Meconium
Meconium is the material collected in the fetal colon prior to birth. The material is green and readily seen in the colon during the autopsy of a stillborn infant. The components include bile secreted from the liver, swallowed fetal amniotic fluid with its shed skin cells and lanugo hair, and proteins and cells contributed by the gastrointestinal tract. The former contributes the green yellow pigments biliverdin and bilirubin from hemoglobin digestion. The meconium is usually sterile, although in previable gestation infants with chorioamnionitis, neutrophils and even bacteria may be seen in the colon at autopsy. Most infants, approximately 75%, do not pass visible meconium until after birth, and this may be due to an active suppression of the anal sphincter by signals such as corticotrophin releasing factor1.
There is some evidence that the fetus passes meconium in utero very early in gestation. Green amniotic fluid is frequently obtained with mid gestation amniocentesis, however this is most likely due to bile pigments formed directly from the breakdown of hemoglobin that has diffused into the fluid2-4. One study relied on meconium corpuscles to identify aspirated meconium in early gestation, but this is suspect since such corpuscles are apoptotic bodies, and apoptotic cells are commonly found in the lungs of previable gestation infants without evidence of other material suggestive of meconium2,5. Meconium passage does occur in many (up to 20%) of mature fetuses for no discernable reason and usually without serious consequence. However, there are exceptions.
The first exception is that meconium can be a marker of fetal asphyxia. Severe acute asphyxia in the experimental model results in passage of meconium 6. Such acute severe asphyxia is relatively uncommon in human labor, but occurs for example in cord prolapse or uterine rupture. Despite the relatively much more common benign passage of meconium, some studies show an association of meconium passage with fetal asphyxia or hypoxic/ischemic brain injury. Conversely, infants with acute asphyxia often demonstrate meconium passage. (See short case #1)
The second exception is meconium aspiration syndrome. Intrauterine asphyxia causes fetal gasping. If there is meconium in the amniotic fluid, whether from asphyxia or otherwise, the predictable result is that amniotic fluid with meconium will also be forcibly aspirated into the lung. This potentially leads to the meconium aspiration syndrome, a topic reviewed in an article aptly entitled “Meconium aspiration syndrome: reflections on a murky subject” 7,8. Asphyxia and shock lung complicate attributing the syndrome of respiratory distress and pulmonary hypertension to the meconium per se.
The pathologist’s contribution to understanding meconium aspiration falls into two categories. First is observation of what is anatomically found in the lungs
of stillborn and newborn infants. Second is the observation of the effects of meconium on tissue.
In almost all stillborn lungs there are some squames, the shed surface epithelial cells of the skin, in the lung. With chorioamnionitis there will be neutrophils and sometimes bacteria seen in the airways. The fetus in utero has shallow respiration that results in some aspiration of amniotic fluid content, including meconium if it is present. If there is deep gasping with asphyxia the content of the amniotic fluid will flood the lungs including invisible substances such as mediators of inflammation. Scattered particles of meconium (squames and pigmented protein) or of vernix in the lung are visible markers of either shallow or deep inspiration. In the absence of chorioamnionitis, they are not associated with inflammation. This does not prove that meconium could not cause lung disease, but at least it is not inflammatory. Some autopsies of stillborn infants demonstrate large quantities of viscous meconium distending and plugging the proximal airways. It is reasonable to speculate that had such an infant lived, this viscous material would have interfered with ventilation (see short case 1). It also seems unlikely that this material could be effectively suctioned out of the airway postpartum.
Meconium would have to be “thick” to physically impede respiration and cause air trapping. Some studies have shown an association with birth complications if there is early thick meconium associated with early passage or with fetal heart rate changes particularly decreased variablity9,10. The distinction of thin versus thick is usually just a visual assessment, although a “meconiumcrit” has been suggested obtained like a hematocrit by centrifuging a tube of stained amniotic fluid11. These reports do not explain the origin of the thickness of the meconium. I don’t know of any evidence that the quantity of meconium passed varies with fetal distress. More likely thick meconium occurs due to oligohydramnios, either because the membrane rupture has left scant fluid behind in the intrauterine cavity or because there was preexistent oligohydramnios perhaps related to chronic intrauterine stress. Autopsies of asphyxiated infants often show some filling of distal bronchi with meconium, and this may be a factor in respiratory distress in some infants that are born alive12.
