The decidua
The placenta itself does not separate from the uterus, but rather the endometrial lining of the uterus splits carrying the placenta with the superficial portion. The marked histological changes in the endometrium are referred to as decidualization, a use of the term analogous with the shedding of decidual leaves from trees in the fall. The portion under the placenta is termed the decidua basalis, and the superficial portion of this remains attached to the delivered placenta. If there is no decidual layer layer, as is the case with abdominal pregnancy when the placental is implanted directly on abdominal organs, there is no separation, and the placenta may be left in situ to avoid hemorrhage.
The separation and expulsion of the placenta has been named the third stage of labor. As a resident, I was warned against pulling the cord to hasten the placental expulsion. There is some variable delay in normal deliveries that can seem prolonged. This delay was recognized in the middle ages as indicated from this quote from a modern translation of the Trotula: “There are some women to whom the afterbirth remains inside after birth, to whom we give aid for its expulsion thus. We extract the juice of a leek and mix it with pennyroyal oil or musk oil or juice of borage, and let us give it to drink, and immediately {the afterbirth} will be brought out perhaps because she will vomit and from the effort of vomiting it will come out. Nevertheless, the juice itself has such power that it is sufficient for expulsion.(1)” I could find no controlled trials. There is some variation in management of the third stage of labor, which is beyond the scope of this book. A true inability of the placenta to separate (placenta accreta) is an important obstetrical complication.
The mechanics of normal placental separation
The endometrium is split and shed during normal menstruation by a mechanism of hormonally controlled contraction of the spiral arteries. This menstrual shedding is prevented in early pregnancy by progesterone from the corpus luteum and later the placenta. In later pregnancy the placenta is likely held in place by the intraamniotic pressure. The contents of the uterus prevent shortening of the myometrial muscle during contractions, at least while the cervix is closed. With expulsion of the contents, the myometrium contracts and shortens markedly (from an average uterine diameter of 21 cm to 5 cm)(2). The placenta cannot contract. The resulting stress tears the decidua, and the placenta is expelled. The best evidence for this shearing hypothesis is that the normal separation of the placenta occurs too rapidly with delivery to be hormonally mediated.
The key role of the decidua had been suspected since the early 18th century. Monro described his dissection of the pregnant uterus “I observed the smooth tense chorion from which the fungous substance separated most easily, and it did likewise from the placenta. (Edin Med 1734)”.(3)This “fungous substance” would later be termed decidua. The chorion, both basal beneath the placenta and that of the membranes, is very firmly adherent to the decidua, and even after delivery in the laboratory the layers cannot be cleanly separated. A consequence of the shearing of the decidua is that the spiral arteries and endometrial veins that traverse it must be torn apart with placental separation.
The details of normal placental separation have been described in frozen sagittal sections of rhesus monkeys in labor (4). The study found that the separation of the placenta begins in the second stage of labor, meaning as the infant is descending in the pelvis. Interval radiographs of placental separation in breech and anencephalic human deliveries demonstrated that 17 started at the lower (cervical side) edge of the placenta, and only three commenced centrally (5). In 20 cases radio-opaque dye was injected into the umbilical artery or vein after cord clamping in breech deliveries prior to delivery of the head. In 18 timed cases, placental separation was complete within 5 minutes of delivery of the infant, and in 6 of those is was complete at the end of the second stage of labor. The authors observed: “Study of the soft tissue outlines of the uterus emphasizes that retraction is a process which occurs in all planes, affecting the circumference, width and depth of the uterus as well as its length. At any one time, however, one part of the organ may be more affected than another, … retraction in mainly one plane may persist until complete expulsion of the fetus …ultimately uniform retraction takes place.” “There were 3 cases in which placental separation occurred before there was evidence of significant uterine retraction. In the remainder, retraction appeared to precede separation.” A plausible hypothesis to explain the three exceptions is that local contraction of the lower uterine segment in the process of cervical dilatation and fetal descent shortens sufficiently to shear the overlying decidua and initiate a hemorrhage that dissected the placenta off the uterus.
