct contact with smooth muscle. However, there is no direct evidence that mast cells or histamine contribute to the mechanism of postpartum intrauterine arterial smooth muscle contraction. Their function in the cord remains unknown.
The failure of the usual vasodilators in the umbilical artery during serotonin induced contraction of human umbilical arterial muscle strips occurs uniquely due to the inability of these agents to initiate the usual cAMP/adenylate cyclase/cAMP dependent protein kinase or cGMP/guanylate cyclase/cGMP protein kinase needed for relaxation of the smooth muscle[131]. The authors note that this phenomenon has not been reported in any other smooth muscle. They also note that if the vasoconstrictor agent of umbilical artery smooth muscle is removed, then the muscle is capable of passive relaxation. These results explain the response to Nicorandil which can hyperpolarize the smooth muscle cell, but lack of a response to nitric oxide. A novel proposed mechanism for the refractoriness of umbilical smooth muscle is the expression of heat shock proteins[132].
As we noted in earlier in sheep studies, in the placenta bed itself irreversible vasoconstriction and very high resistance in the vascular bed is due to vasopressin and is triggered by a lack of blood flow in the fetal circulation when fetal lambs are placed on cardiac bypass for heart surgery. As we will see in the discussion of placental infarction, this mechanism may be part of a system to match placental and maternal perfusion of the placenta. However, overall the signals that normally control the flow through the placental system are poorly understood. An obvious anatomic observation is that the stem vessels in the placenta are all very muscularized and have intimal cushions. In the umbilical cord, only the arteries have intimal cushions that at least appear like a mechanism that assures the blood flow is occluded when the arteries constrict. The possibility that alterations in fetal-umbilical blood flow or in maternal intervillous blood flow in the placenta could trigger umbilical arterial contraction to the best of my knowledge had not been disproven.
We will discuss separately whether exogenous chemicals in meconium can cause arterial contraction in our discussion of the evidence for fetal asphyxia from the fetal autopsy. The possibility that inflammation in the cord can cause or predispose to arterial contraction has not been deeply investigated. There was a suggestive sheep study. Inflammation of the umbilical cord in human infants is relatively common compared to significant fetal asphyxia, but an effect on the umbilical vessels cannot be excluded, as inflammation is more common in preterm infants as in neurologic injury.
The umbilical arteries of stillborn infants are invariably dilated unless the period of post mortem retention is only a few hours in duration (personal observation). If intrauterine constriction occurs it may have relaxed with the death of the smooth muscle cell. In the cases of brief intrauterine retention, the umbilical cord may have constricted in the usual manner following delivery. Thus, there is no evidence for prepartum constriction of umbilical cord arteries in stillborn infants, but this does not refute the possibility.
Overall:
There are published case series of stillbirth associated with umbilical cord abnormalities[93, 104, 133]. The problem is that the array of lesions is varied, and some of the lesions may be postmortem artifacts caused by rotation of the fetus after death, or even compression of the cord after death. In one study, some of the deaths were due to previable delivery and the cord findings could have been incidental[93]. In cases with prenatal history, there was no consensus on a prenatal fetal heart rate pattern that predicted fetal death, although one study found a high incidence of non-reactive non-stress test. Dr. Jason Collins pointed out that in his patients, the two stillbirths with cord abnormalities had prenatal ultrasound identifiable complex cord lesions, a double nuchal cord in one and a triple knot in the other, and that such compound lesions could be a basis for risk screening[134].
Pathologically proving that an umbilical cord finding occurred death, and that it was the cause of death can be difficult. The role of maternal hypoxia from possible sleep apnea or other cause will not leave an anatomic trace. Maternal uterine pressure on the vena cava from supine position may leave no impression. Maternal ketoacidosis or fever may leave also are not going to be detected in the anatomic findings of the placenta or autopsy.
A review of unexplained stillborn deaths found a correlation with fetal thrombotic vascular changes that indirectly suggested compromised umbilical blood flow[135]. These thrombotic lesions had already been associated with abnormalities of the umbilical cord. We will consider the question of thrombi in fetal vessels and the significance of the finding in the next chapter.