Even sterile meconium might be expected to elicit acute inflammation in the lung since it would be expected to have bile and digestive enzymes that could cause tissue damage. Naturally, meconium in the colon does not elicit inflammation, but this lack can be attributed to a specialized mucosa. More telling is the frequent lack of inflammation in fetal membranes with pigment macrophages that are evidence of deep penetration of meconium. Yet even here it could be argued that the lack of vascularity and the effectiveness of the macrophages prevent inflammation. The presence of meconium in the lung of stillborn infants without inflammation does not prove that given more time before death that tissue injury would not have occurred. Yet some autopsies of neonates also demonstrate meconium without inflammation (Fig 1). Despite some experimental evidence of lung injury, the human pathologic evidence is against significant tissue damage from aspirated meconium.
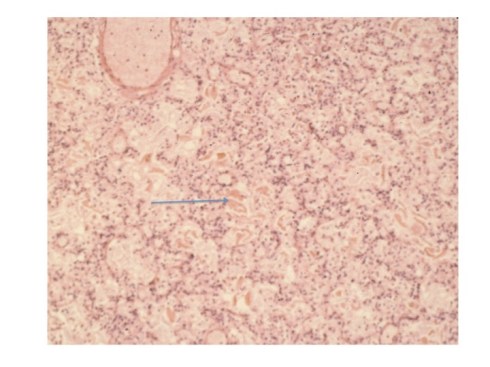
Fig 1: The arrow points to pigmented protein in the airway of an infant who rupture of membranes with thick meconium for 12 hours prior to delivery. At delivery he had severe arthrogryposis and pulmonary hypoplasia and could not be resuscitated. The airways are full of meconium without inflammation. (H&E, 40x)
There is an association of meconium and chorioamnionitis. I see this association routinely, often with neutrophils confined to the subchorionic intervillous fibrinoid (early maternal response to chorioamnionitis) in the context of failure of fetal progress in labor, but this association does not prove cause, but only association, as meconium also occurs without inflammation. As additional proof of a lack of causation, experimental injection of meconium into the amniotic sac of the rabbit does not cause inflammation13.
Meconium appears to cause inflammation with perforation of the bowel usually in the context of the viscous meconium ileus in cystic fibrosis. The ruptured bowel content is accompanied by peritoneal inflammation and calcification likely from digestion of lipids. This model however is significantly different from injecting normal meconium into the peritoneal cavity of the abdomen. The rupture is presumed caused by ischemic tissue necrosis of the bowel, a source of inflammation itself. In addition, the spilled lumenal content is proximal to the colon and may have more digestive enzymes and bile not subjected to changes in the terminal ileum and colon. This exception does not prove that meconium per se induces inflammation.
These observations suggest that meconium alone does not cause inflammatory lung disease. The lack of efficacy of infusing fluid into the amniotic cavity of women with thick meconium in order to thin the meconium is evidence that physical airway obstruction by meconium is not the cause of meconium aspiration syndrome, although an accompanying editorial suggests that the meconium aspiration could have occurred prior to the amnioinfusion14,15. The lack of efficacy in many cases of tracheal suction in preventing the syndrome is also evidence against a direct role for meconium in producing meconium aspiration syndrome, although as with amnioinfusion, a postulated deep aspiration of meconium in utero may defeat the ability of physicians to suction meconium out of the upper airway16,17 18,19. Overall, the evidence does suggest that fetal aspiration of meconium may be insufficient to establish meconium aspiration syndrome.
Another factor to consider in fetal exposure to meconium is duration. Pathologists are often asked to ascertain how long meconium has been in the amniotic fluid. A common assumption is that brown stained fluid is from aging of meconium. This is very unlikely as the brown color in human stool from the breakdown of bilirubin is due to bacterial modification of the bilirubin to stercobilin. At first it seems reasonable that meconium color might age similar to the pattern of blood breakdown in a bruise, but that notion breaks down with recalling that there is no hemosiderin that is protein bound iron, with a brownish red color, and the meconium is not being seen through the skin where dark brown deoxygenated hemoglobin looks blue (cyanosis). Brown meconium may imply hemoglobin not aging. On serial amnioscopy meconium remains green for days in the amniotic fluid in one case for 21 days20.