More recent studies of placental separation have used ultrasound to demonstrate the contraction of the uterus and consequent separation of the placenta in the third stage of labor. A color Doppler flow study in 55 women found that in the third stage of labor, blood flow stopped at a median of 2.5 (0-230) seconds after delivery with median latent phase of 91 (2-367) seconds and detachment phase of 39 (15-250) seconds(6). The separation in the vast majority of cases starts at the caudal pole of the placenta and detachment may occur in phases that may prolong the detachment. Another ultrasound study of 101 women confirmed that separation usually starts at one pole, most often the lower, and is usually multiphasic with separation and the thickening of the adjacent myometrium occurring together(7). In fundal placentas, the fundal portion usually separates last. All of these studies suggest that placental separation is relatively rapid with expulsion of the fetus, and starts in a focus, usually related to the contraction of the lower uterine segment that appears to be pulled cervically.
A different concept of placental separation was suggested by in vitro study of the placenta(8). The author argued that the Bernoulli theorem implies that the intervillous blood flow should reduce blood pressure in the intervillous space compared to the amniotic fluid. Decreased intervillous flow would then result in a higher pressure lifting the placenta upward. This is a very simplified model and there is no evidence that it actually occurs, but it is an interesting idea.
Placenta Accreta
If the decidua is absent due to thinning or scarring of the endometrium, then the placenta remains adherent unable to separate from the uterus. This condition is known as placenta accreta. In 1937, a paper by Irving and Hertig reviewed the microscopic pathology of the placenta accreta, as well as a review of the early literature(9). They cited the first case report from 88 AD in which placenta accreta occurred with placenta previa in a multiparous patient (Fig 1).

Fig 1: A copy of the 1614 paper by Plater of an 88 AD case of accreta, previa cited in Irving and Hertig’s 1937 review.
Clinical studies have repeatedly correlated placenta accreta with placenta previa, high parity, previous adherent placenta, previous Cesarean section, curettage especially with fetal loss or multiple curettages, Asherman syndrome, postpartum endometritis, endometrial ablation, and leiomyomas(10-24).
These correlations can be explained as conditions predisposing to loss of endometrium. The highest risk is in mothers with multiple Cesarean sections and an implantation of a low-lying anterior placenta or placenta previa over the presumed incision site(17). Perhaps the imbrication of the suture line after Cesarean hysterotomy creates a large scar that does not allow regeneration of endometrium, but I have not seen this directly. Most placenta accreta have some areas of normal decidual attachment. I have not found a careful study of the endometrium beneath Cesarean scars in non-gravid uteri that would increase our understanding of placenta accreta. I also could not find sufficient evidence for a counter-hypothesis that over exuberant cytotrophoblast invasion is a cause of placenta accreta(25).
The conventional designations of accreta (on the surface), increta (within the myometrium) and percreta (through the myometrium) likely reflect the thinness of the myometrium underlying rather than actual invasion of placental villi (Fig 2).
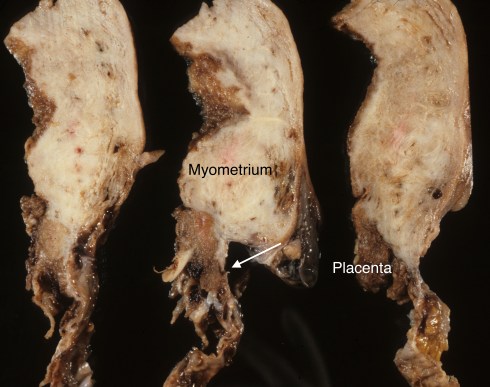
Fig 2a These cut sections from top to bottom of the anterior uterus in a mother with two previous Cesarean sections with placenta accrete that penetrated the thinned lower segment of the uterus. The tissue is fixed with the myometrium appearing white and the placenta brown. The arrow points to an area in which the placenta is against the serosal surface (placenta percreta).
