Introduction:
Obstetricians are very familiar with the gross anatomy of the cervix in pregnancy. They push on it to determine softness and measure how much it has effaced (shortened) and dilated with labor. They measure its length and inspect its internal os by ultrasound, usually through the vagina. They suture it shut for cervical incompetence, and dilate it with laminaria for labor induction. Pathologists are less familiar with the gravid cervix, which is examined only when a catastrophe results in the removal of a pregnant uterus, or the death of the mother. Very few studies have biopsied the pregnant human cervix to understand the microscopic anatomical changes of labor. This section will argue that the cervix is potentially the initiator of preterm labor and chorioamnionitis, and as such merits more study. The topic of chorioamnionitis is presented in more detail in Section 13B, but some background is presented as follows.
The cervical barrier to chorioamnionitis
The cervix is designed to keep the normal resident vaginal flora out of the uterus. Ideally, it would act as a tight seal, to keep all microorganisms outside. The seal, even in the non-pregnant patient, is however less than perfect. Sperm, which can be viewed as microorganisms, must get past the cervix for fertilization. Some microorganisms have developed ways to bypass the cervical defenses and can penetrate through the cervix such as Neisseria gonorrheaand Chlamydia, microorganisms that produce sexually transmitted diseases. To complicate matters, the cervix must also be able to open to allow menstrual discharge to exit the uterus. At the extreme, the cervix dilates to allow the contents of the pregnant uterus to exit.
The cervix does present a substantial barrier to vaginal microorganisms, even if it is not an absolute barrier. There are at least three components of this barrier. The first is geometrical. Pasteur created flasks with a swan neck that kept environmental bacteria from dropping into and contaminating his bacterial cultures. The cervix lacks this option, but does use a relatively long length and interior overlapping corrugations to create a similar barrier. On a microscopic level this provides a long arduous terrain of deep mountains and valleys that bacteria must cross to enter the uterus. The second barrier is a mucus plug of the cervical lumen, much like the cotton plugs that keep environmental bacteria out of culture tubes. Dry cotton plugs were not an evolutionary option, but the glands embedded in the lumen of the cervix secrete dense, sticky mucus. The third barrier is composed of inflammatory cells that act as scouts to attack any microorganisms that enter the cervical tissue. These cells can ingest microorganisms, and through various biochemical pathways, mount an attack against them by recruiting more cells and other bio-molecules that are common to the inflammatory pathways of the body.
The vaginal flora is a mix of microorganisms. Each species has its preferred pH ranges and chemical environments, and they compete with each other for territory and nutrients. Some of the microorganisms travel randomly by Brownian motion, but some are more motile and can respond to a directional signal such as concentration gradient of nutrient. Despite the presence of vast numbers of microorganisms existing as a community in the vagina, few ever reach the uterine cavity via the cervix under normal circumstances.
What are the circumstances in which organisms do ascend the cervix? Unsurprisingly, labor is one such circumstance, the opening of the cervix for labor expels the mucus plug and shortens the cervical length and widens the opening of the lumen. There are numerous studies that have shown a direct correlation of duration of labor and the risk of ascending infection into the uterus (chorioamnionitis) 1,2. This was expected and logical. Before germ theory was established, the obstetrician Semmelwieis proposed hand washing with chlorinated lime to prevent childbed fever.
In labor, infection ascending from vagina to uterus may produce clinical chorioamnionitis with maternal fever, elevated white blood cell count and uterine tenderness, all expected symptoms of an infected uterus. Yet many patients have no symptoms with labor, but still have unequivocal acute inflammation in their placenta and fetal membranes that can be seen by microscopic examination. A surprise discovery was that ascending infection, as detected by the presence of acute inflammatory cells in the fetal membranes, is significantly more frequent in women with preterm labor. Most of these women do not have clinical symptoms of infection; instead they have so called clinically silent infection. The importance of this association is that finding chorioamnionitis in preterm labor is analogous to finding it in prolonged labor. An implication is that the cervical barrier may have been breached in these patients for a period of time, perhaps exceeding 24 hours prior to the onset of active preterm labor.
Cervical labor
If the function of staying closed is critical to the cervix, so is the function of opening when needed. There is ample evidence that the cervix in the pregnant patient may begin the process of opening prior to active labor. It is not uncommon for near term or term pregnant women to have cervical shortening and dilatation prior to the onset of labor. Before labor, cervical changes are evaluated to predict the success of the induction of labor. Bishop proposed a semi-quantitative score of cervical changes based on cervical palpation to predict success in inducing labor. This score has also demonstrated a reasonable predictive value as a test for impending preterm labor3. Individual components of the score have been evaluated as independent predictors and cervical softening and dilatation were significant4-8. The score as a predictor of preterm labor is limited by its subjective technique, and by the need to use intermittent vaginal examinations.