The pathologist usually tries to estimate the duration of meconium exposure in the amniotic cavity on the depth of pigment macrophages in the membranes. The concept is that bile pigment in the meconium is transported across the amnion and that as it diffuses macrophages will ingest it at progressively deeper levels, basically amnion then chorion (Fig 2). In some fetal membranes meconium can be seen on the surface of the amnion or staining the surface epithelium without pigment in macrophages. In others, pigment macrophages are in the amnion connective tissue or the chorion connective tissue or the decidua. This concept was tested experimentally by applying meconium at different concentrations (5-20%) to pieces of placenta in culture21. This was hardly physiologic but has served as a useful estimate for pathologists. The study found no effect of meconium concentration, but that seems unlikely from experience, and the threshold for recognizing the pigment under the microscope may vary among individuals, but they found that it took approximately 2 hours to see pigment in the amnion connective tissue and 3 hours in the chorion connective tissue. They did not have decidua beneath the membranes nor umbilical cord, and therefore could not comment on duration of pigment macrophages in those locations.
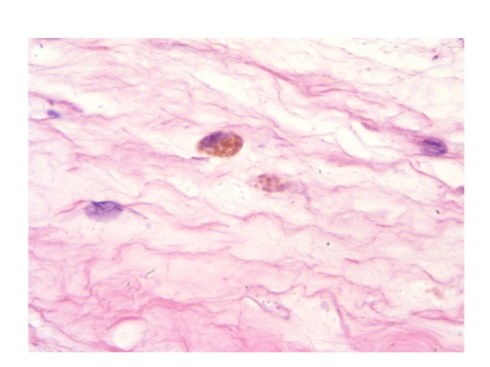
Fig 2: The pigmented macrophage in the center is within the chorion connective tissue of the fetal membranes. The pigment is in granules suggestive of accumulation in lysosomal granules. This may be of longer duration than pigment that diffusely stains the macrophage cytoplasm. (H&E, 100X)
A subsequent study using much more dilute meconium has argued that the timing takes much longer, as the study found few meconium macrophages in the amnion until 24 hours, and never found them in the chorion layer even at 48 hours22. They primarily accessed the depth of free meconium, but the paper is unclear how this was identified. This paper does not correlate with my clinical experience. Yet another approach was a study that looked at meconium macrophages in women who initially had clear amniotic fluid, but had subsequent passage of moderate to thick meconium noted23. This study was more concerned with showing a lack of a quantitative relationship based on number of pigmented macrophages per area of membrane and did not consider the problem of depth of the pigment macrophages. The study is complicated by the presence of meconium passed often with minimal fluid present and thus intense concentration. They comment that: “Our observation that meconium was present in the majority of the placental tissues analysed within an interval as short as 10 minutes is puzzling.” It is, but they provided no further detail. Currently, any estimate of the duration of meconium based on the pathology of the placenta has a high degree of uncertainty likely due to concentration differences of meconium and observer variability, as well as a lack of any in vivo experimental data in primates.
Since other sources of pigment occur in the amniotic fluid, the identification of meconium pigment is made by confirming the gross green staining of the placenta and sometimes by excluding hemosiderin by performing a Prussian Blue reaction to stain the tissue for hemosiderin bound iron. Analogous to the effect of bilirubin lights in the newborn infant, fluorescent lights in the room may substantially reduce the pigment intensity on the microscope slide24
Some authors have turned the association of meconium and fetal distress around and have argued that meconium can cause fetal asphyxia by injury to umbilical cord vessels25-27. There are two types of evidence for this contention. One is the association of meconium stained umbilical cords with vascular smooth muscle necrosis in infants with neurologic injury. The “meconium induced vascular necrosis” is a very distinctive lesion with necrotic muscle cells on the outer muscle layers and toward the umbilical cord surface (Fig3). Yet, it is equally plausible that this pattern is not from diffusion of meconium inward, but from necrosis in the areas of lowest oxygen levels in the umbilical cord vessels.