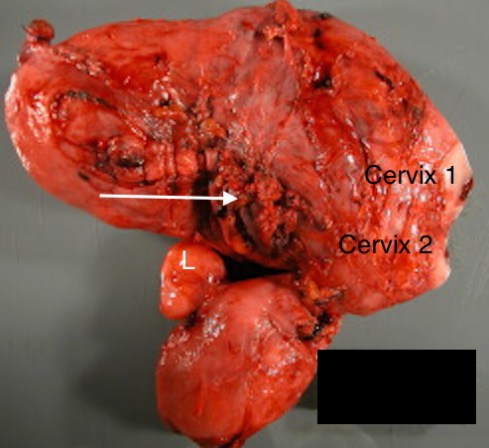
Fig 2b: This is a didelphic uterus with two horns each with its cervix. The arrow points to an area of placenta at the serosal surface of the gravid uterus (placenta percreta) where there had been a classic Cesarean section scar. “L” marks a leiomyoma.
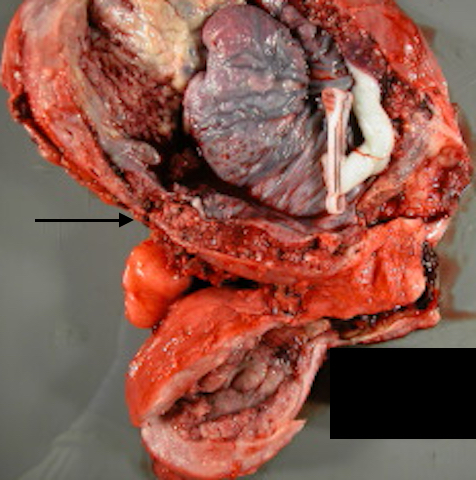
Fig 2c: The same didelphic uterus now opened showing the placenta accreta and the thinned myometrial wall at the arrow.
The outcome of large areas of placenta accreta is massive post partum hemorrhage that may require Cesarean hysterectomy or endovascular occlusion of major arteries(26). In normal separation, uterine contraction after expulsion of the placenta seems to stem the hemorrhage. The importance of uterine contraction is confirmed from two lines of clinical evidence. In placenta previa the placenta separates with a resultant hemorrhage despite an extensive decidual attachement (Fig 3).
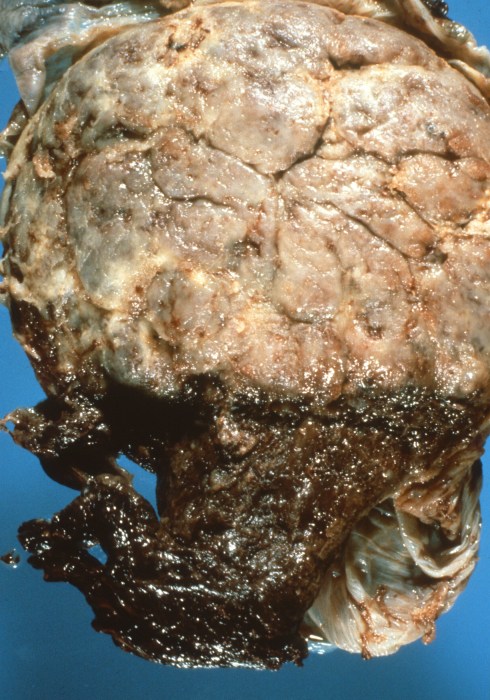
Fig 3: This is the maternal surface of a placenta that was over the cervical os (placenta previa) and was beginning to be delivered through that os with maternal hemorrhage. The red surface is the portion coned into the cervix. The pale portion separated normally after Cesarean delivery and shows a normal smooth maternal floor.
The infant is still inside the uterine cavity preventing contraction. The only effective treatment once the os has dilated is to remove the infant. Of course, today antepartum ultrasound and anticipated in-hospital delivery prevents most emergency Cesarean sections for placenta previa. The other line of evidence is that gravid uteri removed for uterine atony or severe unexplained postpartum hemorrhage have normal decidual separation, but do not contract and continue to hemorrhage after delivery. These atonic placentas may have varied etiology, and more than just not contracting may be the problem. I have seen such uteri with extensive plasma cell infiltration, with angiomatous hypervascularity and with extensive adenomyosis and embolized fetal squames from amniotic fluid (Fig 4).