As implied by the Bishop score, the earliest change in the opening of the cervix is the softening. The shortening and dilatation appear to be passive either from the weight of the amniotic contents pushing down or uterine contractions pulling upward or both.
For simplicity, the cervical changes of softening, shortening, and dilatation, can be lumped as cervical labor, or more commonly, cervical ripening. I prefer the former term. A potentially fruitful hypothesis is that cervical labor is permissive for chorioamnionitis, initiates preterm labor, and causes premature rupture of membranes and in some incidences, cervical incompetence. If this hypothesis is true, then preventing these complications might be aided by understanding the mechanisms and control of cervical labor.
By definition premature rupture of membranes (PROM) occurs before the onset of labor. Both premature rupture and normal rupture occur over the cervical os in almost all cases. There are two plausible mechanisms for PROM. Opening of the internal cervical os would cause membrane to bulge through creating the equivalent of an aneurysm of the membrane sac with a higher probability of rupture at a given pressure. A second mechanism is the weakening of adjacent membranes by activated cervical neutrophils. Both of these mechanisms could result from cervical ripening prior to uterine contractions. Thus, we hypothesize that preterm premature rupture of membranes is a consequence of early cervical ripening before the onset of uterine contractions. In some cases, the fluid pressure against the membranes must not be strong enough to hold them in position, and instead the amnion sac begins to deliver through a relatively narrow opening in the cervix, so called hourglass membranes. The decidua itself is not designed to hold the membranes against the uterine wall, but it designed to shed the membranes if there is lateral pressure. In both PROM and hour glass membranes, opening of the cervix creates conditions that clinically often result in preterm delivery of the infant.
Incompetent cervix is defined as premature delivery of an infant through a dilated cervix without onset of contractions. Those patients with widely dilated cervices on presentation may be part of a spectrum of premature cervical labor with incompetent cervix at one extreme9. IN some cases of incompetent cervix, the women have anatomic abnormalities that weaken the cervix. Even with an anatomically normal cervix, advanced cervical ripening before uterine contractions is a common denominator in many preterm deliveries, including some that present as incompetent cervix.
In many physiologic systems, entry into the lumen of an organ is controlled by an active muscle that circles the opening and can constrict or relax as needed, such as the anal or urethral sphincter. The cervix uses a very different mechanism. The cervix is primarily a connective tissue structure10. Its small amount of superficial muscle does not appear to play a direct role in cervical ripening. The lumen is kept closed by a rigid wall that presses in on a narrow lumen that has interlocking baffles. The walls are kept rigid with struts of collagen and a matrix of glycoproteins and water. Early studies assumed that the softening of the cervix would be via collagenolysis, equivalent to breaking down the supporting struts of the structure. Indeed measurements showed that with dilatation of the cervix in labor the concentration of collagen decreased. However, further study showed that the decreased concentration was not because of any destruction of collagen, but because the matrix was being broken down, drawing in more water, and the collagen beams were collapsing but the collagen protein was intact. The cervix becomes softer due to the gel changes in the glyco-amino-glycans of the matrix 11,12Specifically increases in hyaluronic acid and dermatan sulfate have been measured. The collagen fibers morphologically becomes less ordered 13. These changes appear to be true in species as diverse as the human and the rat and rabbit. This solution to maintaining cervical closure likely requires much less energy than a muscular sphincter would. The glycoprotein matrix has the advantage that it can be biochemically deconstructed and reconstructed more quickly than collagen can be destroyed and resynthesized during labor.
How is cervical labor initiated?
If cervical labor is important in producing complications that lead to preterm delivery, then it becomes important to know what factors that initiate cervical labor. There are multiple means to trigger cervical labor in the human including the administration of oxytocin, progesterone, relaxin, interleukins, prostaglandin E2 and E1. Endocervical dilatation with a balloon or laminaria also results in cervical ripening 14-23. In an experimental bath, prostaglandin can soften the cervix in 5 to 12 minutes. There is also clinical and experimental evidence for the participation of neutrophils 13,24-26. Microorganisms cannot only induce inflammation to produce interleukins and prostaglandins, but can directly produce prostaglandins27. Cervical labor in the sheep can be induced with multiple different manipulations, including dexamethasone, estrogen, oophorectomy, prostaglandin, and oxytocin. As used clinically in humans, cervical labor can be induced with a prostaglandin E2 gel 28. Cervical labor can be experimentally separated from uterine contractions. In one report this was done literally with surgical separation in dexamethasone induced labor. However the separation of cervical and uterine labor can be produced biochemically. Cervical ripening can be induced with prostaglandin, and uterine contractions can be inhibited with progesterone29. In this ovine model, the cervix returned to a competent state following removal of the prostaglandin stimulus. Cervical labor can even be induced in the non-pregnant ewe with oxytocin administration 30.