References
- Yoshida, K., et al., Umbilical cord hematoma: association with chorioamnionitis and funisitis.Acta Obstet Gynecol Scand, 2007. 86(7): p. 895-6.
- Summerville, J.W., J.S. Powar, and K. Ueland, Umbilical cord hematoma resulting in intrauterine fetal demise. A case report.J Reprod Med, 1987. 32(3): p. 213-6.
- Dillon, W.P. and J.A. O’Leary, Detection of fetal cord compromise secondary to umbilical cord hematoma with the nonstress test.Am J Obstet Gynecol, 1981. 141(1): p. 102-3.
- Gardner, R. and R. Trussell, Ruptured Hematoma of the Umbilical Cord.Obstet Gynecol, 1964. 24: p. 791-3.
- Dippel, A.L., Hematomas of the umbilical cord.Surg Gynecol Obstet, 1940. 70: p. 51-57.
- Schwartz, J., Hematoma of the umbilical cord.N Y State J Med, 1949. 49(13): p. 1575.
- Clare, N.M., R. Hayashi, and G. Khodr, Intrauterine death from umbilical cord hematoma.Arch Pathol Lab Med, 1979. 103(1): p. 46-7.
- Lupovitch, A. and T.S. McInerney, Hematoma of the umbilical cord: a dissecting aneurysm of the umbilical vein.Am J Obstet Gynecol, 1968. 102(6): p. 902-4.
- Gualandri, G., et al., Spontaneous umbilical cord hematoma: an unusual cause of fetal mortality: a report of 3 cases and review of the literature.Am J Forensic Med Pathol, 2008. 29(2): p. 185-90.
- Schreier, R. and S. Brown, Hematoma of the umbilical cord. Report of a case.Obstet Gynecol, 1962. 20: p. 798-800.
- Breen, J.L., H.L. Riva, and R.P. Hatch, Hematoma of the umbilical cord; a case report.Am J Obstet Gynecol, 1958. 76(6): p. 1288-90.
- Csecsei, K. and T. Kovacs, Spontaneous haematoma of the umbilical cord with a single umbilical artery.Eur J Obstet Gynecol Reprod Biol, 1996. 64(2): p. 231-3.
- Irani, P.K., Haematoma of the Umbilical Cord.Br Med J, 1964. 2(5422): p. 1436-7.
- Chou, S.Y., et al., Spontaneous umbilical cord hematoma diagnosed antenatally with ultrasonography.Acta Obstet Gynecol Scand, 2003. 82(11): p. 1056-7.
- Seoud, M., et al., Spontaneous umbilical cord hematoma: a rare cause of acute fetal distress.Am J Perinatol, 2001. 18(2): p. 99-102.
- Ruvinsky, E.D., et al., In utero diagnosis of umbilical cord hematoma by ultrasonography.Am J Obstet Gynecol, 1981. 140(7): p. 833-4.
- Sepulveda, W., et al., Fetal death due to umbilical cord hematoma: a rare complication of umbilical cord cyst.J Matern Fetal Neonatal Med, 2005. 18(6): p. 387-90.
- Sutro, W.H., et al., Prenatal observation of umbilical cord hematoma.AJR Am J Roentgenol, 1984. 142(4): p. 801-2.
- Farb, H.F., U. Rowlatt, and W.N. Spellacy, Spontaneous umbilical cord hematoma. A rare cause of fetal death.Minn Med, 1983. 66(5): p. 287-8.
- Ballas, S., S. Gitstein, and J. Kharasch, Fetal heart rate variation with umbilical haematoma.Postgrad Med J, 1985. 61(718): p. 753-5.
- Gregora, M.G. and J. Lai, Umbilical cord haematoma: a serious pregnancy complication.Aust N Z J Obstet Gynaecol, 1995. 35(2): p. 212-4.
- Roberts-Thomson, M.E., The hazards of umbilical cord haematoma.Med J Aust, 1973. 1(13): p. 648-50.
- Feldberg, D., et al., Hematoma of the umbilical cord with acute antepartum fetal distress. A case report.J Reprod Med, 1986. 31(1): p. 65-6.