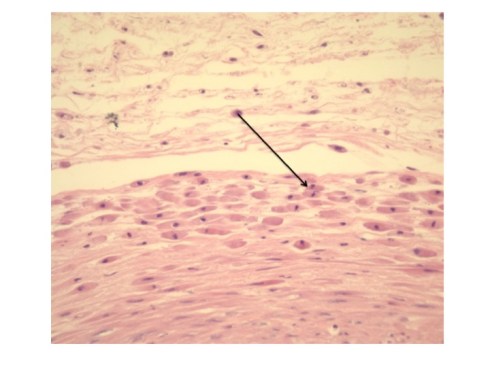
Fig 3: This is from an umbilical cord with meconium macrophages at the back of the arrow and superficial smooth muscle necrosis of the outer layer of the umbilical vein(H&E, 20x)
Another similar line of evidence proposes that meconium causes pulmonary arterial spasm and necrosis was associated with meconium aspiration based on three cases of distal pulmonary infarctions although 2 of the 3 did not have documented meconium passage in utero. The infarctions were attributed to meconium induced vasospasm, but this was only hypothesis28.
The second approach is to show meconium induced vascular injury in segments of the umbilical cord experimentally exposed to meconium29-32. Such studies were non-physiologic since meconium that was effective in causing vasoconstriction or causing vascular injury had direct contact with fetal vessels, (not to mention abnormal oxygen concentration) compared to meconium in the amniotic fluid which must penetrate an epithelial layer and a connective tissue of the umbilical cord and avoid phagocytic macrophages to reach a vessel wall. Meconium may have physiologic effects in the amniotic fluid such as depressing neutrophil function, but these effects in general have not been directly proven to cause fetal asphyxia33. The local effects of meconium are likely to be complex as meconium is a mixture of substances that must diffuse barriers in addition to macrophage scavenging and other tissue defenses. One can also not conflate meconium with bile, which has severe cell detergent properties since bile is likely modified in the fetal gastrointestinal tract. In attempting to explain umbilical cord ulceration in bowel atresias distal to the ampulla of Vater, the lack of other explanation, forced the conclusion that bile reflux into amniotic fluid caused the ulcers. There was no arterial necrosis until ulceration exposed the arteries directly to the amniotic fluid and bile. The case for meconium as a cause of asphyxia remains unproven.
References:
- Lakshmanan J, Oyachi N, Ahanya SA, Liu G, Mazdak M, Ross MG. Corticotropin-releasing factor inhibition of sheep fetal colonic contractility: mechanisms to prevent meconium passage in utero. Am J Obstet Gynecol 2007;196:357 e1-7.
- Mortensen E, Kearney MS. Meconium aspiration in the midtrimester fetus: an autopsy study. Pediatr Dev Pathol 2009;12:438-42.
- King CR, Prescott G, Pernoll M. Significance of meconium in midtrimester diagnostic amniocentesis. Am J Obstet Gynecol 1978;132:667-9.
- Abramovich DR, Gray ES. Physiologic fetal defecation in midpregnancy. Obstet Gynecol 1982;60:294-6.
- Williams L, Bell L. An ultrastructural study of meconium corpuscles in human foetal colon. Anatomy and embryology 1985;171:373-6.
- Block MF, Kallenberger DA, Kern JD, Nepveux RD. In utero meconium aspiration by the baboon fetus. Obstet Gynecol 1981;57:37-40.
- Katz VL, Bowes WA, Jr. Meconium aspiration syndrome: reflections on a murky subject. Am J Obstet Gynecol 1992;166:171-83.
- Hernandez C, Little BB, Dax JS, Gilstrap LC, 3rd, Rosenfeld CR. Prediction of the severity of meconium aspiration syndrome. Am J Obstet Gynecol 1993;169:61-70.
- Meis PJ, Hall M, 3rd, Marshall JR, Hobel CJ. Meconium passage: a new classification for risk assessment during labor. Am J Obstet Gynecol 1978;131:509-13.
- Xu H, Mas-Calvet M, Wei SQ, Luo ZC, Fraser WD. Abnormal fetal heart rate tracing patterns in patients with thick meconium staining of the amniotic fluid: association with perinatal outcomes. Am J Obstet Gynecol 2009;200:283 e1-7.