The relationship of cervical to uterine labor
There is a complex interaction of cervical ripening with uterine contractions. Cervical labor is normally linked to effective uterine contractions for a successful term labor. In the usual explanation of labor, the uterine contractions draw the cervix upward, widening around the presenting part, usually the fetal head, until the infant can be expelled. The observation of cervical dilatation is often considered the hallmark of real labor and the onset of effective uterine contractions. One experiment demonstrated that stretch of cervical tissue released prostaglandins31. Thus, the stretch produced by uterine contractions could produce prostaglandins that initiate cervical labor. Certainly the uterine contractions per se do not simply force open the cervix, but are part of a coordinated change within the cervix. Prior to delivery the cervical compliance changes dramatically in sheep. Nevertheless, uterine contractions for the laboring ewe rarely exceed 20 mm Hg, suggesting that it is the physical properties of the cervix which change dramatically at parturition rather than uterine pressure that causes these changes.
The opposite, namely that cervical labor could induce uterine labor, is suggested by the clinical induction of human uterine labor with cervical prostaglandin administration. This is not a systemic effect but a local one. The mechanisms are unclear. One possibility is that the softening of the cervix would allow the passive weight of the intrauterine contents to stretch the myometrial fibers in the cervix. Stretch of myometrial fibers can induce the cellular changes of labor such as inducing myometrial gap junctions and oxytocin receptor32. These changes could be propagated by cell processes to the lower uterine segment myometrial cells and upward. The mechanism might involve systemic signals as well.
If uterine contractions can induce the cervical changes needed for delivery of the infant, from an evolutionary perspective, are there any potential advantages for the independent initiation of a cervical labor? The ability to experimentally induce cervical labor could simply be an artifact of triggering the intermediate mechanisms that follow from uterine contractions. Pathological cervical labor would then be the result of subtle uterine labor, rather than primary induction of cervical labor. I am not sure that hypothesis has been excluded clinically, but there is a theoretical basis for postulating independent cervical labor, which would make an exclusive uterine initiator less likely.
This theoretical basis follows from a hypothesis presented by Sarah Blaffer Hrdy in her book “Mother Nature A History of Mothers, Infants and Natural Selection”33. Her basic hypothesis is that there are maternal mechanisms in nature that will enable the survival of one living offspring by eliminating a current pregnancy, or sometimes even a newborn. This would be advantageous for example with maternal illness such as infection that would compromise her ability to survive to carry the pregnancy to term. There is evidence that sources of potential systemic inflammatory mediators such as dental inflammation, urinary tract infections, and even appendicitis can induce labor. The mechanism of labor with infection would reasonably have different initiation than that signaled by fetal maturity. Dr. Hrdy also presents evidence that other stresses such as food shortages or loss of status can lead to fetal loss that promotes survival of a living infant. While her hypothesis does not require a specific pathway for environmental stress to result in fetal loss, cervical labor could be one of the mechanisms. If changes in the cervix led to the ascent of benign maternal vaginal flora into the uterus, then an inflammatory response would initiate labor and prematurely expel the fetus. The evidence from mice to monkeys does not suggest that human mothers can consciously effect cervical change, but it remains possible that such preservation mechanisms causing fetal loss persist in humans. Discovering mechanisms that initiate cervical labor might then be useful in developing strategies to prevent preterm delivery.
Pathological examination of the cervix
There are very few anatomical studies of the human gravid cervix. I think that more attention to the cervix at the time of gravid hysterectomy might yield some insight into cervical pathology, particularly the role of inflammation and changes in structure (Fig 1-3).