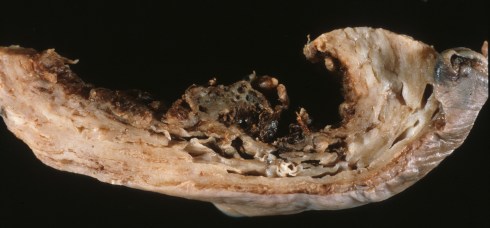
Fig 4a: This slice of a placenta with postpartum hemorrhage demonstrates marked hypervascularity both in a dark area of decidua and throughout the underlying pale myometrium.
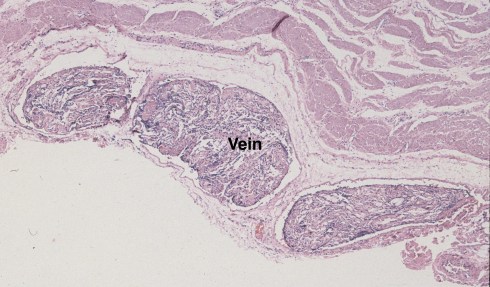
Fig 4b: This photograph from a uterus with atony and postpartum hemorrhage demonstrates a serosal vein packed with vernix as evidence of at least some amniotic fluid embolism. (H&E, 4x)
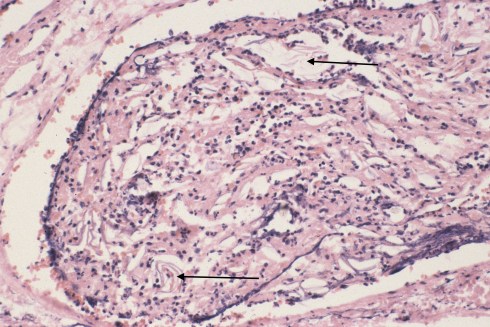
Fig 4c: This higher magnification of the vein shows the scattered fetal squames (shed squamous cells from the skin) with the arrows pointing to a few of the clusters. (H&E 20x)
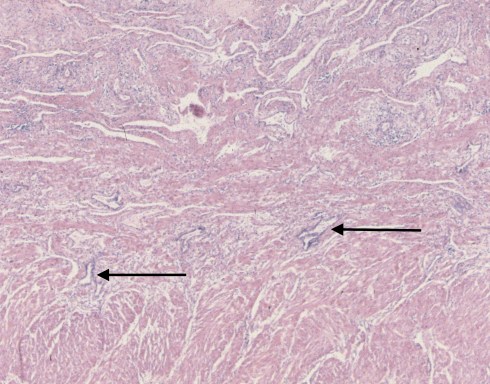
Fig 4d: From the same uterus as in 3b and 3c, the myometrium demonstrated areas of endometrial stroma with chronic inflammation, and with endometrial glands (arrows) deep in the endometrium. Whether this adenomyosis or the amniotic fluid embolism was important in the hemorrhage is unknown. (H&E)
The hemorrhage in placenta accreta defies the expectation that myometrial contraction will stem the bleeding, as efforts to contract the uterus do not stop the hemorrhage even after the placenta has been physically removed. With placenta accreta, the gravid uterus received in pathology may be thick and contracted, yet the continuing hemorrhage had required the Cesarean hysterectomy. The cause of this massive hemorrhage even following complete removal of the adherent placenta has not been fully explained. The lack of a remaining decidua is the most obvious pathological finding to suspect as in some way the cause of continued hemorrhage. In all gravid uteri, the spiral arteries supplying the placenta have had their media destroyed by trophoblast, and therefore lack the first step in normal arterial hemostasis, i.e. arterial constriction. With placenta accreta, two proposed hypothesis for the hemorrhage are 1) the loss of the thrombogenic properties of decidua that are needed to stop hemorrhage in the trophoblastic invaded vessels, and 2) the interference of myometrial contraction around the vessels caused by the deeper invasion of cytotrophoblast due to the lack of a decidua (27). There is also some evidence that trophoblast invasion of myometrial arteries may be abnormal with placenta accreta(10, 28)which could play an undefined role in the hemorrhage. A better understanding of this failed hemostasis might suggest local treatments to stop the hemorrhage without hysterectomy or endoscopic occlusion of large arteries.