- Ratten, G.J., Spontaneous haematoma of the umbilical cord.Aust N Z J Obstet Gynaecol, 1969. 9(2): p. 125-6.
- Moise, K.J., Jr., et al., Umbilical cord hematoma secondary to in utero intravascular transfusion for Rh isoimmunization.Fetal Ther, 1987. 2(2): p. 65-70.
- Keckstein, G., et al., Umbilical cord haematoma as a complication of intrauterine intravascular blood transfusion.Prenat Diagn, 1990. 10(1): p. 59-65.
- Jauniaux, E., et al., Hematoma of the umbilical cord secondary to cordocentesis for intrauterine fetal transfusion [letter; comment].Prenat Diagn, 1990. 10(7): p. 477-8.
- Chenard, E., A. Bastide, and W.D. Fraser, Umbilical cord hematoma following diagnostic funipuncture.Obstet Gynecol, 1990. 76(5 Pt 2): p. 994-6.
- Morin, L.R., et al., Sonography of umbilical cord hematoma following genetic amniocentesis.Acta Obstet Gynecol Scand, 1987. 66(7): p. 669-70.
- Jauniaux, E., et al., Pathologic aspects of the umbilical cord after percutaneous umbilical blood sampling.Obstet Gynecol, 1989. 73(2): p. 215-8.
- Babay, Z.A., et al., A case of varix dilatation of the umbilical vein and review of the literature.Fetal Diagn Ther, 1996.11(3): p. 221-3.
- White, S.P. and A. Kofinas, Prenatal diagnosis and management of umbilical vein varix of the intra-amniotic portion of the umbilical vein.J Ultrasound Med, 1994. 13(12): p. 992-4.
- Zachariah, M., S. Vyjayanthi, and S. Bell-Thomas, Umbilical vein varix thrombosis: a rare pathology.J Obstet Gynaecol, 2004. 24(5): p. 581.
- Viora, E., et al., Thrombosis of umbilical vein varix.Ultrasound Obstet Gynecol, 2002. 19(2): p. 212-3.
- Qureshi, F. and S.M. Jacques, Marked segmental thinning of the umbilical cord vessels.Arch Pathol Lab Med, 1994. 118(8): p. 826-30.
- Mulch, A.D., S.P. Stallings, and C.M. Salafia, Elevated maternal serum alpha-fetoprotein, umbilical vein varix, and mesenchymal dysplasia: are they related?Prenat Diagn, 2006. 26(8): p. 659-61.
- Schrocksnadel, H., et al., Thrombotic occlusion of an umbilical vein varix causing fetal death.Arch Gynecol Obstet, 1991. 248(4): p. 213-5.
- Vandevijver, N., et al., Aneurysm of the umbilical vein: case report and review of literature.Eur J Obstet Gynecol Reprod Biol, 2000. 89(1): p. 85-7.
- Heifetz, S.A., Thrombosis of the umbilical cord: analysis of 52 cases and literature review.Pediatr Pathol, 1988. 8(1): p. 37-54.
- Fortune, D. and A. Östör, Umbilical artery aneurysm.Am J Obstet Gynecol, 1978. 131: p. 339-40.
- Weber, M.A., et al., Third trimester intrauterine fetal death caused by arterial aneurysm of the umbilical cord.Pediatr Dev Pathol, 2007. 10(4): p. 305-8.
- Sentilhes, L., et al., Umbilical artery aneurysm in a severe growth-restricted fetus with normal karyotype.Prenat Diagn, 2007.27(11): p. 1059-61.
- Sepulveda, W., et al., Umbilical artery aneurysm: prenatal identification in three fetuses with trisomy 18.Ultrasound Obstet Gynecol, 2003. 21(3): p. 292-6.
- Berg, C., et al., Prenatal diagnosis of umbilical cord aneurysm in a fetus with trisomy 18.Ultrasound Obstet Gynecol, 2001. 17(1): p. 79-81.
- Siddiqi, T.A., et al., Umbilical artery aneurysm: prenatal diagnosis and management.Obstet Gynecol, 1992. 80(3 Pt 2): p. 530-3.
- Shen, O., et al., Prenatal diagnosis of umbilical artery aneurysm: a potentially lethal anomaly.J Ultrasound Med, 2007. 26(2): p. 251-3.