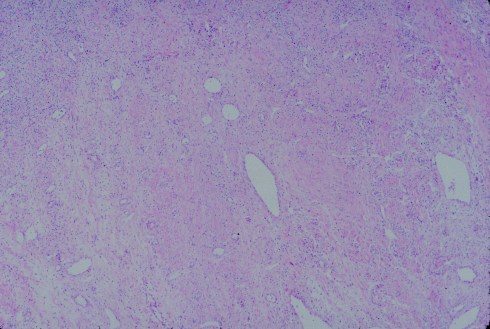
Fig 1: This is a low magnification of a gravid cervix with some myometrial cells to the right, then a denser fibrous area in the middle, and finally an area with loose stroma and many inflammatory cells. (H&E, 4x)

Fig 2: At higher magnification the inflammatory cells appear to be predominantly mononuclear. (H&E 25x)
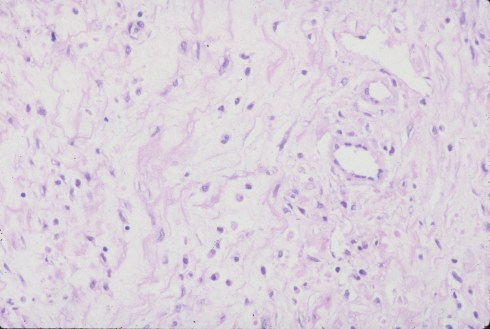
Fig 3: In another area a more edematous, less cellular matrix is evident. (H&E, 25x)
Biopsies of the sheep cervix with induced cervical labor may provide insight in interpreting potential studies of human gravid cervix after preterm labor, etc.
To understand the initiation of cervical labor, and the intermediate pathways of matrix breakdown, there needs to be a quantitative understanding of how communication occurs on the microscopic level. Signals can be produced locally or systemically and propagate via the blood flow, transfer cell-to-cell or diffuse through the microscopic flows and barriers between cells. Signals may have very different half lives and modes of action for example membrane effects versus nuclear binding. Some cells like inflammatory cells are mobile, others may have extensive processes such as myometrial cells. Current histological tools are limited to histochemical stains that rely on gradients of a chemical property or to immuno-stains that yield a qualitative identification of a single antigen per microscope slide. Both rely on a small sample taken at a single point in time. We need to develop better techniques to evaluate the cervical changes.
Conclusion
The uterine cervix is a key organ in pregnancy. Cervical changes of labor are controlled by changes in the matrix where breakdown of glycoproteins can cause rapid softening. While a rigid, closed cervix is needed to prevent ascent of vaginal organisms into the intrauterine cavity, cervical labor must also be able to open in response to signals of uterine labor. The process of cervical labor may also be initiated independently of uterine labor and this may lead to chorioamnionitis, preterm labor and premature rupture of membranes. For this reason, further study of cervical labor mechanisms has potential clinical utility. Fortunately, for clinical care even without a complete understanding of cervical labor, we can ask what clinical events precede the onset of preterm labor, or preterm rupture of membranes in an effort to prevent these complications.
References
- Rouse DJ, Weiner SJ, Bloom SL, et al. Second-stage labor duration in nulliparous women: relationship to maternal and perinatal outcomes. Am J Obstet Gynecol 2009;201:357 e1-7.
- Reid DE, Christian CD, eds. Controvery in Obstetrics and Gynaecology. second ed. Philadelphia: W.B. Saunders Company; 1974.
- EH B. Pelvic scoring for elective induction. Obstet Gynecol 1964;24:266-8.
- Schreyer P, Caspi E, Natan B, Weiraub Z. The predictive value of fetal breathing movement and Bishop score in the diagnosis of “true” preterm labor. Am J Obstet Gynecol 1989;161:886-9.
- Newman R, Godsey R, Ellings J, Campbell B, Eller D, Miller M. Quantification of Cervical Change – Relationship to Preterm Delivery in the Multifetal Gestation. American Journal of Obstetrics and Gynecology165: 2 (AUG 1991);264-271.
- RP L, CH C, SF B, SR M. Randomized prospective trial comparing ultrasonography and pelvic examination for preterm labor surveillance. Am J Obstet Gynecol 1990;162:1603-10.
- B B, X LC, M K, C C, G B, C S. Prediction of preterm delivery: Is it substantially improved by routine vaginal examinations. Am J Obstet Gynecol 1990;162:1042-8.
- RL C, RL G, RO D, et al. Warning symptoms; uterine contractions; and cervical examinationfindings in women at risk of preterm delivery. Am J Obstet Gynecol 1990;162:748-54.
- Iams J, Johnson F, Sonek J, Sachs L, Gebauer C, Samuels P. Cervical competence as a continuum: A study of ultrasonographic cervical length and obstetric performance. Am J Obstet Gynecol 1995;172:1097-106.