Pathology of placenta accreta
I recall as a resident in obstetrics being told to document the completeness of the maternal surface of the placenta. I don’t think I was the only one who was somewhat clueless as to what to look for. As a pathologist, I am aware now that more often than not the surface is partially disrupted and except for large pieces of missing placenta, the completeness of the maternal surface cannot always be reliably determined. The problem can be demonstrated by cutting a small piece of placenta away from the maternal surface, and then re-apposing the edges. The missing chunk is inconspicuous. An extensively disrupted maternal placental surface is likely due the forceful removal of a placenta with placenta accreta (Fig 5).
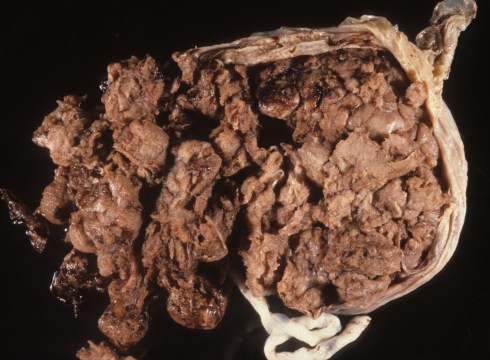
Fig 5a: This is the roughened incomplete maternal surface form a complete placenta accreta
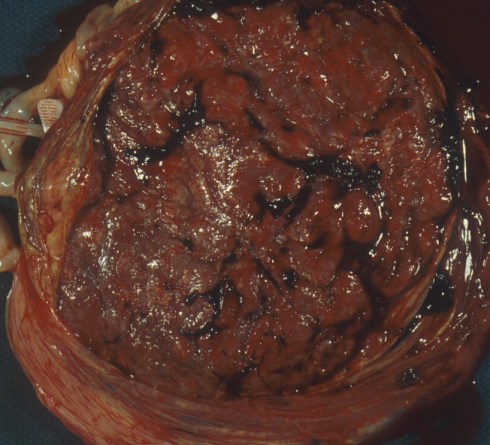
Fig 5b: This is a smooth normal maternal surface.
The examination of the hysterectomy is diagnostically definitive, and the placenta or portions may still be attached (Fig 6).
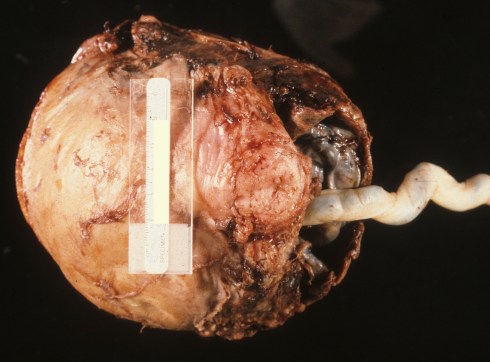
Fig 6a: The infant was delivered through a Cesarean incision and the protruding cord can still be seen, as the placenta could not be separated.
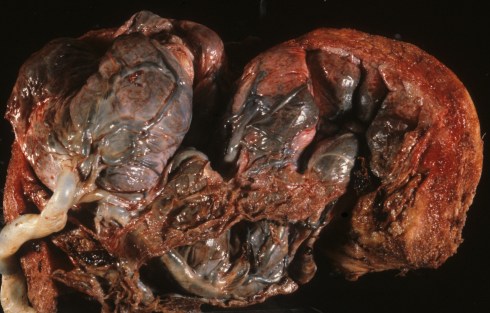
Fig 6b: The incised uterus demonstrates adherent placenta on almost the entire uterine surface (placenta membranacea). There was no decidua over the uterine surface.