- Estroff, J.A. and B.R. Benacerraf, Fetal umbilical vein varix: sonographic appearance and postnatal outcome.J Ultrasound Med, 1992. 11(3): p. 69-73.
- Weissmann-Brenner, A., et al., Isolated fetal umbilical vein varix–prenatal sonographic diagnosis and suggested management.Prenat Diagn, 2009. 29(3): p. 229-33.
- Rahemtullah, A., et al., Outcome of pregnancy after prenatal diagnosis of umbilical vein varix.J Ultrasound Med, 2001. 20(2): p. 135-9.
- Allen, S.L., et al., Thrombosing umbilical vein varix.J Ultrasound Med, 1998. 17(3): p. 189-92.
- Ami, M.B., Y. Perlitz, and M. Matilsky, Prenatal sonographic diagnosis of persistent right umbilical vein with varix.J Clin Ultrasound, 1999. 27(5): p. 273-5.
- Corkill, T.F., The infant’s vulnerable life-line.Aust N Z J Obstet Gynæcol, 1961. 1: p. 154-60.
- Cites refs I need to get.
- Davignon, J., R.R. Lorenz, and J.T. Shepherd, Response of human umbilical artery to changes in transmural pressure.Am J Physiol, 1965. 209: p. 51-59.
- Bendon, R.W., S.P. Brown, and M.G. Ross, In vitro umbilical cord wrapping and torsion: possible cause of umbilical blood flow occlusion.J Matern Fetal Neonatal Med, 2014. 27(14): p. 1462-4.
- Perlitz, Y., I. Ben-Shlomo, and M. Ben-Ami, Acute polyhydramnios in term pregnancy may be caused by multiple nuchal cord loops.Ultrasound Obstet Gynecol, 2010. 35(2): p. 253-4.
- D’Antona, D., et al., Effect of nuchal cord on fetal cerebral haemodynamics and oxygenation measured by near infrared spectroscopy during labour.Eur J Obstet Gynecol Reprod Biol, 1995. 59(2): p. 205-9.
- Plasencia, W., et al., Influence of nuchal cord on ductus venosus assessment at 11 to 13 + 6 weeks’ gestation.Ultrasound Obstet Gynecol, 2010. 35(3): p. 263-6.
- Carey, J.C. and W.F. Rayburn, Nuchal cord encirclements and risk of stillbirth.Int J Gynaecol Obstet, 2000. 69(2): p. 173-4.
- Wang, G., K.E. Bove, and J. Stanek, Pathological evidence of prolonged umbilical cord encirclement as a cause of fetal death.Am J Perinatol, 1998. 15(10): p. 585-8.
- Dursun, P., et al., Nuchal cord type B associated with an excessively long umbilical cord as a cause of stillbirth: a case report.Clin Exp Obstet Gynecol, 2004. 31(2): p. 158-9.
- Verdel, M.J. and N. Exalto, Tight nuchal coiling of the umbilical cord causing fetal death.J Clin Ultrasound, 1994. 22(1): p. 64-6.
- Hamilton, J., Cord around the neck.Clin J, 1947. 76(3): p. 103.
- Rayburn, W.F., A. Beynen, and D.L. Brinkman, Umbilical cord length and intrapartum complications.Obstet Gynecol, 1981. 57(4): p. 450-2.
- Baergen, R.N., et al., Morbidity, mortality, and placental pathology in excessively long umbilical cords: retrospective study.Pediatr Dev Pathol, 2001. 4(2): p. 144-53.
- Berg, T.G. and W.F. Rayburn, Umbilical cord length and acid-base balance at delivery.J Reprod Med, 1995. 40(1): p. 9-12.
- Naeye, R., Umbilical cord length: clinical significance.J Pediatr, 1985. 107: p. 278-281.
- Mills, J.L., E.E. Harley, and A.C. Moessinger, Standards for measuring umbilical cord length.Placenta, 1983. 4(4): p. 423-6.
- Sarwono, E., et al., Umbilical cord length and intra uterine wellbeing.Paediatr Indones, 1991. 31(5-6): p. 136-40.