- Trimmer K, Gilstrap L. Meconiumcrit and Birth Asphyxia. American Journal of Obstetrics and Gynecology 1991;165:1010-3.
- Brown BL, Gleicher N. Intrauterine meconium aspiration. Obstet Gynecol 1981;57:26-9.
- Lauweryns J, Bernat R, Lerut A, Detournay G. Intrauterine pneumonia. An experimental study. Biol Neonate 1973;22:301-18.
- Fraser WD, Hofmeyr J, Lede R, et al. Amnioinfusion for the prevention of the meconium aspiration syndrome. N Engl J Med 2005;353:909-17.
- Ross MG. Meconium aspiration syndrome–more than intrapartum meconium. N Engl J Med 2005;353:946-8.
- Falciglia HS. Failure to prevent meconium aspiration syndrome. Obstet Gynecol 1988;71:349-53.
- Dooley SL, Pesavento DJ, Depp R, Socol ML, Tamura RK, Wiringa KS. Meconium below the vocal cords at delivery: correlation with intrapartum events. Am J Obstet Gynecol 1985;153:767-70.
- Davis RO, Philips JBd, Harris BA, Jr., Wilson ER, Huddleston JF. Fatal meconium aspiration syndrome occurring despite airway management considered appropriate. Am J Obstet Gynecol 1985;151:731-6.
- Manning FA, Schreiber J, Turkel SB. Fatal meconium aspiration “in utero”: a case report. Am J Obstet Gynecol 1978;132:111-3.
- Barham KA. Amnioscopy, meconium and fetal well-being. J Obstet Gynaecol Br Commonw 1969;76:412-8.
- Miller PW, Coen RW, Benirschke K. Dating the time interval from meconium passage to birth. Obstet Gynecol 1985;66:459-62.
- Funai EF, Labowsky AT, Drewes CE, Kliman HJ. Timing of fetal meconium absorption by amnionic macrophages. Am J Perinatol 2009;26:93-7.
- Incerti M, Locatelli A, Consonni S, Bono F, Leone BE, Ghidini A. Can placental histology establish the timing of meconium passage during labor? Acta Obstet Gynecol Scand 2011;90:863-8.
- Morhaime JL, Park K, Benirschke K, Baergen RN. Disappearance of meconium pigment in placental specimens on exposure to light. Arch Pathol Lab Med 2003;127:711-4.
- Naeye R. Can meconium in the amniotic fluid injure the fetal brain. Obstet Gynecol 1995;86:720-4.
- Sienko A, Altshuler G. Meconium-induced umbilical vascular necrosis in abortuses and fetuses: a histopathologic study for cytokines. Obstet Gynecol 1999;94:415-20.
- Burgess AM, Hutchins GM. Inflammation of the lungs, umbilical cord and placenta associated with meconium passage in utero. Review of 123 autopsied cases. Pathol Res Pract 1996;192:1121-8.
- Kearney MS. Chronic intrauterine meconium aspiration causes fetal lung infarcts, lung rupture, and meconium embolism. Pediatr Dev Pathol 1999;2:544-51.
- Kafkasli A, Belfort MA, Giannina G, Vedernikov YP, Schaffner DL, Popek EJ. Histopathologic effects of meconium on human umbilical artery and vein: in vitro study. J Matern Fetal Med 1997;6:356-61.
- Pickens J, Toubas PL, Hyde S, Altshuler G. In vitro model of human umbilical venous perfusion to study the effects of meconium staining of the umbilical cord. Biol Neonate 1995;67:100-8.
- Sepulveda WH, Gonzalez C, Cruz MA, Rudolph MI. Vasoconstrictive effect of bile acids on isolated human placental chorionic veins. Eur J Obstet Gynecol Reprod Biol 1991;42:211-5.
- Hyde S, Altshuler G. Need for placental and experimental pathologic examination to determine pathogenetic influences of chronically present intraamniotic meconium. Am J Obstet Gynecol 1996;174:1669-70.
- Clark P, Duff P. Inhibition of neutrophil oxidative burst and phagocytosis by meconium. Am J Obstet Gynecol 1995;173:1301-5.