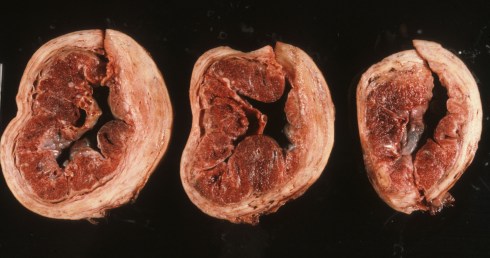
Fig 6c: The same uterus in cross section demonstrates the completely attached placenta. The myometrium is pale and the placenta red.

Fig 6d: This bisected uterus shows an attached placenta over the lower uterine segment that is also over the internal os of the cervix (placenta previa).

Fig 6e: This close up photograph better demonstrates the placenta previa portion of this placental accreta.
If the placenta has been removed, finding the placental bed is not easy, but extensive, systematic samples for histology should demonstrate areas of absent endometrium with basal cytotropblast implantation. In the normally attached placenta, the histology of the maternal surface demonstrates the attachment of the cytotrophoblastic basal plate onto the decidua. With placenta accreta, full thickness samples of the basal plate show direct attachmet to the myometrium that has been torn free, or no basal plate because it remains attached to the uterus (Fig 7).
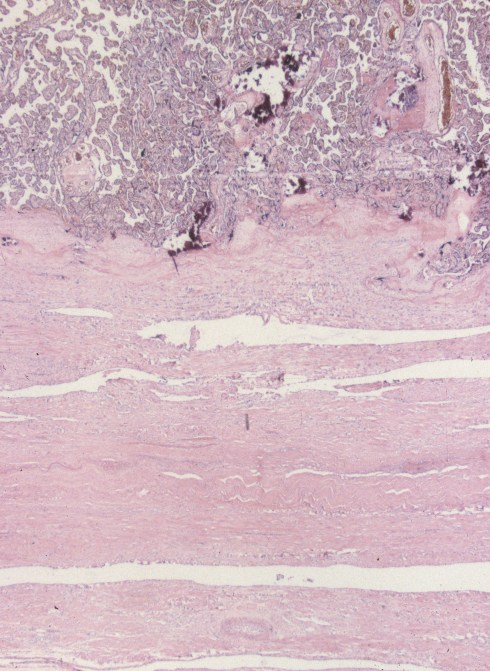
Fig 7a: This is a low magnification of the placenta on top with a thick chorionic base sitting over a relatively thin myometrium that ends at the serosa. This is from the same placenta as in Fig 5a-c. (H&E)
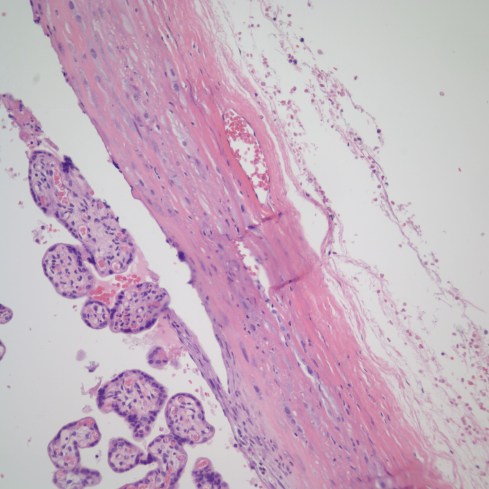
Fig 7b: The placenta in this image has a basal chorion implanted directly on a layer of serosa. This is the same placenta as in Fig 5d-e. (H&E, 20x)
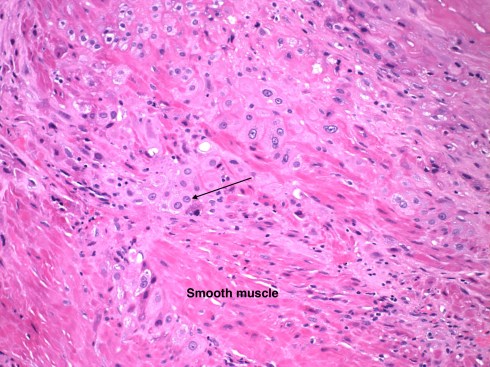
Fig 7c: This high power demonstrates the cytotrophblast cells (arrow points to a typical cell) of the chorionic plate embedding on and infiltrating into the very pink (eosinophilic) smooth muscle cells of the myometrium in a placenta accrete uterus. (H&E, 20x)
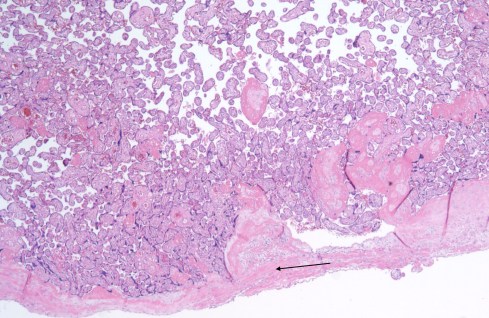
Fig 7d: This microphotograph is from the base of the placenta accreta in which the placenta was removed from the uterus before hysteretomy. The base of the placenta sits directly on the smooth muscle of the myometrium (arrow). (H&E, 4x)
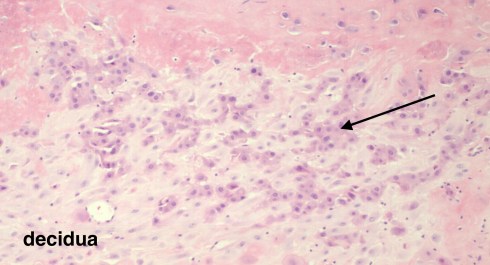
Fig 7e: This is the normal basal plate of the placenta with the cytotrophoblast (arrow) implanting on the pale cells of the decidualized endometrial stroma. (H&E, 20x)