- Malpas, P., Length of the Human Umbilical Cord at Term.Br Med J, 1964. 1(5384): p. 673-4.
- Adinma, J.I., The umbilical cord: a study of 1,000 consecutive deliveries.Int J Fertil Menopausal Stud, 1993. 38(3): p. 175-9.
- Miller, M., M. Higginbottom, and D. Smith, Short umbilical cord: Its origin and relevance.Pediatr, 1981. 67: p. 618-21.
- Miller, M.E., M.C. Jones, and D.W. Smith, Tension: the basis of umbilical cord growth.J Pediatr, 1982. 101(5): p. 844.
- Smit, L.M. and P.G. Barth, Arthrogryposis multiplex congenita due to congenital myasthenia.Dev Med Child Neurol, 1980. 22(3): p. 371-4.
- Thakur, S., L. Pal, and S.R. Phadke, Lethal arthrogryposis with icthyosis: overlap with Neu-Laxova syndrome, restrictive dermopathy and harlequin fetus.Clin Dysmorphol, 2004. 13(2): p. 117-9.
- Jago, R.H., Arthrogryposis following treatment of maternal tetanus with muscle relaxants.Arch Dis Child, 1970. 45(240): p. 277-9.
- Izumi, K., et al., Umbilical cord length in urinary tract abnormalities associated with oligohydramnios: evidence regarding developmental pathogenesis.Fetal Pediatr Pathol, 2006. 25(5): p. 233-40.
- Snider, W., Placental casebook.J Perinatol, 1997. 17(4): p. 327-9.
- Moessinger, A.C., et al., Umbilical cord length as an index of fetal activity: experimental study and clinical implications.Pediatr Res, 1982. 16(2): p. 109-12.
- Sobolewski, K., et al., Wharton’s jelly as a reservoir of peptide growth factors.Placenta, 2005. 26(10): p. 747-52.
- Discher, D.E., P. Janmey, and Y.L. Wang, Tissue cells feel and respond to the stiffness of their substrate.Science, 2005. 310(5751): p. 1139-43.
- Chasnoff, I.J. and M.A. Fletcher, True knot of the umbilical cord.Am J Obstet Gynecol, 1977. 127: p. 425-7.
- Naeye, R.L., Disorders of the Placenta, Fetus and Neonate, Diagnosis and Clinical Significance. 1992, St. Louis: Mosby Year Book.
- McLennan, H., et al., Umbilical cord knots and encirclements.Austral New Zealand J Obstet Gynecol, 1988. 28: p. 116-119.
- Blickstein, I., Z. Shoham-Schwartz, and M. Lancet, Predisposing factors in the formation of true knots of the umbilical cord- analysis of morphometric and perinatal data.Int J Gynecol Obstet, 1987. 25(1288-1290).
- Spellacy, W.N., H. Graven, and R.O. Fisch, The umbilical cord complications of true knots, nuchal coils and cords around the body.Am J Obstet Gynecol, 1966. 94: p. 1136-42.
- Ragucci, N. and C. Morandi, Le distocia del funicolo ombelicale (contributo clinico-statistico).Minerva Ginecologica, 1969. 21: p. 653-655.
- Scheffel, T. and D. Langanke, Die Nabelschnurkomplikationen an der Universitäts-Frauenklinik von 1955 bis 1967.Zentralbl Gynäkol, 1970. 92: p. 429-434.
- Sopracordevole, F. and M.G. Perissinotto, Il nodo vero di funicolo: implicazoni cliniche.Minerva Ginecologia, 1991. 43: p. 109-113.
- Lundgren, A.T. and W.A. Boice, True knotting of the umbilical cord.Illinois Med J, 1939. 76: p. 451-458.
- Sornes, T., Umbilical cord knots.Acta Obstet Gynecol Scand, 2000. 79(3): p. 157-9.
- Hartge, R., Über das Vorkommen von Nabelschnurknoten.Geburstshilfe und Frauenheilkunde, 1979. 32: p. 976-980.
- Javert, C.T. and B. Barton, Congenital and acquired lesions of the umbilical cord and spontaneous abortion.Am J Obstet Gynecol, 1952. 63: p. 1065-1077.