- DN D, JC B, JW R. Connective tissue changes incident to cervical effacement. Am J Obstet Gynecol 1960;80:939-45.
- AM. G. Biochemical basis of cervical maturation. In: Huszar G, ed. The physiology and biochemistry of the uterus in pregnancy and labor. Boca Raton, Florida: CRC Press Inc.; 1986:262-80.
- R K, JF W. Effects of hormonal perturbations on the small dermatan sulfate proteoglycan and mechanical properties of the uterine cervix of late pregnant rats. Conn Tiss Res 1991;138:273-81.
- Junqueira L, Zugaib M, Montes G, Toledo O, Kriszran R, Shigihara K. Morphologic and histochemical evidence for the occurrence of collagenolysis and for the role of neutrophilic polymorphonuclear leukocytes during cervical dilatation. Am J Obstet Gynecol 1980;138:273-81.
- AA C. Prostaglandins and biological control of cervical function. Aust NZ Obstet Gynecol 1994;34:347-351.
- JT C, K U. Reduction of the stretch modulus of human cervical tissue by prostaglandin E2. Am J Obstet Gynecol 1976;126:218-223.
- JT C, K U. The stretch modulus of human cervical tissue in spontaneous; oxytocin-induced; and prostaglandin E2-induced labor. Am J Obstet Gynecol 1979;133:11-14.
- E EM, N K, A H, K M, K S, T T. Interleukin-8 induces cervical ripening in rabbits. Am J Obstet Gynecol 1994;171:77-83.
- EF M, KG PJ, JR DJ, JD B, SP C, JC M. Cervical ripening before medical induction of labor: A comparison of prostaglandin E2; estradiol; and oxytocin. Am J Obstet Gynecol 1995;172:1702-8.
- DA W, MM J, A R, TM G, RH P. A comparison of misoprostol and prostaglandin E2 gel for preinduction cervical ripening and labor induction. Am J Obstet Gynecol 1995;172:1804-10.
- DA W, A R, MM J, TM G, RH P. Misoprosotol: An effective agent for cervical ripening and labor induction. Am J Obstet Gynecol 1995;172:1811-6.
- RJ W, MB Od-B, OD S. Hormonal control of the cervix in pregnant gilts. IV. Relaxin promotes changes in the histological characteristics of the cervix that are associated with cervical softening during late pregnancy in gilts. Endocrinol 1993;133:121-28.
- RD SO, GT C. Preinduction cervical ripening: A comparison of intercervical prostaglandin E2 gel versus Foley catheter. Am J Obstet Gynecol 1995;172:687-90.
- SJ. S. Endocrine regulation of cervical functions during pregnancy and labor. In: Huszar G, ed. The physiology and biochemistryof the uterus in pregnancy and labor. Boca Raton, FL: CRC Press Inc.; 1986:281-95.
- K O, Y Y, Y H, H H, A Y. Nitroblue tetrazolium reduction by leukocytes in the cervix of pregnant women. Am J Obstet Gynecol 1988;159:417-20.
- Osmers, W R, BC A-G, et al. Origin of cervical collagenase during parturition. Am J Obstet Gynecol 1992;166:1455-60.
- Owiny J, Gilbert R, Wahl C, Nathanielsz P. Leukocytic invasion of the ovine cervix at parturition. J Soc Gynecol Invest 1995;2:593-6.
- Bejar R, Curbelo V, Davis C, Gluck L. Premature labor. II Bacterial sources of phospholipase. Obstet Gynecol 1981;57:479-82.
- Stys S, Dresser B, Otte T, Clark K. Effect of prostaglandin E2 on cervical compliance in pregnant ewes. Am J Obstet Gynecol 1981;140:415-19.
- Stys S, Clark K, Clewell W, G M. Hormonal Effects on Cervical Compliance in Sheep. In: Naftolin F, Stubblefield P, eds. Dilatation of the Uterine Cervix. New York: Raven Press; 1980:147-56.
- Khalifa R, Sayre B, Lewis G. Exogenous oxytocin dilates the cervix in ewes. J Anim Sci 1992;70:38-42.
- K H, N C. Synthesis of prostaglandins by the human uterine cervix in vitro during passive mechanical stretch. J Pharm Pharmacol 1982;34:262-63.
- Wathes DC, Porter DG. Effect of uterine distension and oestrogen treatment on gap junction formation in the myometrium of the rat. J Reprod Fertil 1982;65:497-505.
- Blaffer Hrty S. Mother Nature A History of Mothers, Infants, and Natural Selection. New York: Pantheon Books; 1999.