In both the normal placenta and placenta accreta multiple sections of the maternal base are often needed to make a secure diagnosis. Finding samples with a basal decidual attachment does not rule out placenta accreta because 1) not all the placenta is necessarily abnormally attached and 2) the missing diagnostic areas of the placenta may still be in the uterus.
A perplexing finding for the pathologist is the incidental discovery of basal adherent myometrium (Fig 8).

Fig 8: The base of this placenta shows an area of incidental attached myometrium (arrow). (H&E, 10x)
This is anatomically a small area of placenta accreta. The true extent is unknown from a small sample. One study with routine sampling of the placenta, found that the finding portended a small increased risk of retained placenta in subsequent pregnancies(29). Another study found a higher incidence of adherent myometrium using a more extensive sampling of the placenta, but the lesion did not show an increased association with the risk factors for placenta accreta(30). Logically, there must be some line between partial placenta accreta that physically interferes with separation of the placenta and results in maternal hemorrhage and these small areas of accreta. The significance of these incidentally found areas of placenta accreta is not completely resolved(31). A definitive prospective study has not been done.
References:
- The Tortula. Philadelphia: University of Pennsylvania Press; 2001.
- Deyer TW, Ashton-Miller JA, Van Baren PM, Pearlman MD. Myometrial contractile strain at uteroplacental separation during parturition. Am J Obstet Gynecol. 2000;183(1):156-9.
- Boyd JD, Hamilton WJ. The Human Placenta. Cambridge: W. Heffer & Sons LTD; 1970. p. 209.
- Danforth DN, Graham RJ, Ivy AC. The functional anatomy of labor as revealed by frozen sagittal sections in the macacus rhesus monkey. Surg Gyn Obstet. 1942;74:188-203.
- Macpherson J. A radiological study of the placental stage of labour. J Obstet Gynaecol Br Empire. 1956;63:321-30.
- Krapp M, Katalinic A, Smrcek J, Geipel A, Berg C, Germer U, et al. Study of the third stage of labor by color Doppler sonography. Arch Gynecol Obstet. 2003;267(4):202-4.
- Herman A, Zimerman A, Arieli S, Tovbin Y, Bezer M, Bukovsky I, et al. Down-up sequential separation of the placenta. Ultrasound Obstet Gynecol. 2002;19(3):278-81.
- Groeber WR. Antiabruption dynamics of the intervillous circulation in an artificial uterus. Am J Obstet Gynecol. 1966;95(5):640-7.