- Collins, J.C., R.J. Muller, and C.L. Collins, Prenatal observation of umbilical cord abnormalities: a triple knot and torsion of the umbilical cord.Am J Obstet Gynecol, 1993. 169(1): p. 102-4.
- Hershkovitz, R., et al., Risk factors associated with true knots of the umbilical cord.Eur J Obstet Gynecol Reprod Biol, 2001. 98(1): p. 36-9.
- Gembruch, U. and A.A. Baschat, True knot of the umbilical cord: transient constrictive effect to umbilical venous blood flow demonstrated by Doppler sonography.Ultrasound Obstet Gynecol, 1996. 8(1): p. 53-6.
- Browne, F.J., On the abnormalities of the umbilical cord which may cause antenatal death.J Obstet Gynæcol Brit Emp, 1925. 32: p. 17-48.
- diTerlizzi, G. and G.F. Rossi, Studio clinico-statistico sulle anomalie del funicolo.Annali di Obstet Ginecol, 1955. 77: p. 459-474.
- Hennessy, J.P., True knots of the umbilical cord.Am J Obstet Gynecol, 1944. 48: p. 528-536.
- Tuxen, A.J., et al., Factors affecting umbilical venous perfusion during experimental cord knotting.Placenta, 2005. 26(10): p. 753-7.
- Benirschke, K. and P. Kaufmann, Pathology of the Human Placenta. Third ed. ed. 1995, New York: Springer-Verlag.
- Maher, J.T. and J.A. Conti, A comparison of umbilical cord blood gas values between newborns with and without true knots.Obstet Gynecol, 1996. 88(5): p. 863-6.
- Airas, U. and S. Heinonen, Clinical significance of true umbilical knots: a population-based analysis.Am J Perinatol, 2002. 19(3): p. 127-32.
- Ghosh, A., et al., Fetal loss from umbilical cord abnormalities- a difficult case for prevention.Europ J Obstet Gynec Reprod Biol, 1984. 18: p. 183-98.
- Goldstein, I., I.E. Timor-Trisch, and I. Zaidise, Sinusoidal pattern together with signs of moderate fetal hypoxia associated with a true knot of cord.Europ J Obstet Gynecol Reprod Biol, 1981. 11: p. 221-225.
- Abuhamad, A., Three-dimensional ultrasound with color Doppler imaging of an umbilical cord true knot.Ultrasound Obstet Gynecol, 2014. 43(3): p. 360.
- Vasilj, O., et al., Do we sometimes see too much? Prenatal diagnosis of a true umbilical cord knot.Eur J Obstet Gynecol Reprod Biol, 2015. 187: p. 73-4.
- Rodriguez, N., et al., Three-dimensional high-definition flow imaging in prenatal diagnosis of a true umbilical cord knot.Ultrasound Obstet Gynecol, 2012. 39(2): p. 245-6.
- Hasbun, J., J.L. Alcalde, and W. Sepulveda, Three-dimensional power Doppler sonography in the prenatal diagnosis of a true knot of the umbilical cord: value and limitations.J Ultrasound Med, 2007. 26(9): p. 1215-20.
- Ramon, Y.C.C.L. and R.O. Martinez, Prenatal diagnosis of true knot of the umbilical cord.Ultrasound Obstet Gynecol, 2004. 23(1): p. 99-100.
- Abuhamad, A., et al., Umbilical artery Doppler waveform notching: is it a marker for cord and placental abnormalities?J Ultrasound Med, 2002. 21(8): p. 857-60.
- Stempel, L.E., Beyond the pretty pictures: giving obstetricians just enough (umbilical) cord to hang themselves.Am J Obstet Gynecol, 2006. 195(4): p. 888-90.
- Gupta, I., V.F. Hillier, and J.M. Edwards, Multiple vascular profiles in the umbilical cord; an indication of maternal smoking habits and intrauterine distress.Placenta, 1993. 14(1): p. 117-23.
- Lacro, R.V., K.L. Jones, and K. Benirschke, The umbilical cord twist: origin, direction, and relevance.Am J Obstet Gynecol, 1987. 157(4 Pt 1): p. 833-8.