- Irving F, Hertig A. A study of placenta accreta. Surg Gynecol Obstet. 1937;64:178-200.
- Tantbirojn P, Crum CP, Parast MM. Pathophysiology of placenta creta: the role of decidua and extravillous trophoblast. Placenta. 2008;29(7):639-45.
- Deaton JL, Maier D, Andreoli J, Jr. Spontaneous uterine rupture during pregnancy after treatment of Asherman’s syndrome. Am J Obstet Gynecol. 1989;160(5 Pt 1):1053-4.
- Luke RK, Sharpe JW, Greene RR. Placenta accreta: the adherent or invasive placenta. Am J Obstet Gynecol. 1966;95(5):660-8.
- Clark SL, Koonings PP, Phelan JP. Placenta previa/accreta and prior cesarean section. Obstet Gynecol. 1985;66(1):89-92.
- Beavis ELG. Placenta accreta a report of six cases. Aust NZJ Obstet Gynaec. 1961;1:161-7.
- Kistner RW, Hertig AT, Reid DE. Simultaneously occurring placenta previa and placenta accreta. Surg Gynecol Obstet. 1952;94(2):141-51.
- Brock JA. The etiology of placenta previa accreta. J Reprod Med. 1969;11:111-5.
- Miller DA, Chollet JA, Goodwin TM. Clinical risk factors for placenta previa-placenta accreta. Am J Obstet Gynecol. 1997;177(1):210-4.
- Van Thiel DH, Grodin JM, Ross GT, Lipsett MB. Partial placenta accreta in pregnancies following chemotherapy for gestational trophoblastic neoplasms. Am J Obstet Gynecol. 1972;112(1):54-8.
- Morison JE. Placenta accreta. A clinicopathologic review of 67 cases. Obstet Gynecol Annu. 1978;7:107-23.
- El-Miligy M, Gordon A, Houston G. Focal myometrial defect and partial placenta accreta in a pregnancy following bilateral uterine artery embolization. J Vasc Interv Radiol. 2007;18(6):789-91.
- Al-Serehi A, Mhoyan A, Brown M, Benirschke K, Hull A, Pretorius DH. Placenta accreta: an association with fibroids and Asherman syndrome. J Ultrasound Med. 2008;27(11):1623-8.
- Barss P, Misch KA. Endemic placenta accreta in a population of remote villagers in Papua New Guinea. Br J Obstet Gynaecol. 1990;97(2):167-74.
- Hoffman MK, Sciscione AC. Placenta accreta and intrauterine fetal death in a woman with prior endometrial ablation: a case report. J Reprod Med. 2004;49(5):384-6.
- Friedman A, DeFazio J, DeCherney A. Severe obstetric complications after aggressive treatment of Asherman syndrome. Obstet Gynecol. 1986;67(6):864-7.
- Cramer SF. Invasion and destruction of neoplastic tissue by “normal” tissue. Pediatr Pathol. 1987;7(4):473-5.
- Goh WA, Zalud I. Placenta accreta: diagnosis, management and the molecular biology of the morbidly adherent placenta. J Matern Fetal Neonatal Med. 2016;29(11):1795-800.
- Earl U, Bulmer JN, Briones A. Placenta accreta: an immunohistological study of trophoblast populations. Placenta. 1987;8(3):273-82.
- Khong TY, Robertson WB. Placenta creta and placenta praevia creta. Placenta. 1987;8(4):399-409.
- Jacques SM, Qureshi F, Trent VS, Ramirez NC. Placenta accreta: mild cases diagnosed by placental examination. Int J Gynecol Pathol. 1996;15(1):28-33.
- Khong TY, Werger AC. Myometrial fibers in the placental basal plate can confirm but do not necessarily indicate clinical placenta accreta. Am J Clin Pathol. 2001;116(5):703-8.
- Stanek J, Drummond Z. Occult placenta accreta: the missing link in the diagnosis of abnormal placentation. Pediatr Dev Pathol. 2007;10(4):266-73.