- Strong, T.H., Jr., H.J. Finberg, and J.H. Mattox, Antepartum diagnosis of noncoiled umbilical cords.Am J Obstet Gynecol, 1994. 170(6): p. 1729-31; discussion 1731-3.
- Machin, G.A., J. Ackerman, and E. Gilbert-Barness, Abnormal umbilical cord coiling is associated with adverse perinatal outcomes.Pediatr Dev Pathol, 2000. 3(5): p. 462-71.
- Collins, J., Prenatal observation of umbilical cord torsion with subsequent premature labor and delivery of a 31-week infant with mild nonimmune hydrops.Am J Obstet Gynecol, 1995. 172: p. 1048.
- Hathout, H., The vascular pattern and mode of insertion of the umbilical cord in abortion material.J Obstet Gynaecol: p. 961-4.
- Uyanwah-Akpom, P. and H. Fox, The clinical significance of marginal and velamentous insertion of the cord.Br J Obstet Gynaecol, 1977. 84(12): p. 941-3.
- Heinonen, S., et al., Perinatal diagnostic evaluation of velamentous umbilical cord insertion: clinical, Doppler, and ultrasonic findings.Obstet Gynecol, 1996. 87(1): p. 112-7.
- Eddleman, K.A., et al., Clinical significance and sonographic diagnosis of velamentous umbilical cord insertion.Am J Perinatol, 1992. 9(2): p. 123-6.
- Yao, A.C., J. Lind, and T. Lu, Closure of the human umbilical artery: a physiological demonstration of Burton’s theory.Eur J Obstet Gynecol Reprod Biol, 1977. 7(6): p. 365-8.
- McCallum, W.D., Thermodilution measurement of human umbilical blood flow at delivery.Am J Obstet Gynecol, 1977. 127(5): p. 491-6.
- McCallum, W.D. and R.M. McAreavey, Human umbilical vessel diameters and bloodgas tensions in the first two minutes after delivery.Gynecol Invest, 1976. 7(4): p. 201-12.
- Boura, A.L. and W.A. Walters, Autacoids and the control of vascular tone in the human umbilical-placental circulation.Placenta, 1991. 12(5): p. 453-77.
- Meirelles, R.S., et al., In vitro contractility of human umbilical artery.Europ J Obstet Gyn Reprod Biol, 1977. 7: p. 219-226.
- Tiritilli, A., 5-hydroxytryptamine induces vasoconstriction of the human umbilical artery: effects of hypoxia and nicorandil.Gynecol Obstet Invest, 2000. 50(2): p. 77-83.
- White, R.P., Pharmacodynamic study of maturation and closure of human umbilical arteries.Am J Obstet Gynecol, 1989. 160(1): p. 229-37.
- Cruz, M.A., et al., Effect of histamine on human placental chorionic veins: interaction with serotonin.Pharmacology, 1991. 42(2): p. 86-90.
- Gebrane-Younes, J., N.M. Hoang, and L. Orcel, Ultrastructure of human umbilical vessels: a possible role in amniotic fluid formation?Placenta, 1986. 7(2): p. 173-85.
- Bergh, C.M., et al., Impaired cyclic nucleotide-dependent vasorelaxation in human umbilical artery smooth muscle.Am J Physiol, 1995. 268(1 Pt 2): p. H202-12.
- Brophy, C.M., et al., Heat shock protein expression in umbilical artery smooth muscle.J Reprod Fertil, 1998. 114(2): p. 351-5.
- Singh, V., S. Khanum, and M. Singh, Umbilical cord lesions in early intrauterine fetal demise.Arch Pathol Lab Med, 2003.127(7): p. 850-3.
- Collins, J.H., Two cases of multiple umbilical cord abnormalities resulting in stillbirth: prenatal observation with ultrasonography and fetal heart rates.Am J Obstet Gynecol, 1993. 168(1 Pt 1): p. 125-8.
- Parast, M.M., C.P. Crum, and T.K. Boyd, Placental histologic criteria for umbilical blood flow restriction in unexplained stillbirth.Hum Pathol, 2008. 39(6): p. 948-53.